Received: Mon 17, Nov 2025
Accepted: Fri 02, Jan 2026
Abstract
Background: The role of age in peripheral nerve reconstruction remains controversial, with no clear consensus on an upper age limit for surgical candidacy. While age-related declines in neuroregeneration have been observed, it remains unclear if these justify withholding surgery in older patients. This review critically evaluates the existing literature on peripheral nerve reconstruction outcomes in individuals aged ≥ 65 compared to younger cohorts, including human and animal studies, to assess whether age cutoffs for surgery are evidence-based.
Methods: A systematic review was conducted on January 8, 2025, following PRISMA guidelines. Searches of EMBASE, PubMed, Scopus, Web of Science, Cochrane, and Google Scholar identified studies assessing peripheral nerve reconstruction, nerve transfer, and nerve grafting in relation to age. Inclusion criteria required studies comparing older adults (≥ 65) to younger adults, even as secondary analyses, or animal models with human-equivalent age comparisons. Exclusion criteria included neonatal, central nervous system, or non-mammalian studies. Risk of bias was assessed using ROBINS-I.
Results: Of 398 studies screened, 60 met initial screening criteria, but only three directly compared older to younger adults, none involving injury or reconstruction. Remaining studies compared juveniles, adults aged 18-64, or adults to juveniles. Across studies, aging was associated with reduced axonal regeneration, increased fibrosis, and impaired neuromuscular recovery. However, no study demonstrated an absolute threshold beyond which nerve reconstruction was futile. Functional recovery was observed in older patients, particularly when rehabilitation and adjunct therapies were utilized.
Conclusion: Current evidence does not support an arbitrary age cutoff for nerve reconstruction. While aging impacts nerve regeneration, it does not preclude functional recovery, and no study clearly demonstrated surgical ineffectiveness in older adults. Due to the lack of direct outcome comparisons and prospective studies, surgical decisions should be individualized rather than age-based.
Keywords
Nerve regeneration / physiology, aging / physiology, brachial plexus / surgery, facial nerve / surgery, health care rationing, quality of life
1. Introduction
Peripheral nerve injuries (PNIs) cause significant motor and sensory deficits, chronic pain, and disability that dramatically affect quality of life, often necessitating surgical intervention. Age-related changes in nerve physiology, including reduced axonal plasticity, slower regeneration, and impaired inflammatory resolution, may affect surgical outcomes following nerve repair [1-5]. While advanced age is traditionally considered a negative prognostic factor, there is no consensus on whether a specific age cutoff (e.g., 65 years) should influence surgical decision-making for nerve reconstruction, grafting, or transfer after PNI.
Historical literature has long debated the influence of age on nerve reconstruction outcomes. Early experts like Sunderland emphasized that younger patients exhibit superior regeneration capacity as compared to older adults [6]. Nagano later proposed that exploration of PNIs was not indicated for patients older than 50 years, due to poor outcomes [7]. Rosén and Lundborg reported that age accounted for nearly 50% of sensory recovery variance after nerve repair [8]. However, when evaluating a 35-year history of 3,000 nerve injuries, Spinner and Kline only mentioned that children do better than adults, and Kanaya et al. found no association with age in a series of 52 brachial plexus surgeries [9, 10]. More recent studies have challenged strict age-based exclusion, demonstrating that patients over 60 years could still benefit from nerve transfers and reconstruction, arguing that physiological rather than chronological age should guide decision-making [11, 12].
Epidemiologic data estimate an incidence of PNI of 13.6 people per 1,000,000 for lower extremity PNI and 43.8 people per 1,000,000 for upper extremity PNI, although this data is over a decade old [13, 14]. While most PNIs occur in young patients, demographic shifts and longer life expectancies are increasing the prevalence of nerve injuries in older adults [15]. Advances in healthcare have enabled seniors to remain more active, thus contributing to a rise in nerve injuries from trauma, falls, and iatrogenic causes. Surgical intervention rates mirror these demographic shifts. Currently, patients aged 65 and older are more likely than any other age group to have surgery [16]. This trend challenges longstanding hesitancy to offer nerve reconstruction to older patients, and signals a paradigm shift towards broader surgical inclusion.
Given the anticipated rise in nerve injury cases among older adults, it is both timely and essential to re-evaluate age-related assumptions in surgical candidacy and outcomes. The present systematic review aims to synthesize current evidence comparing surgical outcomes in patients aged 65 and versus younger cohorts, as well as in animal models of equivalent biological ages. Ultimately, we seek to determine whether the current literature supports the notion of an age cutoff for surgical intervention in PNI.
2. Methods
2.1. Data Source, Search Strategy and Studies Selection
This systematic review was conducted on Jan 8th, 2025, by querying the electronic databases EMBASE, PubMed, Scopus, Web of Science, Cochrane, and Google scholar. A search strategy was generated using the following strategy: i) ‘‘age,’’ OR ‘‘aging,’’ OR “older” in the title AND ii) “nerve transfer” OR “nerve graft” OR “nerve regeneration” in the abstract AND iii) ‘‘sciatic” OR “tibial” OR “peroneal” OR “facial” OR “oculomotor” OR “brachial plexus” OR “radial” OR “ulnar” OR “median” OR “peripheral” in the abstract. Additional sources included reference lists of included studies. We followed Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Phase 1 exclusion criteria: i) non-mammalian studies, ii) obstetric/neonatal studies, iii) intervention of only free muscle transfers, iv) central nervous system, v) cochlear, ocular, or olfactory nerve pathway focus, vi) unavailable full texts, and vii) editorials, letters, reviews.
2.2. Data Collecting Process
After collecting the studies, all duplicates were removed. Studies were filtered based on their titles and abstracts, ensuring they met the eligibility criteria. The remaining studies were evaluated by reading the full texts. Variables collected included author, title, journal, year, country, genus, species, number of groups, number of participants, age availability, youngest age, oldest age, second oldest age, primary outcome, secondary outcomes, nerve studied, and type of nerve injury.
2.3. Age Comparisons
All animal ages were converted to human ages based on developmentally appropriate equations [17-20]. The cutoff of 65 years was selected a priori based on its frequent clinical use. Comparative studies specifically analyzing two distinct age groups were required to substantiate any proposed cutoff age. Studies using only regression analysis or evaluating single age cohorts, while valuable in demonstrating successful outcomes in older patients, were insufficient for establishing a precise age threshold. Studies without comparing adults at least older than 65 to adults at least older than 18 were excluded.
2.4. Risk of Bias Assessment
The risk of bias was evaluated for each study using the ROBINS-I tool for non-randomized studies.
