Received: Wed 02, Jul 2025
Accepted: Fri 18, Jul 2025
Abstract
Acalculia is an acquired disorder of numerical skills whose assessment is not traditionally included in the three-stage protocol specific to awake glioma neurosurgery. Nevertheless, this disorder can significantly impact patient’s daily life. This review aims to describe the importance of assessing numerical and practical skills to optimize quality of life.
Ninety-eight articles from three databases were reviewed by at least two authors, selecting 17 for qualitative analysis (n = 297 patients). Most scored 75-100% on the CARE grid, with robustness classifying 10 as C, 3 as D, and 4 as B.
Among the 297 patients, 239 were preoperatively investigated for a potential acalculia with 8.8% (n = 21) showing numerical disorders. Postoperatively, among 132 patients assessed for an acalculia, 14.4% (n = 19) had numerical disorders. Of these, 42.1% (n = 8) were inaugural cases while 36.8% (n = 7) had no specific numerical skills assessment. Lastly, among 162 patients assessed at all three phases, 88.9% (n = 144) showed no postoperative numerical disorder. Then, this review highlights disparities in numerical assessment during awake neurosurgery: none of the 17 studies follows the same evaluation protocol, especially for numerical tasks.
These findings suggest assessing numerical skills during operative phases specific to awake surgery helps prevent the onset of postoperative acalculia. However, it is regrettable that these data cannot be completed and compared with the percentage of patients who performed some numerical tasks 90.6% (n = 269) without being screened for a postoperative acalculia 39.7% (n = 107). Finally, this analysis highlights significant disparities between protocols used to assess numerical skills.
Keywords
Glioma, acalculia, assessment, awake neurosurgery
1. Introduction
1.1. Gliomas and Awake Condition
Gliomas are infiltrative brain tumors that are estimated to represent 24.7% of primary tumors [1]. Among these are low-grade diffuse gliomas (LGDG), for which the first-line treatment is awake neurosurgery with pre- and intra-operative brain mapping. The awake condition is also used for certain high-grade tumors (HGDG) when the location allows it. It aims to preserve post-operative quality of life by optimizing resection while maintaining an “onco-functional balance” [2, 3]. It involves assessing the patient pre-, intra-, and post-operatively, using various tests, including cognitive tests such as those for language, to map the brain regions that need to be preserved during the resection and to evaluate potential post-operative deficits. However, while it is now clear that prioritizing the patient’s cognitive and language abilities is crucial, a deficit in numerical skills can also significantly impact their everyday life.
1.2. Acalculia in Gliomas
Acalculia is an acquired disorder of numerical abilities that occurs following brain injury and can be primary (due to a loss of numerical concepts), secondary (associated with other cognitive disorders: aphasic, memory, executive) or found in Gerstmann syndrome which is an association of four symptoms: digital skills disorders, digital agnosia, right-left disorientation and agraphia [4]. Moreover, numerical skills are not localized to a single area of the cortex but can be affected by lesions in the parietal, occipital, temporal, or even frontal and subcortical regions [4]. The evaluation of numerical disorders is not standardized across neurosurgery departments. It may involve a counting task [1-3, 5, 6], sometimes referred to as a numerical task but often considered more of an automatic linguistic process, an addition, subtraction, or multiplication task [2, 3, 7-12], or, in some cases, the use of a battery such as the number processing and calculation (NPC) battery [7]. Moreover, currently, the literature on the assessment of acalculia during neurosurgery is quite limited, and most of the evaluation protocols available focus primarily on language.
Given the proximity between the areas involved in numerical skills and the brain regions considered eloquent from a linguistic and cognitive perspective, as well as the impact that acalculia can have on a patient’s everyday life, it seems essential to consider these disorders, which should potentially be subject to systematic evaluation. Highlight the prevalence of acalculia during neurosurgery of gliomas would allow the systematic integration of numerical skills assessment into the three-step protocol specific to awake surgery.
2. Materials and Methods
We followed PRISMA recommendations to write the systematic review [13]. The review was registered on Prospero in July 2024 as Acalculia Assessment in Glioma Patients: A Systematic Review (ID: CRD42024471675).
2.1. Search Strategy
The search was firstly translated into keywords. The HeTOP (Health Terminology/Ontology Portal) platform was chosen to select the most appropriate Medical Subject Headings (MeSH) terms. Secondly, synonyms were identified so as not to exclude any terms for terminological reasons. A time filter from January 2000 to May 2024 was systematically applied. Articles were selected between February and July 2024. Table 1 summarizes the different search equations according to the databases.
Table. 1. Search equations according to
the selected databases.
|
DATA
BASES |
search
equation |
|
PubMed |
(((glioma) OR ("glial cell tumor")) OR
("low grade glioma")) AND (((mathematic) OR (acalculia) OR
(dyscalculia) OR ("Numerical concept") OR ("Numerical
skill") OR (Transcoding) OR ("Calculation ability") OR
("calculation skill") OR ("calculation task") OR
("Arithmetical ability") OR ("Arithmetical skill"))) AND
((("glioma surgery") OR ("Awake craniotomy") OR
("Awake surgery") OR ("Intraoperative electrical
stimulation") OR ("Direct electrical stimulation") OR
("Wakefulness") OR ("tumor surgery") OR ("brain surgery")
OR ("surgical resection"))) NOT ((child*) OR (pediatric) OR
(teenager)) AND (1997:2024[pdat])
|
|
Science Direct |
(glioma OR
"glial cell tumor") AND (acalculia OR dyscalculia OR
"numerical skill" OR "calculation task" OR
"arithmetical ability") AND ("brain surgery" OR
"surgical resection ») filter:1997-2024
|
|
Web of Science |
(((ALL=((glioma OR "glial cell tumor" OR
"low grade glioma"))) AND (ALL=((mathematic) OR (acalculus) OR
(dyscalculia) OR ("Numerical concept") OR ("Numerical
skill") OR (Transcoding) OR ("Calculation ability") OR
("calculation skill") OR ("calculation task") OR
("Arithmetical ability") OR ("Arithmetical skill") OR
("gerstmann syndrome")))) AND (ALL=((("glioma surgery")
OR ("Awake craniotomy") OR ("Awake surgery") OR
("Intraoperative electrical stimulation") OR ("Direct
electrical stimulation") OR ("Wakefulness") OR ("tumor
surgery") OR ("brain surgery") OR ("surgical
resection"))))) |
2.2. Eligibility Criteria
i) Patients with glioma (diffuse gliomas in adults according to the 2021 WHO classification [14]). All diffuse gliomas in adults were included to ensure no patient was excluded for terminological reasons. ii) Adult patients (>18 years old). iii) Numerical skills mentioned. iv) Case Reports or Original Articles only. v) Article written in French or English. vi) Publication between 2000 and 2024.
2.3. Article Selection
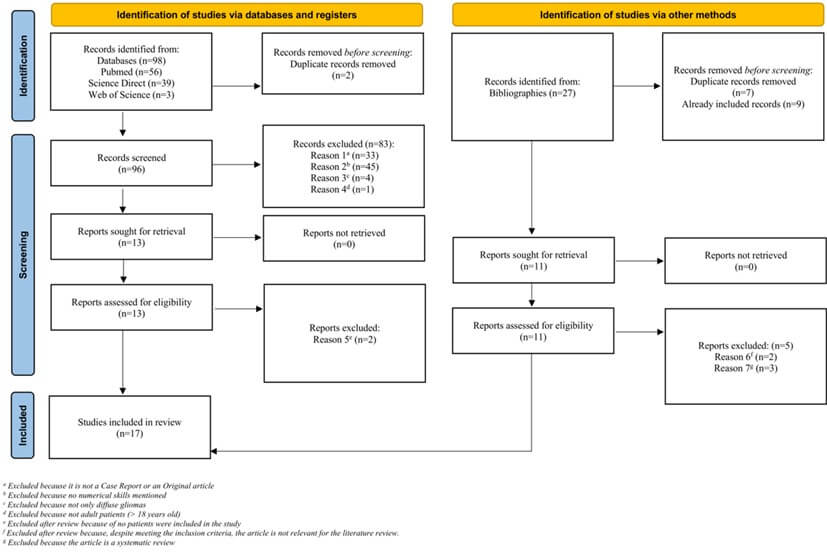
The selection of the articles has been achieved according to a process respecting the recommendations in force (PRISMA) [13]. Figure 1 summarizes the study selection process.