3. Results
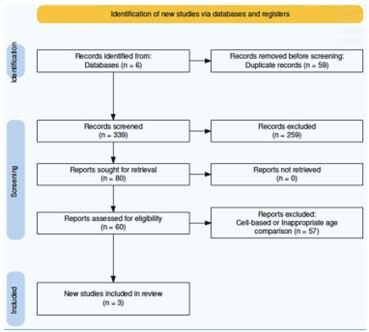
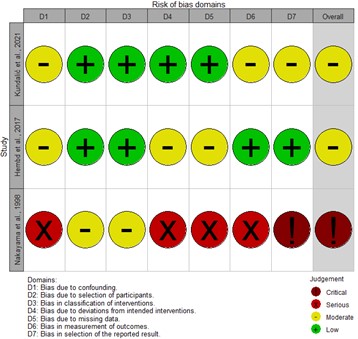
Following the initial screening, 398 studies met the search criteria, and 60 studies were ultimately included (Figure 1) [21].. Each selected study was assessed for its methodology, age stratification, and outcomes related to nerve regeneration, functional recovery, and structural changes. Among the included studies, only three directly compared individuals aged 65 and older to those between 18 and 64 (Table 1) [22-24]. Bias was noted to be moderate in the two human studies and critical in the animal study (Figure 2). The distribution of age comparisons in the included studies was as follows: nine studies focused exclusively on subjects younger than 18 [25-33], 29 studies compared adults aged 18-64 to juveniles [1, 2, 4, 5, 34-57], and one study examined individuals older than 65 against younger cohorts under 18 [58]. Eighteen studies compared adults aged 18-64 to other adults within the same range (Figure 3) [3, 59-75].
Table. 1. Included studies.
|
Author |
Nakayama [21] |
Kundalic [23] |
Hembd [22] |
|
Journal |
Journal of the Autonomic Nervous System |
Archives of Biological Sciences |
Plastic and Reconstructive Surgery |
|
Year |
1998 |
2021 |
2017 |
|
Country |
Japan |
Serbia |
USA |
|
Genus |
Rattus |
Homo |
Homo |
|
Number of Participants |
18 (9 per group) |
15 cadavers |
126 (63 hemifaces) |
|
Youngest (years) |
13 years* |
25 years |
22 years |
|
Oldest (years) |
82 years* |
86 years |
97 years |
|
Second Oldest (years) |
66 years* |
64 years |
61 years |
|
Primary Outcome |
Conduction velocities and morphometric
changes in pelvic nerve fibers |
Morphometric changes in ECM proteins
(collagen type I, IV, laminin) |
Negative correlation between age and axonal
load in the facial nerve |
|
Secondary Outcomes |
Unmyelinated fiber loss, size distributions,
conduction velocity |
Endoneurial ECM remodeling, implications for
regeneration |
Axonal count at buccal and zygomatic
branches, histomorphometric analysis |
|
Nerve Studied |
Pelvic nerve |
Sural nerve |
Facial nerve |
|
Type of Nerve Injury |
No injury (age-related analysis) |
No injury (aging study) |
No injury (anatomical study) |
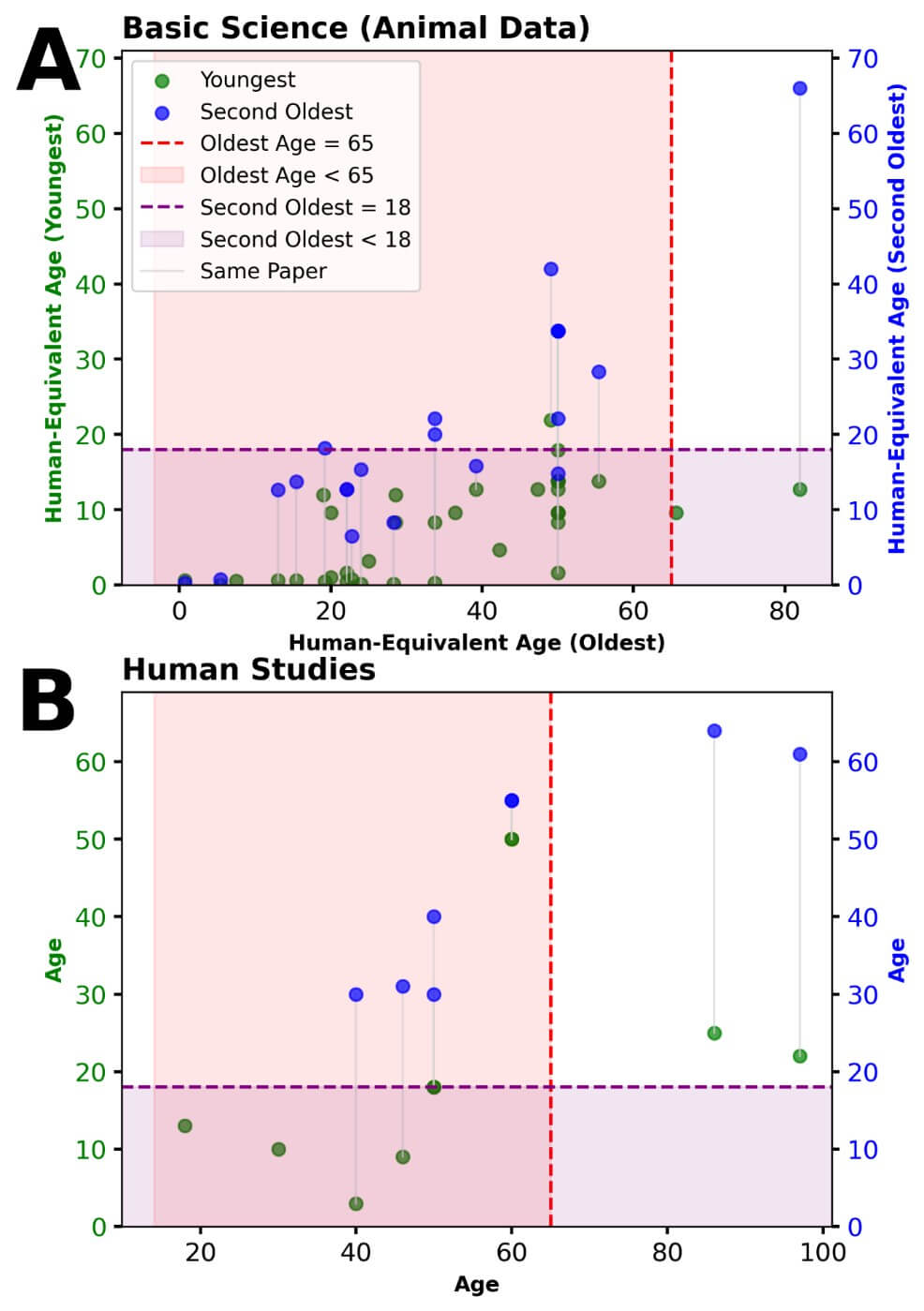
Nakayama et al. studied age-related differences in peripheral nerve physiology using female Wistar rats, comparing a young cohort (3-9 months, equivalent to humans <65 years) and an aged cohort (30-37 months, equivalent to humans ≥65 years). They primarily examined conduction velocities and morphometric changes in pelvic nerve fibers, with secondary outcomes including unmyelinated fiber loss, size distributions, and conduction velocity. In doing so, the authors demonstrated a significant reduction in conduction velocity and fiber size in the aged group, indicating a physiological decline in nerve function with aging. These findings suggest potential implications for nerve grafting and transfer outcomes in older patients. Importantly, this study was an age-related analysis without nerve injury [22].
Similarly, Hembd et al. conducted a cadaveric anatomical study analyzing facial nerve axonal density across three human age groups: young adults (22-59 years, <65 years), middle-aged adults (61-79 years, transitional), and elderly individuals (80-97 years, ≥65 years). They found a significant decline in axonal load with increasing age, particularly after 60 years. This suggests that donor nerves may be less viable in older individuals, which could adversely impact the effectiveness of nerve grafting procedures. The primary outcome was the correlation between age and axonal load, while secondary outcomes included axonal count at the buccal and zygomatic branches and histomorphometric analysis of the nerve structure [23].
Kundalic et al. examined age-related remodeling of the extracellular matrix (ECM) within human sural nerves, stratifying subjects into three groups: young adults (25-44 years, < 65 years), middle-aged adults (45-64 years, < 65 years), and elderly individuals (65-86 years, ≥ 65 years). Their study identified substantial ECM alterations in the older cohort, characterized by increased collagen type IV and decreased collagen type I and laminin content. These compositional changes impede nerve regeneration following injury, suggesting delayed or incomplete recovery after nerve grafting in older patients. The study’s primary outcome focused on morphometric changes in ECM proteins, while secondary outcomes assessed endoneurial ECM remodeling and its implications for nerve regeneration [24].
Despite accumulating evidence of age-related declines in nerve regeneration, no study in this systematic review identified a strict age threshold beyond which nerve grafting or transfers should not be performed. Furthermore, no study directly compared an age group >= 65 to an adult age group < 65 after injury or after repair.
3.1. Alternative Age Cutoffs
If the cutoff had been set at 60 years, two additional studies would have been included; however, these studies demonstrated no statistically significant differences in elbow and shoulder function between age groups based on this cutoff [74, 75]. Similarly, if the cutoff had been set at 70 or 80 years, no additional eligible studies were identified, thus leaving our results unchanged. Collectively, these findings further reinforce that currently available evidence does not strongly support any specific age threshold for peripheral nerve reconstruction outcomes.
4. Discussion
Nerve reconstruction, grafting, and transfer represent essential surgical interventions to restore function in patients suffering from nerve injuries. Although the influence of age on neuroregeneration is well recognized, the critical question remains: should age be a determining factor in the decision to offer nerve reconstruction surgery? Based on the existing literature, there is insufficient support for an absolute age cutoff in offering these procedures. While animal and cadaveric studies, including those by Nakayama, Hembd, and Kundalic, provide valuable insights into the effects of aging on nerve structure and function, they do not offer definitive evidence that patients over 65 years of age (or older cutoff) should be excluded from reconstructive nerve surgery.
4.1. Neuroregeneration and the Aging Nervous System
Aging is accompanied by measurable declines in regenerative potential, as shown in both animal models and human studies. Nakayama et al. (1998) reduced number and conduction velocities of both myelinated and unmyelinated fibers in the pelvic nerve of aged mice, suggesting diminished neural transmission and regeneration. Similarly, Hembd et al. (2017) demonstrated clinically that axonal load in the human facial nerve declines with age, a finding with potential implications for facial reanimation procedures. Kundalic et al. (2021) further illustrated in cadavers structural changes in the extracellular matrix of the aging sural nerve, emphasizing the morphological alterations that occur with advancing age. While these studies confirm age-related deterioration in nerve architecture and function, they do not establish that these changes preclude meaningful functional recovery following nerve reconstruction.
4.2. The Lack of Evidence for a Defined Age Threshold
A major limitation in the existing body of research is the paucity of high-quality, large-scale studies comparing nerve reconstruction outcomes in patients above and below an age cutoff of ≥ 65. While we know that neuroplasticity and regenerative potential decline with age, there is no clear point at which nerve surgery becomes futile. The studies reviewed here do not examine post-operative functional recovery in aged patients undergoing nerve repair. Instead, they focus on anatomical and histological changes, which, while relevant, do not provide sufficient justification for denying patients surgical intervention.
In other fields of medicine, age alone is rarely used as a strict criterion for excluding patients from surgery. In orthopedic surgery, for example, joint arthroplasty is routinely performed in elderly patients despite the presence of age-related physiological changes that may impact bone quality and healing [76]. Similarly, in cardiovascular surgery, operative eligibility is determined by consideration of physiologic reserve, functional status, and comorbidities together, rather than age alone [77].
4.3. The Potential for Functional Recovery in Older Patients
Despite reduction in neuroplasticity, older adults are still capable of meaningful functional recovery following nerve repair. The concept of compensatory neuroplasticity suggests that even when the rate of regeneration is slower, reorganization of adjacent neural circuits allow for partial recovery and functional improvement. Similar principles are observed and well documented in stroke rehabilitation, where elderly patients demonstrate substantial gains in function despite diminished neuroplasticity when compared to younger individuals [78].
Additionally, the role of rehabilitation and adjunct therapies should not be overlooked. The success of nerve reconstruction is not solely dependent on the intrinsic regenerative capacity of the nervous system but also on rehabilitation strategies, physical therapy, and neurostimulation techniques that should be prioritized in postoperative management.
4.4. Cost-Effectiveness and Quality of Life
When evaluating the cost-effectiveness of nerve reconstruction in elderly patients, the balance between quality-adjusted life years (QALY) and overall financial justification is complex. Cost-effectiveness studies on nerve reconstruction in older adults are limited, but insights from related surgical fields suggest that these procedures can still be economically justified. Studies on brachial plexus injuries in all patients report that most patients experience significant functional improvement and high satisfaction post-surgery [79, 80]. Comparable findings have been observed in cardiac and orthopedic surgery, where procedures like bypass surgery and joint replacement remain cost-effective even when the per-QALY cost is higher in the elderly [81, 82]. Additionally, analogous procedures such as carpal tunnel release and lumbar decompression have been deemed cost-effective in older adults [83, 84].
2.3. Statistical Analysis
However, elderly patients inherently have fewer remaining years to benefit from functional recovery, increasing the relative per-QALY cost. Older adults typically lack indirect cost benefits, such as return-to-work productivity, unlike younger patients who can offset surgery costs through restored workforce participation. Nevertheless, quality of life improvements remain critically valuable—regaining functional independence from motor improvement or pain relief can significantly reduce long-term expenses related to caregiving, assisted living, and healthcare utilization associated with disability. Hence, nerve reconstruction can still be economically reasonable due to its potential to enhance autonomy and reduce broader societal costs from dependency.