2.4. Data Extraction
A coding grid was designed to facilitate statistical processing. This grid was duplicated, and the data was extracted twice (EC and A-VO) in a double-blind process to minimize the risk of error during data retrieval.
2.5. Assessing the Quality of the Studies
This systematic review includes only case series or case studies. Consequently, the CARE (consensus-based clinical case reporting guideline) grid [15], designed to assess the completeness, transparency, and analysis of data in case reports, was selected and used. Initially, this grid has 30 items, but only 28 were selected because of two items which were not relevant to the study (timeline and patient perspectives). One point was assigned per item, and each score was then converted into a percentage (excellent quality above 90%, good quality between 75% and 89%, average quality between 50% and 74%, and poor quality below 50%). Moreover, to assess the robustness of all the studies, the article by Ebbels [16] serves as a reference. The study’s design and the number of participants are taken into account, categorizing them into one of four proposed models: A, B, C and D. Models A and B exhibit strong experimental control (model B involving a larger participant pool than model A). Conversely, models C and D demonstrate weaker experimental control (model D including more participants than model C). As a result, model B is deemed the most reliable, while model C is considered the least reliable.
2.6. Results of the Systematic Search
Initially, ninety-eight results were obtained from the search equations in three databases. Two duplicate records were removed before the analysis of titles and abstracts. Then, eighty-three articles were excluded by reading titles and abstracts because they did not meet the eligibility criteria. All the above steps were carried out under double-blind conditions to control for selection bias. Therefore, thirteen articles were read in their entirety and all bibliographies were analyzed. Eleven articles were included out of thirteen - two of them met the inclusion criteria but were purely theoretical without inclusion of patient, and therefore were excluded after reading. Six articles reviewed and found in bibliographies were also added. Therefore, seventeen articles were selected for the systematic literature review. Figure 1 shows all steps of the selection process, according to the PRISMA flowchart model.
Table. 2. Patient-related clinical data.
|
Author(s) |
No. of Patients (%)* |
Sex |
Age (yrs) |
Handedness |
Pathology |
Localization |
|||
|
Barbagallo et al. [1] |
n = 4 (1.3%) |
male female |
2 2 |
mean range |
48,5 38-60 |
n/a |
2 LGG; 1 cavernous
hemangioma; 1 HGG |
right hem n
= 2 left hem n
= 2 |
Insula (n
=1); fronto-opercular (n = 2); temporal lobe (n =
1) |
|
D’Andrea et al. [5] |
n = 27 (9%) |
male female |
15 12 |
mean range |
65,8 41-79 |
assessed but not
reported |
HGG (IV) |
left hemisphere |
parietal lobe
(posterior temporo-parietal junction; dominant and subcortical areas of AG,
SMG, IPS) |
|
Della Puppa et al. [7] |
n = 3 (1%) |
male female |
1 2 |
mean range |
60 50-68 |
right-handed |
HGG (IV) |
right hemisphere |
parietal lobe |
|
Della Puppa et al. [8] |
n = 2 (0.9%) |
male female |
1 1 |
mean range |
62 57-67 |
right-handed |
HGG (IV) |
right hemisphere |
parietal lobe |
|
Duffau et al. [25] |
n = 1 (0.3%) |
male female |
/ 1 |
mean range |
44 |
right-handed |
LGG |
left hemisphere |
parieto-occipital junction |
|
Ius et al. [26] |
n = 58 (19.5%) |
male female |
30 28 |
mean range |
38 19-61 |
left-handed n
= 15 right-handed n
= 43 |
LGG (II) |
right hem n
= 25 left hem n
= 33 |
primarily situated
in the frontal, temporal, and insular lobes |
|
Kurimoto et al. [18] |
n = 1 (0.3%) |
male female |
1 / |
mean range |
67 |
right-handed |
HGG (IV) |
left hemisphere |
parietal lobe,
near the AG |
|
Lima Maldonado et al. [6] |
n = 14 (14.3%) |
male female |
6 8 |
mean range |
44,1 27-62 |
right-handed |
13 LGG (II) ;1 HGG
(III) |
left hemisphere |
inferior parietal
lobe |
|
Matsuda et al. [2] |
n = 1 (0.3%) |
male female |
/ 1 |
mean range |
56 |
right-handed |
HGG (III) |
left hemisphere |
parietal lobe |
|
Pallud et Dezamis
[3] |
n = 107 (36%) |
male female |
59 48 |
mean range |
40,8 16-74 |
assessed but not
reported |
49 LGG (II); 58
HGG (44 grade III and 14 grade IV) |
right hem n
= 26 left hem n
= 81 |
frontal lobe (n = 64); temporal lobe
(n = 21); parietal lobe (n = 14); insula (n = 8) |
|
Pu et al. [9] |
n = 5 (1.7%) |
male female |
3 2 |
mean range |
25 17-33 |
right-handed |
LGG |
right hem n
= 1 left hem n
= 4 |
parietal lobe (n
= 2); temporal lobe (n = 1); parieto-temporal lobe (n = 1) c; frontal lobe (n = 1) |
|
Roux et al. [10] |
n = 6 (2%) |
male female |
2 4 |
mean range |
52 / |
right-handed |
2 HGG (III); 2 metastases; 1 cavernoma and 1 cortical dysplasia |
right hem n
= 1 left hem n
= 5 |
close to the AG or
involving the posterior parietal area |
|
Roux et al. [11] |
n = 20a (6.7%) |
male female |
10 10 |
mean range |
42,7 21-75 |
left-handed n
= 1 right-handed n
= 15 |
various brain
lesions: low-grade or high-grade astrocytoma, metastasis, cavernoma |
n/a |
parietal lobe (n
= 10) (including AG n = 9); F2 (n = 5) 1 patient with “no
area found”; n = 4a: n/a |
|
Russell et al. [19] |
n = 28 (9.4%) |
male female |
18 10 |
mean range |
44 18-75 |
left-handed n
= 2 right-handed n
= 26 |
24 HGG (13
glioblastomas and 11 anaplastic tumors); 4 LGG |
n/a |
parietal lobe |
|
Scarone et al. [23] |
n = 15 (5%) |
male female |
6 9 |
mean range |
37,6 28-54 |
left-handed n
= 2 right-handed n
= 13 |
LGG |
right hem n
= 2 left hem n
= 13 |
parietal lobe (SPL
n = 4; SMG n = 4); frontal lobe (SMA n = 4; F2-F3 n
= 2); insula (n =1) |
|
Wu et alb
[24] |
n = 4 (1.3%) |
male female |
3 1 |
mean range |
38,7 34-41 |
right-handed |
LGG (2
astrocytomas and 2 oligodendrogliomas) |
left hemisphere |
frontal lobe
(around Broca’s area) |
|
Yu et al. [12] |
n = 1 (0.3%) |
male female |
1 / |
mean range |
41 |
right-handed |
LGG |
left hemisphere |
temporal lobe |
LGG: Low-Grade Glioma (I, II); HGG: High-Grade Glioma (III, IV); hem:
Hemisphere; AG: Angular Gyrus; SMG: Supramarginal Gyrus; IPS: Intraparietal
Sulcus; AF: arcuate fasciculus; GBM: glioblastoma multiforme; F2: middle
frontal gyrus; SPL: superior parietal lobe; SMA: supplementary motor area;
F2-F3: middle and Inferior Frontal Gyri
aIn this study, cortical stimulation
was performed on only sixteen cases because four patients had preoperative
symptoms of acalculia and were excluded from the brain mapping procedure.