Importantly, recovery after nerve reconstruction often spans up to 24 months to achieve maximal functional gains. For older patients with significant comorbidities or limited life expectancy, alternative treatment strategies such as tendon transfers or advanced prosthetics (e.g., myoelectric devices), may offer more immediate functional restoration. Although nerve transfers are generally comparable in cost to tendon transfers, which are typically less costly than myoelectric prosthetics, the wide range and rapidly advancing technology of myoelectric prosthetics can complicate direct cost comparisons. Nevertheless, despite the complexity and variability in costs associated with myoelectric devices, their immediate functional benefits and user adaptability may justify their use in select elderly populations.
4.5. Ethical and Clinical Implications of Denying Surgery Based on Age
Restricting access to nerve reconstruction based purely on age raises ethical concerns. It risks contributing to ageism in medicine, where older patients are denied potentially beneficial treatments due to assumptions about their prognosis rather than individualized assessments [85]. Surgical decisions should be made based on functional status, comorbidities, and patient goals rather than an arbitrary age threshold [86, 87]. Given the profound impact nerve reconstructive procedures can have on independence, mobility, and psychosocial well-being, withholding potentially restorative interventions based on age alone is ethically untenable.
4.6. Basic Science Correlates
4.6.1. Adult to Adult Comparisons
Comparative animal studies modeling human-equivalent ages ranging 18-64 offer insights into how nerve regeneration and functional recovery vary across stages of adulthood. Several studies demonstrated that functional recovery after peripheral nerve injuries declined progressively with age, with older adults showing decreased axonal regeneration and increased fibrosis compared to younger adults [3, 59, 60, 68]. Morphometric analyses of peripheral nerves confirmed that aging results in reduced Schwann cell density, increased axonal atrophy, and decreased myelin integrity, all of which contribute to slower conduction velocity and impaired nerve regeneration [61, 64, 67]. Studies on vascularization and extracellular matrix remodeling suggested that nerve regeneration in older adults is hampered by diminished angiogenesis and increased collagen deposition, reducing the ability of regenerating axons to traverse the nerve repair site [59, 63]. Additionally, electrophysiological studies found that age-related declines in nerve conduction velocity were accompanied by altered muscle activation patterns and delayed neuromuscular transmission, further contributing to impaired functional recovery [62, 65]. However, certain studies suggested that while older adults experience more delayed recovery, meaningful functional gains were still achievable with rehabilitation and optimized nerve repair strategies [69]. These findings emphasize that while nerve regeneration declines with age, even within adulthood, functional recovery is still possible in older adults, underscoring that diminished regeneration does not equate to futility.
4.6.2. Adult to Juvenile Comparisons
Studies comparing adult animals with human-equivalent ages of 18-64 to juveniles provide valuable insights into nerve regeneration and functional recovery differences between younger and mature individuals. Several studies demonstrated that neuromuscular junction stability and recovery following nerve transection were significantly impaired in older subjects due to molecular expression differences in acetylcholine receptors and muscle regulatory factors [4, 34, 38]. Collateral and sudomotor nerve reinnervation studies showed robust reinnervation in younger nerves, whereas older nerves exhibited reduced regenerative capacity and incomplete functional restoration [33, 35, 41, 60]. Motor axon regeneration studies highlighted a preferential motor reinnervation effect in younger subjects, with superior axonal transport and Schwann cell migration [37, 46, 47]. Investigations of Schwann cell-mediated immune responses revealed age-related delays in macrophage infiltration and myelin debris clearance, negatively affecting axonal regeneration [39, 49, 55]. Younger subjects also exhibited enhanced Schwann cell survival and neurotrophic support compared to older nerves [48, 50, 51].
Morphometric and electrophysiological studies showed older subjects had slower nerve conduction recovery, increased axonal atrophy, and reduced Schwann cell-axon interactions, prolonging functional deficits [43, 44, 53, 54, 56]. Genetic analyses indicated diminished immune recruitment, lower neurotrophic factor expression, and slower recovery in aged nerves [43, 47, 48]. Functional outcomes following nerve grafting and conduit repair consistently demonstrated superior recovery in younger subjects, notably in muscle reinnervation, CMAP amplitude, and rehabilitation compliance [52, 55, 57]. Regenerative delays were also noted in facial, brachial plexus, and tibial nerve injuries, with older subjects showing slower axonal outgrowth and increased synkinesis post-repair [40, 54, 56]. Collectively, these findings underscore reduced regenerative capacity, impaired neuromuscular recovery, and diminished functional outcomes in adults versus younger individuals. Although age-related declines in regeneration exist from youth to adulthood, meaningful recovery remains achievable through nerve reconstruction interventions.
4.7. Clinical Correlates
4.7.1. Adult-to-Adult Comparisons
Clinical data mirror basic science findings, revealing age-associated attenuation of outcomes, but not their absence. Older adults undergoing brachial plexus nerve transfer surgeries demonstrated significantly lower rates of successful functional outcomes compared to younger patients. Gillis et al. reported shoulder abduction outcomes in patients aged 50-77 years, finding no significant difference between patients aged 50-59 years (n = 27; 33% achieving ≥ M3, 9/27) and those aged 60-77 years (n = 13; 38% achieving ≥ M3, 5/13) [74]. In a separate study focusing on elbow flexion, the same group reported functional recovery (≥ M3) in 61% (23/38) of patients aged 50-59 years compared to 59% (10/17) of those aged 60-72 years, also without a significant difference between these two older adult age groups [75]. In contrast, Bhatia et al. found that younger adults under 30 years (n = 118) achieved functional elbow flexion (MRC grade ≥ 3) at a significantly higher rate (81%) compared to older adults over 30 years (49%; n = 82) [73]. Sharma et al. further supported this age-related trend, showing among 17 patients that those younger than 40 years (n = 10) had superior elbow flexion outcomes (70% achieving M4 strength and 30% achieving M3), compared to patients older than 40 years (n = 7), among whom only 43% achieved M3 and none achieved M4 strength; however, this difference did not reach statistical significance (p = 0.14) [70]. Similarly, Socolovsky et al. showed numerically better elbow flexion outcomes (MRC ≥ 4) in younger adults aged 20-29 years (75%, 15/20) compared to adults aged ≥40 (57.1%, 8/14), though this difference was not statistically significant (Chi-square = 0.52, p = 0.47). Shoulder abduction outcomes also did not significantly differ across these adult age groups [71]. Conversely, Ertem et al. demonstrated a significant age-related decline in median and ulnar nerve recoveries, reporting excellent and good motor recovery in 95% of adults aged 18-25 (n = 20), 57.7% in those aged 26-45 (n = 52), and only 20% in those aged 46-62 (n = 15), highlighting a statistically significant and progressive deterioration with advancing age (p < 0.001) [66]. This stark difference suggests evidence supporting an age cutoff around 25 years for optimal median and ulnar nerve repair outcomes, which may be considerably younger than current clinical practice typically considers.
An additional analysis within the adult subgroup from Fornander et al. suggested numerically better sensory outcomes among younger adults (19-23 years, 2 of 3 patients achieving fair or better outcomes) compared to older adults (> 23 years, 0 of 2 patients). Although limited by small sample size, these findings support the possibility of decreasing sensory recovery potential with advancing adult age, reflecting diminishing cortical plasticity [72]. Collectively, these studies validate basic science findings, confirming that while regenerative potential diminishes progressively with age, meaningful recovery remains achievable well into later adulthood.
4.7.2. Adult-to-Juvenile Comparisons
Comparisons between adult and juvenile patients undergoing nerve reconstruction consistently show that younger individuals demonstrate superior nerve regeneration due to greater neuroplasticity and regenerative capacity. Studies comparing neonatal and older patients undergoing brachial plexus nerve transfers have shown significantly superior cortical reorganization and independent volitional control in infants and young children. Socolovsky et al. observed that 100% of neonatal patients (mean age 10 months, n = 22) attained full volitional elbow flexion without simultaneous wrist flexion, compared to only 47.7% of older patients (mean age 30.2 years, n = 44) who required compensatory motor commands (p < 0.001) [1]. Fornander et al. similarly noted minimally better sensory recovery outcomes in patients who sustained median nerve injuries at or before 7-18 years of age (50% achieving fair or better outcomes) compared to those injured after 21-57 years of age (40% achieving fair or better outcomes). However, this difference was not statistically significant (p = 1.0), likely reflecting the low sample size (n = 11) [72]. Younger patients' tendency toward better sensory outcomes may reflect greater cortical plasticity. Collectively, these findings reinforce that younger individuals benefit substantially from superior Schwann cell support, faster axonal regrowth, and enhanced cortical plasticity, whereas adults frequently exhibit slower and incomplete neural recovery.
5. Conclusion
The current body of literature does not support denying nerve reconstruction surgery to patients due to age. While aging affects neuroregeneration, the available studies do not demonstrate that these changes make surgical intervention ineffective or futile. The lack of functional outcome data in older populations highlights a critical gap in research, emphasizing the need for further studies before any age-related surgical restrictions can be justified. Until then, treatment decisions should be made on a case-by-case basis, ensuring that older adults who could benefit from nerve reconstruction are not unfairly excluded from potentially life-changing interventions.
Funding
None.
Conflicts of Interest
None.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Abbreviations
PNI: Peripheral Nerve Injury
ECM: Extracellular Matrix
QALY: Quality-Adjusted Life Year
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
ROBINS-I: Risk of Bias in Non-randomized Studies - of Interventions
CMAP: Compound Muscle Action Potential
REFERENCES
[1] Mariano Socolovsky, Ana
Lovaglio, Gonzalo Bonilla, et al. “Brain plasticity and age after restoring
elbow flexion with distal nerve transfers in neonatal brachial plexus palsy and
nonneonatal traumatic brachial plexus injury using the plasticity grading scale.”
J Neurosurg, vol. 139, no. 6, pp. 1568-1575, 2023. View at: Publisher Site | PubMed
[2] Peter J Apel, Jianjun Ma,
Michael Callahan, et al. “Effect of locally delivered IGF-1 on nerve
regeneration during aging: an experimental study in rats.” Muscle Nerve,
vol. 41, no. 3, pp. 335-341, 2010. View at: Publisher Site | PubMed
[3] L A Bharali, S J Lisney
“Success of Regeneration of Peripheral-Nerve Axons in Rats After Injury at
Different Postnatal Ages.” J Neurol Sci, vol. 100, no. 1-2, pp. 203-210,
1990. View at: Publisher Site | PubMed
[4] J J Campbell, B Pomeranz
“A new method to study motoneuron regeneration using electromyograms shows that
regeneration slows with age in rat sciatic nerve.” Brain Res, vol. 603,
no. 2, pp. 264-270, 1993. View at: Publisher Site | PubMed
[5] M A Khalifa, E A
Montgomery, N Ismiil, et al. “Immunohistochemical expression of CD34 in
connective tissue sheaths of aging human peripheral nerve.” Surg Radiol Anat
SRA, vol. 45, no. 10, pp. 1331-1398, 2000. View at: Publisher Site | PubMed
[6] S Sunderland “The anatomy
and physiology of nerve injury.” Muscle Nerve, vol. 13, no. 9, pp.
771-784, 1990. View at: Publisher Site | PubMed
[7] A Nagano “Treatment of
brachial plexus injury.” J Orthop Sci, vol. 3, no. 1, pp. 71-80, 1998.
View at: Publisher Site | PubMed
[8] B Rosén, G Lundborg, L B
Dahlin, et al. “Nerve repair: correlation of restitution of functional
sensibility with specific cognitive capacities.” J Hand Surg Edinb Scotl,
vol. 19, no. 4, pp. 452-458, 1994. View at: Publisher Site | PubMed
[9] R J Spinner, D G Kline
“Surgery for peripheral nerve and brachial plexus injuries or other nerve
lesions.” Muscle Nerve, vol. 23, no. 5, pp. 680-695, 2000. View at: Publisher Site | PubMed
[10] F Kanaya, M Gonzalez, C M
Park, et al. “Improvement in motor function after brachial plexus surgery.” J
Hand Surg, vol. 15, no. 1, pp. 30-36, 1990. View at: Publisher Site | PubMed
[11] Andreas Gohritz, Gregor
Laengle, Anna Boesendorfer, et al. “Nerve Transfers for Brachial Plexus
Reconstruction in Patients over 60 Years.” J Pers Med, vol. 13, no. 4,
pp. 659, 2023. View at: Publisher Site | PubMed
[12] Kota Hayashi, Johnny
Chuieng-Yi Lu, Tommy Nai-Jen Chang, et al. “Outcomes for the shoulder and elbow
following surgical reconstruction for traumatic brachial plexus injury in
patients over 60 years.” J Hand Surg Eur Vol, vol. 50, no. 8, pp.
1041-1049, 2025. View at: Publisher Site | PubMed
[13] Michael Karsy, Ryan
Watkins, Michael R Jensen, et al. “Trends and Cost Analysis of Upper Extremity
Nerve Injury Using the National (Nationwide) Inpatient Sample.” World
Neurosurg, vol. 123, pp. e488-e500, 2019. View at: Publisher Site | PubMed
[14] Chase H Foster, Michael
Karsy, Michael R Jensen, et al. “Trends and Cost-Analysis of Lower Extremity
Nerve Injury Using the National Inpatient Sample.” Neurosurgery, vol.