Therefore, some results in (Table 1) are based on sixteen cases instead of
twenty.
bIn
this study, there are 4 patients as well as 14 control subjects. Only the data
concerning the 4 patients will be reported in the table.
cIn
this case, for the review, the patient with the temporo-parietal lesion was
grouped with the patients who had parietal lesions.
*The value represents the number of cases unless otherwise indicated.
Table. 3.
Characteristics of the interventions.
|
|
|
|
Assessment |
||||||||
|
|
|
|
Preoperative |
Perioperative |
Postoperative |
||||||
|
Author(s) |
Localization(s) |
No. of Patients |
Assessors |
Assessment(s) (batteries, tests…) |
Mention of acalculia (primary /secondary) |
Assessors |
Assessment(s) (batteries, tests…) |
Mention of acalculia (primary/ secondary) |
Assessors |
Assessment(s) (batteries, tests…) |
Mention of acalculia (primary/ secondary) |
|
Barbagallo et al. (2018) [1] |
Insula;
fronto-opercular; temporal lobe |
n = 4 |
n/a |
Linguistic
assessment: naming and word generation tasks Other cognitive function assessment: / Calculation skills
assessment: n/m |
n = 0 |
a surgeon |
Linguistic
assessment: naming task,
counting task Other cognitive function assessment: visuo-spatial recognition |
n = 1 |
1 NP |
Linguistic
assessment: performed but
not detailed Other cognitive function assessment: / Calculation skills
assessment: n/m |
n = 0 |
|
D’Andrea et al. (2016) [5] |
parietal lobe and
posterior temporo-parietal junction |
n = 27 |
2 NS; 1 ST
|
EHI Linguistic
assessment: BDAE (italian
version) including number naming task Other cognitive function assessment: / Calculation skills
assessment: / |
n = 3 |
none |
n/a
|
n = 0 |
2 NS
|
Linguistic
assessment: BDAE (italian
version) including number naming task Other cognitive function assessment: / Calculation skills
assessment: / |
n = 0 |
|
Della Puppa et al. (2013) [7] |
parietal lobe |
n = 3 |
n/a |
EHI; KPS Linguistic
assessment: AAT (fluency
task); DO80 + neurologic assessment including spontaneous speech, word
generation, repetition, picture naming, reading, or writing Other cognitive function assessment: Visual-spatial functions, executive functions, memory, praxis Quality of life and mood state: EORTC QLQ-C30; HADS Calculation skills
assessment: Number Processing
and calculation battery (35 tasks) |
n = 0 |
1 ST |
Linguistic
assessment: DO80 and
counting Calculation skills
assessment: Single-digit
addition and multiplication, each with a single operand |
n = 3 |
n/a |
Linguistic
assessment: AAT (fluency
task); DO80 + neurologic assessment including spontaneous speech, word
generation, repetition, picture naming, reading, or writing Other cognitive function assessment: Visual-spatial functions, executive functions, memory, praxis Quality of life and mood state: QLQ-C30; Hospital
Anxiety and Depression Scale tests Calculation skills
assessment: Number Processing
and calculation battery (35 tasks) |
n = 0 |
|
Della Puppa et al. (2015) [8] |
parietal lobe |
n = 2 |
1 ST for
calculation tasks (at least) |
EHI Quality of life and mood state: EORTC QLQ-C30; HADS Linguistic
assessment: Subtest of AAT; Token test; Repetition; Written language Reading; DO80
Other cognitive function assessment: ENB Calculation skills
assessment: counting abilities (simple fact retrieval,
rule-based processing, and mental and written calculation in all 4
operations), number comprehension, numerical transcoding, calculation,
arithmetic reasoning, and conceptual knowledge |
n = 0 |
1 ST |
Calculation skills
assessment: Single-digit
addition and multiplication, each with a single operand |
n = 2 |
n/a |
Quality of life and mood state: EORTC QLQ-C30; HADS Linguistic
assessment: Subtest of AAT; Token test; Repetition; Written language Reading; DO80 Other cognitive function: ENB Calculation skills
assessment: counting abilities (simple fact retrieval, rule-based processing, and
mental and written calculation in all 4 operations), number comprehension,
numerical transcoding, calculation, arithmetic reasoning, and conceptual
knowledge |
n = 0 |
|
Duffau et al. (2002) [25] |
parieto-occipital junction |
n = 1 |
n/a |
Harris test of lateral dominance Linguistic
assessment: spontaneous speech, word generation,
repetition, picture naming, writing, and reading Other cognitive function assessment: Visuospatial tests Calculation skills
assessment: Reading Arabic
numbers, writing them from dictation, comparing single and multi-word
numbers, reciting times tables, writing complex calculations, mentally
subtracting seven in sequence, and recalling number sequences forwards and
backwards |
n = 0 |
n/a |
Linguistic assessment: verbal counting, picture naming Calculation skill’s assessment: Simple multiplication problems; Subtraction of seven from one or two
digit numbers (only presented verbally and not also visually) |
n = 1
|
n/a |
Linguistic
assessment: spontaneous speech, word generation,
repetition, picture naming, writing, and reading Other cognitive function assessment: Visuospatial tests Calculation skills
assessment: Reading aloud Arabic numerals, writing them from dictation, comparing
single and multi-word numbers, mentally solving simple multiplication and
subtraction problems |
n = 1
|
|
Ius et al. (2011) [26] |
primarily situated
in the frontal, temporal, and insular lobes |
n = 58 |
1 NS |
EHI Linguistic
assessment: n/a Other cognitive function assessment: n/a Calculation skill
assessment: n/a
|
n/a |
1 NS |
Linguistic
assessment: counting,
picture naming, and reading tests, a repetition or semantic task* Other cognitive function assessment: a bisection line task* Calculation skill
assessment: calculation
task* *depending the
tumor location |
n/a |
1 NS |
Linguistic
assessment: n/a Other cognitive function assessment: n/a Calculation skill
assessment: n/a
|
n/a |
|
Kurimoto et al. (2006) [18] |
parietal lobe,
near the AG |
n = 1 |
n/a |
KPS Linguistic
assessment: object naming,
repetition, spontaneous conversation Calculation skill
assessment: simple addition
and subtraction |
n = 1 > suspected Gerstmann syndrome |
n/a |
Linguistic
assessment: object naming,
repetition, spontaneous conversation Calculation skill
assessment: simple addition
and subtraction (no complex tasks) |
n = 1 |
n/a |
KPS Calculation skill
assessment: simple and complex
addition and subtraction; multiplication and division |
n = 1 |
|
Lima Maldonado et al. (2011) [6] |
inferior parietal
lobe |
n = 14 |
1 NS; 1 ST |
EHI Linguistic
assessment: DO80; BDAE
(including counting) Other cognitive function assessment: / Calculation skill
assessment: / |
n = 0 |
1 NS; 1 ST |
Linguistic
assessment: picture naming
with DO80; counting |
n = 0 |
1 NS; 1 ST |
Linguistic
assessment: DO80; BDAE
(including counting) Other cognitive function: / Calculation skill
assessment: /
|
n = 2 |
|
Matsuda et al. (2019) [2] |
parietal lobe |
n = 1 |
1 senior NS; 1 ST |
Linguistic
assessment: performed but
not detailed Neurologic
assessment: performed but
not detailed |
n = 0 |
1 senior NS; 1 ST |
Linguistic
assessment: picture naming
and counting tasks Calculation skill
assessment: 1-digit addition
and 1-digit multiplication alternately |
n = 1 |
1 senior NS; 1 ST |
Linguistic
assessment: performed but
not detailed Neurologic
assessment: performed
but not detailed |
n = 0 |
|
Pallud &
Dezamis (2017) [3] |
frontal; temporal lobe; parietal lobe; the insular lobe |
n = 107 |
1 senior NS; 1 ST |
EHI; KPS Linguistic
assessment: BDAE; timed
picture naming task with the DO80; fluency semantic and phonological tasks;
timed semantic association task; timed reading task Other cognitive function assessment: praxis* (Rey Figure); memory*; attention*; executive function* Calculation skill
assessment: calculus* (addition, subtraction, and multiplication tasks) *depending the tumor location |
n = 9
|
1 senior NS; 1 ST |
Linguistic
assessment: picture naming
(DO80) and counting tasks
|
n/a |
1 senior NS; 1 ST |
KPS Linguistic
assessment: BDAE; timed
picture naming task with the DO80; fluency semantic and phonological tasks;
timed semantic association task; timed reading task
Other cognitive function assessment: praxis* (Rey Figure); memory*; attention*; executive function* Calculation skill
assessment: calculus* (addition, subtraction, and multiplication tasks) *depending the tumor location |
n/a |
|
Pu et al. (2011) [9] |
parietal lobe; temporal lobe; frontal lobe |
n = 5 |
NP team; 1 ST |
EHI; KPS Linguistic
assessment: WAB (Chinese version): picture naming Other cognitive function assessment: MMSE Calculation skill
assessment: numerical
processing examinations (number comparison, single-digit multiplication and
simple subtraction) |
n = 0 |
n/a |
Linguistic
assessment: language
function mapping Calculation skill
assessment: subtraction and
multiplication tasks
|
n = 0 |
NP team; 1 ST |
KPS Linguistic
assessment: WAB (Chinese version): picture naming Other cognitive function: MMSE Calculation skill
assessment: numerical
processing examinations (number comparison, single-digit multiplication and
simple subtraction) |
n = 1 |
|
Roux et al. (2003) [10] |
close to the AG or
involving the posterior parietal area |
n = 6 |
1 physician |
EHI Linguistic assessment: Written and oral comprehension, naming, language fluency, reading,
computation, dictation, repetition, written transcription, object handling,
and writing abilities Other cognitive functions assessed: A finger-recognition task; a color-naming task Calculation skills assessment: Oral and written calculation abilities, comprehension of
arithmetical signs and numbers, estimation of quantities, reading numbers,
and counting abilities |
n = 4 |
n/a |
Linguistic assessment: A naming task; a reading task; a writing task Other functions assessed: A finger-recognition task; a color-naming task Calculation skills assessment: basic addition tasks
|
n = 6 |
1 physician |
Linguistic assessment: Written and oral comprehension, naming, language fluency, reading,
computation, dictation, repetition, written transcription, object handling,
and writing abilities Other functions assessed: A finger-recognition task; a color-naming task Calculation skills assessment: Oral and written calculation abilities, comprehension of
arithmetical signs and numbers, estimation of quantities, reading numbers,
and counting abilities |
n = 1 |
|
Roux et al. (2009) [11] |
parietal lobe
(including AG); F2 |
n = 20a |
NP team |
EHI Linguistic
assessment: naming and
reading tasks (but not detailed) Other cognitive function assessment: / Calculation skill
assessment: number
production, numerosity judgement, numerical facts, various calculation tests
(written and mental), number comparisons, number knowledge, and suite
production |
n = 4 |
NP team |
Linguistic
assessment: performed but
not detailed Calculation skill
assessment: naming,
alphabetic script reading, and Arabic number reading; calculating tasks |
n = 14 |
NP team |
Linguistic
assessment: naming and
reading tasks (but not detailed) Calculation skill
assessment: number
production, numerosity judgement, numerical facts, various calculation tests
(written and mental), number comparisons, number knowledge, and suite
production |
n = 8 (4 preoperatively and 4 others
postoperatively) whom n
= 2 (at 12 months) |
|
Russell et al. (2005) [19] |
parietal lobe |
n = 28 |
n/a |
Wada test Linguistic
assessment: performed but
not detailed Other cognitive function assessment: performed but not detailed Calculation skill
assessment: n/a (but
signs of Gerstman syndrome investigated) |
n = 0 |
2 surgeons |
/ |
n = 0 |
n/a |
Linguistic
assessment: performed but
not detailed Other cognitive function: performed but not detailed Calculation skill
assessment: n/a (but
signs of Gerstman syndrome investigated) |
n = 1 |
|
Scarone et al. (2009) [23] |
parietal lobe
(SPL; SMG); frontal lobe (SMA; F2); insula |
n = 15 |
1 neurologist; 1
ST; 3 NS |
EHI Linguistic
assessment: BDAE (French
version); writing abilities (“Mechanics of writing examination” and “Recall
of written symbols”) Other cognitive function assessment: / Calculation skill
assessment: / |
n = 0 |
1 ST (at least) |
Linguistic
assessment: picture naming
with DO80; counting (1 to 10) |
n = 0 |
1 ST (at least) |
Linguistic
assessment: BDAE (French
version); auditory comprehension, counting, object naming, spontaneous
speech; writing Other cognitive function: / Calculation skill
assessment: / |
n = 4 (immediate postoperative
among whom n = 1 (at 9
months) |
|
Wu et al. (2008)b [24] |
Frontal lobe
(around Broca’s area) |
n = 4 |
1 ST |
EHI; KPS Linguistic
assessment: WAB (chinese
version): auditory word judgment, auditory word repetition, picture naming Other cognitive function assessment: MMSE Calculation skill
assessment: / |
n = 0 |
1 ST |
Linguistic
assessment: picture naming,
counting, repetition |
n = 0 |
1 ST |
KPS Linguistic
assessment: WAB (chinese
version) Other cognitive function: MMSE Calculation skill
assessment: / |
n = 0 |
|
Yu et al. (2011) [12] |
temporal lobe |
n = 1 |
n/a |
EHI Linguistic
assessment: naming tasks Other cognitive function assessment: a simple reaction time test, a verbal working memory (digit span)
test Calculation skill
assessment: single digit subtraction, Stroop-like number comparison; calculation
tasks |
n = 0 |
n/a |
Linguistic
assessment: picture naming
task Other cognitive function: a simple reaction time test, a verbal working memory (digit span)
test Calculation skill
assessment: simple subtraction and simple and single-digit multiplication tasks |
n = 1 |
n/a |
Linguistic
assessment: naming tasks Other cognitive function: a simple reaction time test, a verbal working memory (digit span)
test Calculation skill
assessment: single digit subtraction, Stroop-like number comparison; calculation
tasks |
n = 0 |
LGG: Low-Grade Glioma (I, II); HGG: High-Grade Glioma (III, IV); hem:
Hemisphere; AG: Angular Gyrus; SMG: Supramarginal Gyrus; IPS: Intraparietal
Sulcus; AF: Arcuate Fasciculus; GBM: Glioblastoma Multiforme; F2: Middle
Frontal Gyrus; SPL: Superior Parietal Lobe; SMA: Supplementary Motor Area;
F2-F3: Middle And Inferior Frontal Gyri; NS: Neurosurgeon; ST:
speech-therapist; NP: Neuropsychologist
AAT: Aachener Aphasia Test; BDAE: Boston Diagnostic Aphasia Examination; DO-80: Test de Dénomination Orale D’images; EHI: Edinburgh Handedness Inventory; ENB: Esame Neuropsicologico Breve; EORTC QLQ-C30: European Organization for Research and Treatment of Cancer Quality of
Life Questionnaire-C30; HADS:
Hospital Anxiety and Depression Scale; HTLD: Harris test of lateral
dominance; KPS: Karnofsky Performance Scale; MMSE: Mini Mental State Examination; NPCB:
Number Processing and calculation
battery; WAB: West Aphasia
Battery
*None, as a controlled variable.
aIn this study, cortical stimulation
was performed on only sixteen cases because four patients had preoperative
symptoms of acalculia and were excluded from the brain mapping procedure.