85, no. 2, pp. 250-256, 2019. View at: Publisher Site | PubMed
[15] Mohammed K Al Harbi, Salah
M Alshaghroud, Mohammed M Aljahdali, et al. “Regional anesthesia for geriatric
population.” Saudi J Anaesth, vol. 17, no. 4, pp. 523-532, 2023. View
at: Publisher Site | PubMed
[16] Mark C Bicket, Kao-Ping
Chua, Pooja Lagisetty, et al. “Prevalence of Surgery Among Individuals in the
United States.” Ann Surg Open, vol. 5, no. 2, pp. e421, 2024. View at: Publisher Site | PubMed
[17] Chełmińska, D. Kowalska “The Effectiveness of Maize
DDGS in Rabbit Diets / Efektywność zastosowania suszonego
wywaru kukurydzianego w dietach dla królików.” Ann Anim Sci, vol. 13, no. 3, pp. 571-585. View at: Publisher Site
[18] Sulagna Dutta, Pallav
Sengupta “Men and mice: Relating their ages.” Life Sci, vol. 152, pp.
244-248, 2016. View at: Publisher Site | PubMed
[19] Pallav Sengupta “The
Laboratory Rat: Relating Its Age With Human’s.” Int J Prev Med, vol. 4,
no. 6, pp. 624-630, 2013. View at: PubMed
[20] Pallav Sengupta, Sulagna
Dutta “Mapping the Age of Laboratory Rabbit Strains to Human.” Int J Prev
Med, vol. 11, pp. 194, 2020. View at: Publisher Site | PubMed
[21] H Nakayama, K Noda, H
Hotta, et al. “Effects of aging on numbers, sizes and conduction velocities of
myelinated and unmyelinated fibers of the pelvic nerve in rats.” J Auton
Nerv Syst, vol. 69, no. 2-3, pp. 148-155, 1998. View at: Publisher Site | PubMed
[22] Austin Hembd, Purushottam
Nagarkar, Justin Perez, et al. “Correlation between Facial Nerve Axonal Load
and Age and Its Relevance to Facial Reanimation.” Plast Reconstr Surg,
vol. 139, no. 6, pp. 1459-1464, 2017. View at: Publisher Site | PubMed
[23] Braca K. Kundalić, Slađana Z, Ugrenović, Ivan D. Jovanović, et al. “Morphometric analysis of the human endoneurial extracellular
matrix components during aging.” Arch Biol Sci, vol. 73, no. 1, pp. 103-110, 2021.
View at: Publisher Site
[24] W P Ng, A M Lozano
“Neuronal age influences the response to neurite outgrowth inhibitory activity
in the central and peripheral nervous systems.” Brain Res, vol. 836, no.
1-2, pp. 49-61, 1999. View at: Publisher Site | PubMed
[25] K Kuzis, J D Coffin, F P
Eckenstein “Time course and age dependence of motor neuron death following
facial nerve crush injury: role of fibroblast growth factor.” Exp Neurol,
vol. 157, no. 1, pp. 77-87, 1999. View at: Publisher Site | PubMed
[26] A Fex Svenningsen, M Kanje
“Estrogen and progesterone stimulate Schwann cell proliferation in a sex- and
age-dependent manner.” J Neurosci Res, vol. 57, no. 1, pp. 124-130,
1999. View at: Publisher Site | PubMed
[27] S I Zacks, M F Sheff
“Age-related impeded regeneration of mouse minced anterior tibial muscle.” Muscle
Nerve, vol. 5, no. 2, pp. 152-161, 1982. View at: Publisher Site | PubMed
[28] F Piehl, H Hammarberg, G
Tabar, et al. “Changes in the mRNA expression pattern, with special reference
to calcitonin gene-related peptide, after axonal injuries in rat motoneurons
depends on age and type of injury.” Exp Brain Res, vol. 119, no. 2, pp.
191-204, 1998. View at: Publisher Site | PubMed
[29] B M Belin, D J Ball, J C
Langer, et al. “The effect of age on peripheral motor nerve function after
crush injury in the rat.” J Trauma, vol. 40, no. 5, pp. 775-777, 1996.
View at: Publisher Site | PubMed
[30] Zhou YQ, Chen L, Gu YD
“Study of expression of slit2 mRNA in rats of different age after sciatic nerve
injury.” Zhonghua Yi Xue Za Zhi, vol. 84, no. 13, pp. 1110-1113, 2004.
[31] F. Johannes Plate,
Jiaozhong Cai, Thomas L. Smith, et al. “The Influence of Age on Chemotactic and
Inflammatory Marker Expression in Rats Following Peripheral Nerve Injury.” J
Hand Surg, vol. 38, no. 10, pp. e40, 2013. View at: Publisher Site
[32] Phillip L Lamoureux,
Matthew R O'Toole, Steven R Heidemann, et al. “Slowing of axonal regeneration
is correlated with increased axonal viscosity during aging.” BMC Neurosci, vol. 11, pp. 140, 2010. View at: Publisher Site | PubMed
[33] Peter J
Apel, Timothy Alton, Casey Northam, et al. “How age impairs the response of the neuromuscular junction to
nerve transection and repair: An experimental study in rats.” J Orthop Res
Off Publ Orthop Res Soc, vol. 27, no. 3, pp. 385-393, 2009. View at: Publisher Site | PubMed
[34] X Navarro, H Kamei, W R
Kennedy “Effect of age and maturation on sudomotor nerve regeneration in mice.”
Brain Res, vol. 447, no. 1, pp. 133-140, 1988. View at: Publisher Site | PubMed
[35] X Navarro, W R Kennedy
“Effect of age on collateral reinnervation of sweat glands in the mouse.” Brain
Res, vol. 463, no. 1, pp. 174-181, 1988. View at: Publisher Site | PubMed
[36] T B Le, O Aszmann, Y G
Chen, et al. “Effects of pathway and neuronal aging on the specificity of motor
axon regeneration.” Exp Neurol, vol. 167, no. 1, pp. 126-132, 2001. View
at: Publisher Site | PubMed
[37] Bradley J Willcox, James N
Scott “Growth-associated proteins and regeneration-induced gene expression in
the aging neuron.” Mech Ageing Dev, vol. 125, no. 7, pp. 513-516, 2004.
View at: Publisher Site | PubMed
[38] Jami L Scheib, Ahmet Höke
“An attenuated immune response by Schwann cells and macrophages inhibits nerve
regeneration in aged rats.” Neurobiol Aging, vol. 45, pp. 1-9, 2016.