Therefore, some results in (Table 1) are based on sixteen cases instead of
twenty.
bIn this study, there are 4 patients as well as 14 control subjects. Only the data concerning the 4 patients will be reported in the table.
3. Results
3.1. Characteristics of the Population
Across all studies combined, a total of 297 patients were included (Table 2). The cohorts range is from 1 [2, 12, 17, 18] to 107 [3] participants depending on studies. Among this population, 47% (n = 139) were female and 53% (n = 158) were male and the mean age is 47.4 years ± 20.7 years.
Among 277 patients (Only 277 patients are included because the article by Roux et al. [11] does not specify the number of patients per lesion), 54.9% had a low-grade diffuse glioma, 43.3% had a high-grade diffuse glioma, and 1.8% had another type of lesion (cavernous hemangioma, cavernoma, cortical dysplasia, etc.). Among the 249 patients, lesions were predominantly located in the left hemisphere 75.1% (n = 187), which corresponds to the majority of right-handed patients (87%) when handedness is reported (available for only 82.8% of the 187 patients). Across all studies, so 297 patients, the most commonly affected lobe was the parietal lobe with 118 cases, followed by the frontal lobe with 81 cases and the temporal lobe with 25 cases. Additionally, 10 cases involved lesions in the insula. Furthermore, 63 patients (Ius et al., 2011; Roux et al., 2009) were not classified due to the precise location of the lesions not being specified for each case. Finally, it is important to underline that in the study by Roux et al. [11], 20 patients were initially included, but four were excluded due to preoperative acalculia.
3.2. Characteristics of the Interventions
Of the 297 patients, 242 underwent awake conditions for tumor resection (Table 3). Indeed, in two studies [5, 19], this approach was not proposed: in the first case, general anesthesia with sensory and motor monitoring during the operation was used, while in the second, the study aimed to evaluate the benefits of an alternative method to awake surgery.
3.3. Assessments of Numerical Abilities
Among the seventeen studies, there is a lack of consistency across departments in the methods of assessment. Some rely on standardized test batteries, while others use isolated tasks or automatic routines such as numerical counting rhymes.
3.3.1. Standardized Batteries of Tests
Regarding the assessment of numerical abilities, seven studies mention the use of standardized test batteries like the Boston Diagnosis Aphasia Examination (BDAE) [20], the Western Aphasia Battery (WAB) [21] or the NPC [22].
The BDAE, which is not specifically designed to assess impairments in digital skills, is used by four research teams using a subtest consisting of oral and written automatic sequences and a 5-number dictation [3, 5, 6, 23]. However, only D’Andrea et al. [5] explicitly mention using the “number naming task” whereas the other teams only state that they used the BDAE without specifying which tasks were employed. Similarly, the WAB, which is also not specifically designed to assess impairments in numerical skills, is used by two teams [9, 24]. This battery includes a subtest that assesses the ability to solve mathematical problems, ranging from simple to more complex ones. On the other hand, Della Puppa et al. [7] employed the NPC [22], which provides a more targeted evaluation in this area.
Among all the included studies, Della Puppa et al. [7] is the only one to have administered a comprehensive battery dedicated to assessing numerical skills. The other studies do include tasks aimed at assessing numerical skills, although these are not necessarily linked to a standardized test battery. Depending on the study, the tasks vary both in number and content, including activities such as calculation, numerical transcoding, number comparisons, or recitation of the numerical sequence.
3.3.2. Assessments of Digital Skill Subdomains
Some subdomains are more thoroughly investigated than others. In fact, numerical transcoding, mental arithmetic, written calculation abilities, comparisons, counting task (recitation of the numerical sequence) are among the most commonly studied.
In addition to the study by D’Andrea et al. [5] which used BDAE subtest, three other studies assessed a numerical transcoding task [8, 10, 25] among other numerical tasks for some of them (Table 3). Four out of the seventeen studies focused on mental arithmetic tasks (addition, subtraction or multiplication) [3, 9, 12, 18]. The investigation of written calculation abilities is found in four studies [8, 10, 11, 17], but only during pre- and postoperative phases. Four studies also investigated numerical comparisons. Then, several studies included a counting task, also referred to as “verbal numerical sequence recitation” [1, 3, 6, 23, 24].
3.3.3. Operative Phases and Numerical Assessment
It is interesting to highlight that assessments differ across the three characteristic phases of awake surgery. Generally, the pre-operative and post-operative phases are more exhaustive than the perioperative phase, which is primarily explained by the more limited time available in the operating room [5, 8, 12, 17, 18]. Moreover, all articles mentioned pre-, peri-, and postoperative phases except the study by D’Andrea et al. [5], in which an alternative method is assessed and there is not a three phases protocol. In the study by Russell et al. [19], three phases are mentioned, but the perioperative phase is not conducted in an awake state.
In the preoperative phase, three main areas of assessment are identified: language, cognitive functions, and numerical skills, as well as the patient’s handedness. Among the proposed assessments, a handedness questionnaire is typically included, notably the Edinburgh Handedness Inventory [3, 5-12, 23, 24, 26, 27]. Duffau et al. [25] proposed the Harris test of lateral dominance [28]. Moreover, a language assessment is conducted with an oral picture naming test such as the DO80 naming test [29], which is the most referenced [3, 6-8, 23], or (BDAE) for certain department [3, 5, 6, 23]. Other cognitive skills, such as numerical skills [3, 7-12, 18, 25], visuo-spatial skills [3, 7, 25], or memory skills [3, 7, 12] may also be investigated depending on the studies, their objectives, as well as the areas and needs of the patients.
Then, the perioperative phase is typically defined, in most articles, by a language assessment which is conducted using the same oral picture naming test proposed preoperatively, except for the study by Scarone et al. [23], in which they mentioned the use of the DO80 [29] without confirming its use in the preoperative phase. Perioperatively, among the 17 studies, 10 conducted a somewhat exhaustive numerical assessment [12, 18, 19, 25], and 4 more if we include the 1-to-10 counting task [3, 6, 23, 24]. For some others team, a mental arithmetic task is proposed [2, 7-12, 18, 25, 26]. Ius et al. [26] clarified, however, that this evaluation was not systematic for the included patients and depended on the location of the glioma. Lastly, for most articles, the postoperative assessment is similar, if not identical, to the preoperative assessment, except regarding the patient’s handedness.