View at: Publisher Site | PubMed
[39] A C Fullarton, D V
Lenihan, L M Myles, et al. “Obstetric brachial plexus palsy: a large animal
model for traction injury and its repair. Part 1: age of the recipient.” J
Hand Surg Edinb Scotl, vol. 25, no. 1, pp. 52-57, 2000. View at: Publisher Site | PubMed
[40] Chong Jian Zhou, Masaru
Kawabuchi, Songyan Wang, et al. “Age differences in morphological patterns of
axonal sprouting and multiple innervation of neuromuscular junctions during
muscle reinnervation following nerve crush injury.” Ann Anat Anat Anz Off
Organ Anat Ges, vol. 184, no. 5, pp. 461-472, 2002. View at: Publisher Site | PubMed
[41] Shimura K “Study on
differences in regeneration of peripheral nerves with age (based on
histological data).” J Showa Med Assoc, vol. 33, no. 6, pp. 791-822,
1973.
[42] Weixiao Huang, Sheng Yi,
Lili Zhao “Genetic Features of Young and Aged Animals After Peripheral Nerve
Injury: Implications for Diminished Regeneration Capacity.” Cell Mol
Neurobiol, vol. 43, no. 8, pp. 4363-4375, 2023. View at: Publisher Site | PubMed
[43] Yong-Jun Wang 1,
Chong-Jian Zhou, Qi Shi, et al. “Aging delays the regeneration process
following sciatic nerve injury in rats.” J Neurotrauma, vol. 24, no. 5,
pp. 885-894, 2007. View at: Publisher Site | PubMed
[44] Hu Liang Low, Antal
Nogradi, Gerta Vrbová, et al. “Axotomized motoneurons can be rescued from cell
death by peripheral nerve grafts: the effect of donor age.” J Neuropathol
Exp Neurol, vol. 62, no. 1, pp. 75-87, 2003. View at: Publisher Site | PubMed
[45] S J Choi, K Harii, M J
Lee, et al. “Electrophysiological, morphological, and morphometric effects of
aging on nerve regeneration in rats.” Scand J Plast Reconstr Surg Hand Surg,
vol. 29, no. 2, pp. 133-140, 1995. View at: Publisher Site | PubMed
[46] Hiroyuki Obata, Kiyohito
Naito, Ayaka Kikui, et al. “Age‑related differences for
expression of the nerve‑specific proteins after
peripheral nerve injury.” Exp Ther Med, vol. 24, no. 5, pp. 682, 2022. View at:
Publisher Site | PubMed
[47] Yoshihiro Sowa, Tetsuya
Imura, Toshiaki Numajiri, et al. “Adipose-derived stem cells produce factors
enhancing peripheral nerve regeneration: Influence of age and anatomic site of
origin.” Stem Cells Dev, vol. 21, no. 11, pp. 1852-1862, 2012. View at: Publisher Site | PubMed
[48] Jason R Hess, Michael J
Brenner, Terence M Myckatyn, et al. “Influence of aging on regeneration in
end-to-side neurorrhaphy.” Ann Plast Surg, vol. 57, no. 2, pp. 217-222,
2006. View at: Publisher Site | PubMed
[49] Takuya Yokoi, Takuya
Uemura, Kiyohito Takamatsu, et al. “Bioabsorbable nerve conduits coated with
induced pluripotent stem cell-derived neurospheres enhance axonal regeneration
in sciatic nerve defects in aged mice.” J Biomed Mater Res PART B-Appl
Biomater, vol. 106, no. 5, pp. 1752-1758, 2018. View at: Publisher Site | PubMed
[50] T David
Luo, Timothy B Alton, Peter J Apel, et al. “Effects of age and insulin-like growth factor-1 on rat
neurotrophin receptor expression after nerve injury.” Muscle Nerve, vol.
54, no. 4, pp. 769-775, 2016. View at: Publisher Site | PubMed
[51] R Cuppini “Age-related
changes in maturation of regenerated motor innervation.” J Neurol Sci,
vol. 114, no. 1, pp. 99-103, 1993. View at: Publisher Site | PubMed
[52] M Kawabuchi, Z Chongjian,
A T Islam, et al. “The effect of aging on the morphological nerve changes
during muscle reinnervation after nerve crush.” Restor Neurol Neurosci,
vol. 13, no. 3-4, pp. 117-127, 1998. View at: PubMed
[53] M Kawabuchi, C J Zhou, S
Wang, et al. “The spatiotemporal relationship among Schwann cells, axons and
postsynaptic acetylcholine receptor regions during muscle reinnervation in aged
rats.” Anat Rec, vol. 264, no. 2, pp. 183-202, 2001. View at: Publisher Site | PubMed
[54] Ayaka Kaneko, Kiyohito
Naito 1, Shinji Nakamura, et al. “Influence of aging on the peripheral nerve
repair process using an artificial nerve conduit.” Exp Ther Med, vol.
21, no. 2, pp. 1-9, 2021. View at: Publisher Site | PubMed
[55] M Streppel, D N Angelov, O
Guntinas-Lichius, et al. “Slow axonal regrowth but extreme hyperinnervation of
target muscle after suture of the facial nerve in aged rats.” Neurobiol
Aging, vol. 19, no. 1, pp. 83-88, 1998. View at: Publisher Site | PubMed
[56] L He, A Yadgarov, S
Sharif, et al. “Aging profoundly delays functional recovery from gustatory
nerve injury.” Neuroscience, vol. 209, pp. 208-218, 2012. View at: Publisher Site | PubMed
[57] Qiuju
Yuan, Huanxing Su, Jiasong Guo, et al. “Decreased
c-Jun expression correlates with impaired spinal motoneuron regeneration in
aged mice following sciatic nerve crush.” Exp Gerontol, vol. 47, no. 4,
pp. 329-336, 2012. View at: Publisher Site | PubMed
[58] E Verdú, D Ceballos, J J
Vilches, et al. “Influence of aging on peripheral nerve function and
regeneration.” J Peripher Nerv Syst, vol. 5, no. 4, pp. 191-208, 2000.
View at: Publisher Site | PubMed
[59] X Navarro, W R Kennedy
“Changes in sudomotor nerve territories with aging in the mouse.” J Auton
Nerv Syst, vol. 31, no. 2, pp. 101-107, 1990. View at: Publisher Site | PubMed
[60] André Jeronimo, Cláudia
Além Domingues Jeronimo, et al. “A morphometric study on the longitudinal and
lateral symmetry of the sural nerve in mature and aging female rats.” Brain
Res, vol. 1222, pp. 51-60, 2008. View at: Publisher Site | PubMed
[61] Stefan Milde, Robert
Adalbert, M Handan Elaman, et al. “Axonal transport declines with age in two
distinct phases separated by a period of relative stability.” Neurobiol
Aging, vol. 36, no. 2, pp. 971-981, 2015. View at: Publisher Site | PubMed
[62] Roberto Pola, Tamar R
Aprahamian, Marta Bosch-Marcé, et al. “Age-dependent VEGF expression and
intraneural neovascularization during regeneration of peripheral nerves.” Neurobiol
Aging, vol. 25, no. 10, pp. 1361-1368, 2004. View at: Publisher Site | PubMed
[63] Uros Kovacic, Tilen Zele,
Tomaz Mars, et al. “Aging impairs collateral sprouting of nociceptive axons in
the rat.” Neurobiol Aging, vol. 31, no. 2, pp. 339-350, 2010. View at: Publisher Site | PubMed
[64] Dingding Shen, Qi Zhang,
Xiaorong Gao, et al. “Age-related changes in myelin morphology,
electrophysiological property and myelin-associated protein expression of mouse
sciatic nerves.” Neurosci Lett, vol. 502, no. 3, pp. 162-167, 2011. View
at: Publisher Site | PubMed
[65] Kadir Ertem, Yurdaer
Denizhan, Saim Yoloğlu, et al. “The effect of injury level, associated injuries, the type of
nerve repair, and age on the prognosis of patients with median and ulnar nerve
injuries.” Acta
Orthop Traumatol Turc, vol.