3.4. Acalculia Investigation
Among the 297 included patients, 90.6% (n = 269) underwent at least one numerical skills assessment during the pre-, peri-, or postoperative phases [1-3, 5-12, 18, 23-26]. However, in the study by Ius et al. [26] this assessment depended on tumor location, and we therefore have no precise idea of the number of patients assessed for their numerical abilities. Assessment at all three phases: 65% (n = 175) of the 269 patients were assessed at all three phases (pre-, peri-, and postoperative) [3, 6-12, 18, 23-25]. Perioperative assessment only: 23.4% (n = 63) were assessed only during the pre- and postoperative phases [1, 2, 26]. Assessment pre- and postoperatively: 10% (n = 27) were assessed solely during the perioperative phase [5]. In some studies, assessments were limited to a 1-to-10 counting task. For 13.7% of the patients (n = 37) [1, 6, 23, 24], this was therefore more of an automatic, language-based task than a comprehensive numerical skills evaluation.
Some studies did not report investigating acalculia (n/a in Table 2), whether in the pre- [26], peri- [3, 26], or postoperative [3, 26] phases. Consequently, the following data are based on patients who were explicitly assessed for acalculia: Preoperative phase: among the 239 patients investigated, 8.8% (n = 21) had numerical disorders [3, 5, 10, 11, 18]. It should be highlighted that Roux et al. [11] excluded 4 patients from their perioperative protocol due to preoperative acalculia. Perioperative phase: acalculia was observed in 23.2% (n = 30) of the 129 (Considered 4 patients out of 20 in Roux et al. [11] study who do not underwent surgery because of their preoperative acalculia) patients who underwent evaluation during surgery [1, 2, 7, 8, 10-12, 25]. This was sometimes due to the protocol, aiming to map eloquent regions associated with numerical skills, thereby inducing a transient acalculia during stimulation. Postoperative phase: among the 132 evaluated patients at this phase, 14.4% (n = 19) had a numerical disorder. Of these, 42.1% (n = 8) [6, 9, 19, 23] were inaugural cases, with no prior acalculia detected during the pre- or perioperative phases. However, for 36.8% (n = 7) of these 19 patients, no specific numerical skills assessment was conducted, except a potential BDAE subtest for 31.5% (n = 6) of the 19 [6, 23]. Lastly, among the 19 patients, one patient [19] appears not to have undergone evaluation across all three phases, or these evaluations were not explicitly reported by the authors.
Then, among the 65% (n = 175) of the 269 patients assessed at all three operative phases, 93.3% (n = 251) did not present postoperative acalculia. However, if we focus on studies that both evaluated patients at all three phases and explicitly investigated potential postoperative acalculia, the study by Pallud et Dezamis [3] must be excluded. The total number of patients to consider is therefore 162, with 88.9% (n = 144) showing no postoperative numerical disorder.
3.5. Quality of Life Impact
Roux et al. [11] mentioned that acalculia can have a huge impact on the daily life of a patient suffering from brain damage, including damage caused by a glioma, especially as number manipulation is necessary for many activities other than pure mathematics. Moreover, they demonstrated that patients may experience complaints that arise later, distantly from the surgery, which should also be taken into account. Those arguments are also evocated by Della Puppa et al. [8].
3.6. Characteristics of the Included Studies
3.6.1. Characteristics of the Studies
The systematic review of the literature includes 17 articles. All of them were published between 2003 and 2019, even though the inclusion criteria spanned from 2000 to 2024. None of our articles were published before 2002; 44% were published between 2002 and 2010, and 56% between 2011 and 2020. Additionally, no studies on the subject have been published since 2019. Only original papers were included in the systematic review.
3.6.2. Methodological Quality and Robustness of Studies
Using the Gagnier and colleague’s grid (2013), most of the articles selected in this review are between 75% and 100% concerning the methodological quality (excellent quality above 90%, good quality between 75 and 89%, average quality between 50 and 74%): 8 were of excellent methodological quality [2, 7, 10, 11, 18, 19, 23, 25] according the 28 items of the CARE grid, 10 of good quality [1, 3, 5, 6, 8, 9, 12, 24] and one of average quality [26]. Regarding the robustness of the 17 studies according the article by Ebbels [16], 10 fall under model C [1, 2, 7, 8-10, 12, 18, 24, 25], 3 lean towards model D [6, 11], 3 lean towards model B [5, 19, 23], and 1 to model B [3].
4. Discussion
The aim of this review of the literature was to provide an overview of current practices concerning acalculia and glioma awake surgery. Notably, no previous review has attempted to determine a prevalence of acalculia in this context. Consequently, this review underlines two points of interest. Firstly, none of the included studies has proposed a comprehensive evaluation of acalculia, both analytical and functional, and above all, this disorder is not systematically investigated as evidenced by the seventeen studies reviewed. In fact, there is significant variability in how numerical skills are assessed in this context, making it difficult to compare findings across studies. Secondly, it has been observed a lack of consideration and understanding of the functional impact that numerical skill disorders can have on patients’ daily life, beyond the analytical aspect and impact.
According to these two points of interest, one of the key challenges in estimating the prevalence of acalculia in awake glioma surgery, stems from the lack of harmonization in international practices and the lack of awareness regarding the wider implications of this disorder. First of all, it is important to highlight the distinction between dyscalculia, which refers to neurodevelopmental disorders, and acalculia, which is still not sufficiently clear, making precise terminology crucial. In fact, “dyscalculia” should not be used in the context of acquired brain injuries such as glioma.
Then, it is crucial to consider patient as an active participant in their own care, especially concerning a glioma resection and the possibility of an awake surgery. While most of patients included have lesions predominantly located in the left hemisphere, it would be an oversimplification to conclude that only left-sided lesions could lead to deficits in numerical skills. Indeed, some studies emphasize the role of the right hemisphere, particularly the posterior part of the parietal lobe, which plays a key role in certain numerical tasks [5, 7, 17, 18]. Arsalidou and Taylor [30] demonstrate that the parietal, frontal, and prefrontal lobes, the insula, as well as certain gyri such as the left fusiform gyrus, the bilateral inferior, medial, and superior frontal gyri, are involved in numerical processing to varying degrees. Awake surgery for gliomas is distinctive in that it takes into account the patient’s complaints. However, numerical skills are not always highlighted when assessing potential postoperative deficits, as their preservation is generally not a priority compared to other cognitive functions such as language, except in cases where the patient’s profession directly relies on these skills [2]. Consequently, both before and after surgery, numerical impairments are often underestimated. This tendency is reinforced by the widespread belief that acalculia firstly affects calculation abilities (e.g., addition, subtraction) rather than transcoding, estimations or problems solving, in other words, various numerical abilities which can also have a significant impact on patients’ daily life and potentially even more so than calculation itself. While some authors are beginning to underscore the importance of numerical skills in daily life and their relevance during awake glioma surgery [7, 9-11, 25], this perspective remains in the minority.
Indeed, these observations indicate that patients rarely report difficulties related to acalculia spontaneously, and this is not limited to glioma resections [31, 32]. Therefore, the tailored assessment approach presented by Duffau in his 2022 paper, which is one of the fundamental principles of awake glioma surgery [17], must necessarily take into account the potential impact of an acalculia on a patient’s everyday life, even in the absence of any explicitly stated complaint before the operation. The review highlights a lack of harmonization regarding the assessment of acalculia, which complicates the ability to determine the potential prevalence of these kinds of disorders. The 1-to-10 counting task, frequently used to assess numerical abilities in the articles analyzed as part of this review, also raises concerns due to its automatic nature. Like reciting multiplication tables, numbers from 1 to 10 are often recited automatically. However, it is worth noting that this aspect is not systematically discussed by the authors who used this task.
One other limitation of some of the included articles, concerning the assessment, is that some used the awake condition during glioma resection to explore numerical cartography [6-8, 12]. This suggests that their aim was not to assess numerical skills in the context of glioma surgery, but rather to use this setting to gain more insight into the numerical areas of the brain. Others, though a minority, underline the importance of considering a numerical disorder and the role of the awake condition in this neurosurgery in maximizing the preservation of the patient’s calculation abilities in order to optimize the postoperative quality of life for patients [2, 9, 11, 25]; nevertheless, the limitation of these studies is that they focus solely on the analytical aspect of a numerical disorder, without considering the functional impact, which can nevertheless be just as limiting as aphasia [11].
Secondly, it remains to be asked whether we ensure that the patient is aware of the omnipresence of numerical tasks in their daily life, like telling the time, cooking, managing one’s budget, etc. [33], beyond just basic calculation tasks. It is interesting to add the perspective of Benn et al. [33] because they highlighted, for the first time in the literature, the alarming lack of diagnosis and appropriate support for patients with acalculia. They also emphasised the emotional impact this can have on these patients, potentially leading to social isolation. Indeed, among the complaints gathered, patients reported difficulties in managing passwords and numerical information more generally, for tasks such as cooking (measurements, cooking times, etc.), handling phone numbers, managing their finances independently, and following temporal instructions (schedules, telling the time, etc.). Acalculia remains an under-investigated disorder, although it can have a significant impact on patients’ quality of life. It would therefore be pertinent to adopt a more systematic approach in its investigation, particularly in the context of glioma. At the very least, it is crucial to inform patients, from the preoperative phase onwards, about the potential consequences of acalculia on their daily lives after the surgery. This would allow patients to make informed decisions regarding surgical options in collaboration with the neurosurgeon and the medical and paramedical team, in line with the personalized approach inherent to this type of surgery.
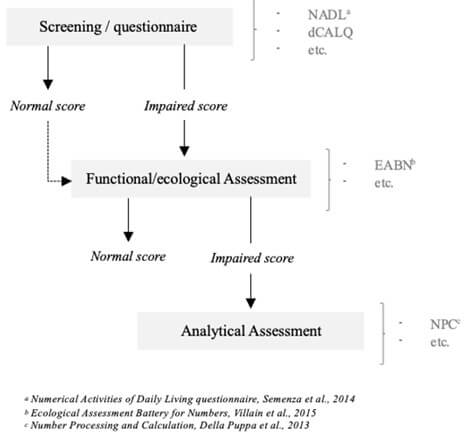
Moreover, considering this functional impact, it may be relevant to recommend using a more functional and patient-centered assessment protocol to evaluate numerical skills appropriately in both clinical and research settings. At the very least, it would be advisable to propose a questionnaire like the Numerical Activities of Daily Living questionnaire [32] or to conduct a screening using tools such as those recently proposed by the dCALQ [34]. In fact, this kind of questionnaire or screening could be particularly useful for better targeting patients' complaints and identifying more persons who may specifically report difficulties with calculation, rather than relying on a broad question about numerical skills during anamnesis which does not always elicit a clear complaint. According to the results, a more in-depth assessment using more “functional” batteries, such as the Ecological Assessment Battery for Numbers [35] could be suggested. Then, in cases where these tests show pathological results, a more analytical investigation could be implemented with transcoding, estimation, mental calculation, or problem-solving, always in a discussion with the patients and their wishes. Indeed, some more analytical test batteries like NPC [22] could, therefore, complete the investigation if necessary. Acalculia should, therefore, be considered in its globality, taking into account the various disorders it may cause. Figure 2 presents a proposed standardized protocol for evaluating numerical skills during awake glioma surgery.
Finally, this review could permit to determine a potential prevalence of acalculia, but the variety of proposed assessments, as well as whether they were conducted at all operative stages or only at one or two, and finally, the more or less systematic nature of the investigation of numerical disorders, make determining this prevalence difficult. Moreover, the authors provide very limited raw data, making it impossible to differentiate between pathological and non-pathological scores or to assess the severity of the disorders. In fact, future prospective studies are recommended to use standardized tools and collect clear and precise data to better understand the true prevalence of acalculia and its clinical implications in this population. From a methodological perspective, most of the included articles are of good to very good quality. However, several studies are limited to single cases, and few articles report on large cohorts. This reinforces the idea that it is difficult to extract a clear guiding principle from the body of literature regarding the assessment of acalculia in the context of awake neurosurgery.
5. Conclusion
To conclude, all in all, the lack of standardized protocols for assessments has led to the use of different tests and batteries across studies to assess numerical skills in the context of glioma awake surgery. This variability in methodologies and the lack of awareness regarding acalculia, complicates the interpretation and comparison of results. Without a unified framework, researchers and clinicians face challenges in drawing consistent conclusions and developing evidence-based guidelines. This variability highlights the urgent need for harmonization to ensure reliable and comparable outcomes in this field.
Data Availability
No data were generated or analyzed during the current study.
Conflicts of Interest
None.
Funding
None.
Author Contributions
Elise Chaulet: Manuscript writing, data collection, data analysis and interpretation. Marie Villain: Study conception and design; key editorial revisions. Others: Double-blind evaluation tasks; revisions. All authors have read and approved the final version of the manuscript.
Guarantor
Marie Villain and Elise Chaulet.
REFERENCES
[1] Giuseppe M V
Barbagallo, Antonio Morrone, Francesco Certo “Intraoperative Computed
Tomography and Awake Craniotomy: A Useful and Safe Combination in Brain
Surgery.” World Neurosurg, vol. 119, pp. 159‑166, 2018. View at: Publisher Site | PubMed
[2] Ryosuke
Matsuda, Kentaro Tamura, Fumihiko Nishimura, et al. “Subcortical Calculation
Mapping During Parietal Glioma Surgery in the Dominant Hemisphere : A Case
Report.” World Neurosurg, vol. 121, pp. 205‑210, 2019. View at: Publisher Site | PubMed
[3] J Pallud, E
Dezamis “Functional and oncological outcomes following awake surgical resection
using intraoperative cortico-subcortical functional mapping for supratentorial
gliomas located in eloquent areas.” Neurochirurgie, vol. 63, no. 3, pp.
208‑218, 2017.
View at: Publisher Site | PubMed
[4] Alfredo
Ardila, Mónica Rosselli “Acalculia and Dyscalculia.” Neuropsychol Rev,
vol. 12, no. 4, pp. 179‑231, 2002.
View at: Publisher Site | PubMed
[5] Giancarlo
D'Andrea, Pietro Familiari, Antonio Di Lauro, et al. “Safe Resection of Gliomas
of the Dominant Angular Gyrus Availing of Preoperative FMRI and Intraoperative
DTI : Preliminary
Series and Surgical Technique.” World
Neurosurg, vol. 87, pp. 627‑639, 2016. View at: Publisher Site | PubMed
[6] Igor Lima
Maldonado, Sylvie Moritz-Gasser, Nicolas Menjot de Champfleur, et al. “Surgery
for gliomas involving the left inferior parietal lobule : New
insights into the functional anatomy provided by stimulation mapping in awake
patients.” J Neurosurg, vol. 115, no. 4, pp. 770‑779, 2011. View at: Publisher
Site | PubMed
[7] Alessandro
Della Puppa, Serena De Pellegrin, Elena d'Avella, et al. “Right parietal cortex
and calculation processing: Intraoperative functional mapping of multiplication
and addition in patients affected by a brain tumor.” J Neurosurg, vol.
119, no. 5, pp. 1107‑1111, 2013.
View at: Publisher Site | PubMed
[8] Alessandro
Della Puppa, Serena De Pellegrin, Anna Lazzarini, et al. “Subcortical mapping
of calculation processing in the right parietal lobe.” J Neurosurg, vol.