39, no. 4, pp. 322-327, 2005. View at: PubMed
[66] Elisa
Giorgetti, Michael Obrecht, Marie Ronco, et al. “Magnetic Resonance Imaging as a Biomarker in Rodent
Peripheral Nerve Injury Models Reveals an Age-Related Impairment of Nerve
Regeneration.” Sci Rep, vol. 9, no. 1, pp. 13508, 2019. View at: Publisher Site | PubMed
[67] E Verdú, M Butí, X Navarro
“The effect of aging on efferent nerve fibers regeneration in mice.” Brain
Res, vol. 696, no. 1-2, pp. 76-82, 1995. View at: Publisher Site | PubMed
[68] Michio W Painter, Amanda
Brosius Lutz, Yung-Chih Cheng, et al. “Diminished Schwann Cell Repair Responses
Underlie Age-Associated Impaired Axonal Regeneration.” Neuron, vol. 83,
no. 2, pp. 331-343, 2014. View at: Publisher Site | PubMed
[69] Raman Sharma, Sunil Gaba,
Manish Modi “Age correlation in upper brachial plexus injury patients
undergoing nerve transfer surgeries.” Brain Spine, vol. 2, pp. 101695,
2022. View at: Publisher Site | PubMed
[70] Mariano Socolovsky, Gilda
di Masi, Gonzalo Bonilla, et al. “Age as a Predictor of Long-Term Results in
Patients with Brachial Plexus Palsies Undergoing Surgical Repair.” Oper
Neurosurg, vol. 15, no. 1, pp. 15-24, 2018. View at: Publisher Site | PubMed
[71] Lotta Fornander, Torbjörn
Nyman, Thomas Hansson, et al. “Age- and time-dependent effects on functional
outcome and cortical activation pattern in patients with median nerve injury: a
functional magnetic resonance imaging study.” J Neurosurg, vol. 113, no. 1, pp. 122-128, 2010. View at: Publisher Site | PubMed
[72] Anil
Bhatia, Aditi Kulkarni, Pablo Zancolli, et al. “The Effect of Age and the Delay before Surgery on the
Outcomes of Intercostal Nerve Transfers to the Musculocutaneous Nerve: A
Retrospective Study of 232 Cases of Posttraumatic Total and Near-total Brachial
Plexus Injuries.” Indian J Plast Surg, vol. 53, no. 2, pp. 260-265,
2020. View at: Publisher Site | PubMed
[73] Seung Hoon Lee, Dong Hyun
Kim, Yong Seuk Lee “Is there an optimal age for total knee arthroplasty?: A
systematic review.” Knee Surg Relat Res, vol. 32, no. 1, pp. 60, 2020.
View at: Publisher Site | PubMed
[74] Wei Wang, Sean M Bagshaw,
Colleen M Norris, et al. “Association between older age and outcome after
cardiac surgery: a population-based cohort study.” J Cardiothorac Surg,
vol. 9, no. 1, pp. 177, 2014. View at: Publisher Site | PubMed
[75] Siew Kwaon Lui, Minh Ha
Nguyen “Elderly Stroke Rehabilitation: Overcoming the Complications and Its
Associated Challenges.” Curr Gerontol Geriatr Res, vol. vol. 2018, pp. 9853837,
2018. View at: Publisher Site | PubMed
[76] Lukas Rasulić, Andrija Savić, Bojana Živković, et al. “Outcome after brachial plexus injury surgery and impact on
quality of life.” Acta Neurochir (Wien), vol. 159, no. 7, pp.
1257-1264, 2017. View at: Publisher
Site | PubMed
[77] P D Choi, C B Novak, S E
Mackinnon, et al. “Quality of life and functional outcome following brachial
plexus injury.” J Hand Surg, vol. 22, no. 4, pp. 605-612, 1997. View at:
Publisher Site | PubMed
[78] Sandro Gelsomino, Roberto
Lorusso, Ugolino Livi, et al. “Cost and cost-effectiveness of cardiac surgery
in elderly patients.” J Thorac Cardiovasc Surg, vol. 142, no. 5, pp.
1062-1073, 2011. View at: Publisher Site | PubMed
[79] Samuel T Kunkel, Matthew J
Sabatino, Ravinder Kang, et al. “The Cost-Effectiveness of Total Hip
Arthroplasty in Patients 80 Years of Age and Older.” J Arthroplasty,
vol. 33, no. 5, pp. 1359-1367, 2018. View at: Publisher Site | PubMed
[80] Young Il Won, Chi Heon
Kim, Hee-Pyoung Park, et al. “A cost-utility analysis between decompression
only and fusion surgery for elderly patients with lumbar spinal stenosis and
sagittal imbalance.” Sci Rep, vol. 12, no. 1, pp. 20408, 2022. View at: Publisher Site | PubMed
[81] Zachary S Hubbard, Tsun
Yee Law, Samuel Rosas, et al. “Economic benefit of carpal tunnel release in the
Medicare patient population.” Neurosurg Focus, vol. 44, no. 5, pp. E16,
2018. View at: Publisher Site | PubMed
[82] Becca R Levy “The Role of
Structural Ageism in Age Beliefs and Health of Older Persons.” JAMA Netw
Open, vol. 5, no. 2, pp. e2147802, 2022. View at: Publisher Site | PubMed
[83] Alexander Y Shin, Allen T
Bishop, Michelle F Loosbroch, et al. “A multidisciplinary approach to the
management of brachial plexus injuries: experience from the Mayo Clinic over
100 years.” J Hand Surg Eur Vol, vol. 47, no. 11, pp. 1103-1113, 2022.
View at: Publisher Site | PubMed
[84] Nicholas Pulos 1, Emily H Shin, Robert J Spinner, et al. “Management of Iatrogenic Nerve Injuries.” J Am Acad Orthop Surg, vol. 27, no. 18, pp. e838-e848, 2019. View at: Publisher Site | PubMed