122, no. 5, pp. 1038‑1041, 2015.
View at: Publisher Site | PubMed
[9] Song Pu,
Yong-nian Li, Chen-xing Wu, et al. “Cortical Areas Involved in Numerical
Processing: An Intraoperative Electrostimulation Study.” Stereotact Funct
Neurosurg, vol. 89, no. 1, pp. 42‑47, 2011. View at: Publisher
Site | PubMed
[10] Franck-Emmanuel
Roux, Sergio Boetto, Oumar Sacko, et al. “Writing, calculating, and finger
recognition in the region of the angular gyrus : A cortical stimulation study of
Gerstmann syndrome.” J
Neurosurg, vol. 99, no. 4, pp. 716‑727, 2003. View at: Publisher Site | PubMed
[11] Franck-Emmanuel
Roux, Leila Boukhatem, Louisa Draper, et al. “Cortical calculation localization
using electrostimulation.” J Neurosurg, vol. 110, no. 6, pp. 1291‑1299, 2009. View at: Publisher Site | PubMed
[12] Xiaodan Yu,
Chuansheng Chen, Song Pu, et al. “Dissociation of subtraction and
multiplication in the right parietal cortex : Evidence from intraoperative cortical
electrostimulation.” Neuropsychologia,
vol. 49, no. 10, pp. 2889‑2895, 2011.
View at: Publisher Site | PubMed
[13] Matthew J
Page, Joanne E McKenzie, Patrick M Bossuyt, et al. “The PRISMA 2020 statement : An updated
guideline for reporting systematic reviews.” PLOS
Medicine, vol. 18, no. 3, pp. e1003583, 2021. View at: Publisher Site | PubMed
[14] David N
Louis, Arie Perry, Pieter Wesseling, et al. “The 2021 WHO Classification of
Tumors of the Central Nervous System : A summary.” Neuro
Oncol, vol. 23, no. 8, pp. 1231‑1251, 2021. View at: Publisher Site | PubMed
[15] Joel J
Gagnier, Gunver Kienle, Douglas G Altman,, et al. “The CARE guidelines :
Consensus-based clinical case reporting guideline development.” BMJ Case Rep, vol. 2013, pp. bcr2013201554, 2013. View at: Publisher Site | PubMed
[16] Susan H
Ebbels “Intervention research : Appraising study designs, interpreting findings and creating research
in clinical practice.” Int J
Speech Lang Pathol, vol. 19, no. 3, pp. 218-231, 2017. View at: Publisher Site | PubMed
[17] Hugues Duffau
“A Personalized Longitudinal Strategy in Low-Grade Glioma Patients : Predicting
Oncological and Neural Interindividual Variability and Its Changes over Years
to Think One Step Ahead.” J Pers
Med, vol. 12, no. 10, pp. 1621, 2022. View at: Publisher
Site | PubMed
[18] Masanori
Kurimoto, Takashi Asahi, Takashi Shibata, et al. “Safe Removal of Glioblastoma
Near the Angular Gyrus by Awake Surgery Preserving Calculation Ability-Case
Report : Case
Report.” Neurol Med Chir (Tokyo), vol. 46, no. 1,
pp. 46‑50, 2006.
View at: Publisher
Site | PubMed
[19] Stephen M
Russell, Robert Elliott, David Forshaw, et al. “Resection of parietal lobe
gliomas : Incidence
and evolution of neurological deficits in 28 consecutive patients correlated to
the location and morphological characteristics of the tumor.” J Neurosurg, vol. 103, no. 6, pp. 1010‑1017, 2005. View at: Publisher Site | PubMed
[20] Goodglass Harold,
Kaplan Edith “Assessment of Aphasia and Related Disorders.” Lea and Febiger,
1972.
[21] Andrew
Kertesz “Western Aphasia Battery.” Grune & Stratton, 1982.
[22] M Delazer, L
Girelli, A Granà, et al. “Number Processing and Calculation – Normative Data
from Healthy Adults.” Clin Neuropsychol, vol. 17, no. 3, pp. 331‑350, 2003. View at: Publisher Site | PubMed
[23] Pietro
Scarone, Peggy Gatignol, Sophie Guillaume, et al. “Agraphia after awake surgery
for brain tumor : New
insights into the anatomo-functional network of writing.” Surg Neurol, vol. 72, no. 3, pp. 223‑241, 2009. View at: Publisher Site | PubMed
[24] Chen-xing Wu,
Song Pu, Yi Lin, et al. “Fractionated resection on low grade gliomas involving
Broca’s area and insights to brain plasticity.” Chin Med J (Engl), vol.
121, no. 20, pp. 2026‑2030, 2008.
View at: PubMed
[25] H Duffau, D
Denvil, M Lopes, et al. “Intraoperative mapping of the cortical areas involved
in multiplication and subtraction : An electrostimulation study in a patient with a
left parietal glioma.” J Neurol
Neurosurg Psychiatry, vol. 73, no. 6, pp. 733‑738, 2002. View at: Publisher
Site | PubMed
[26] Tamara Ius,
Elsa Angelini, Michel Thiebaut de Schotten, et al. “Evidence for potentials and
limitations of brain plasticity using an atlas of functional resectability of
WHO grade II gliomas : Towards a “minimal
common brain”.” NeuroImage,
vol. 56, no. 3, pp. 992‑1000, 2011.
View at: Publisher Site | PubMed
[27] R C Oldfield
“The assessment and analysis of handedness : The Edinburgh inventory.” Neuropsychologia, vol. 9, no. 1, pp. 97‑113, 1971. View at: Publisher Site | PubMed
[28] Harris AJ
“Harris test of lateral dominance.” Manual of directions for administration and
interpretation. Psychological Corporation, 1947.
[29] Deloche G,
Hannequin D “DO 80 : Test de dénomination orale d’images.” Éditions du Centre de Psychologie Appliquée, 1997.
[30] Marie
Arsalidou, Margot J Taylor “Is 2+2=4? Meta-analyses of brain areas needed for
numbers and calculations.” NeuroImage, vol. 54, no. 3, pp. 2382‑2393, 2011. View at: Publisher Site | PubMed
[31] Hélène
Robert, Marie Villain, Cécile Prevost-Tarabon, et al. “Ecological assessment of
numerical skills in adults with left stroke.” Ann Phys Rehabil Med, vol.
64, no. 1, pp. 101383, 2021. View at: Publisher Site | PubMed
[32] Carlo
Semenza, Francesca Meneghello, Giorgio Arcara, et al. “A new clinical tool for
assessing numerical abilities in neurological diseases : Numerical
activities of daily living.” Front
Aging Neurosci, vol. 6, pp. 112, 2014. View at: Publisher Site | PubMed
[33] Yael Benn,
Mark Jayes, Martin Casassus, et al. “A qualitative study into the experience of
living with acalculia after stroke and other forms of acquired brain injury.” Neuropsychol
Rehabil, vol. 33, no. 9, pp. 1512‑1536, 2023. View at: Publisher Site | PubMed
[34] Joël Macoir,
Marjorie Dionne, Julie Bilodeau, et al. “dCALQ – a new screening test for the
acquired deficits of number processing: Development and normative data.” Appl
Neuropsychol Adult, vol. 11, pp. 1‑10, 2025. View at: Publisher Site | PubMed
[35] M Villain, C Tarabon-Prevost, E Bayen, et al. “Ecological Assessment Battery for Numbers (EABN) for brain-damaged patients: Standardization and validity study.” Ann Phys Rehabil Med, vol. 58, no. 5, pp. 283‑288, 2015. View at: Publisher Site | PubMed
