Received: Sat 29, Mar 2025
Accepted: Wed 16, Apr 2025
Abstract
Background: Acute basilar artery occlusion (BAO) is a devastating form of stroke, often leading to poor outcomes despite advanced recanalization techniques. Atrial fibrillation (AF) is frequently associated with embolic stroke, but its impact on the outcomes of BAO remains debatable. Additionally, the role of bridging thrombolysis needs further exploration.
Methods: This study included 124 patients with acute BAO, divided into AF (n = 57) and non-AF (n = 67) groups. The baseline demographics, use of intravenous thrombolysis (IVT), procedural details and outcomes were compared between the groups. Univariate and multivariate analyses were conducted to identify the predictors of outcomes at discharge and 90 days after treatment.
Results: The patients with AF were older (median age 73 vs. 67 years, P = 0.012) and more likely to have favourable outcomes at discharge (38.6% vs. 19.4%, P = 0.018) and 90 days after treatment (40.4% vs. 23.9%, P = 0.049). AF was independently associated with better functional outcomes at discharge (OR 0.236, 95% CI 0.088 to 0.629; P = 0.004) and 90 days after treatment (OR 0.346, 95% CI 0.143 to 0.841; P = 0.019); however, IVT was not. Lower baseline NIHSS scores and shorter procedure times were also linked to favourable outcomes. However, non-AF patients had higher rates of stent implantation (14.9% vs. 1.8%, P = 0.024) and balloon dilation (41.8% vs. 3.5%, P<0.001), reflecting differences in stroke mechanisms and treatment approaches.
Conclusion: AF was associated with better outcomes of BAO, likely due to differences in stroke pathophysiology and corresponding intervention strategies. Bridging thrombolysis did not affect the functional outcomes of BAO patients, regardless of whether BAO was comorbid with AF. Tailored management strategies based on stroke etiology and patient characteristics are essential for improving outcomes.
Keywords
Acute basilar artery occlusion, atrial fibrillation, bridging thrombolysis, functional outcomes, stroke management
1. Introduction
Acute basilar artery occlusion (BAO) is a catastrophic subtype of acute ischemic stroke that is associated with high mortality and disability rates [1]. Owing to the unique vascular anatomy of the posterior circulation and the limited blood flow of the collateral circulation, the basilar artery may become occluded and therefore lead to devastating outcomes [2]. Although BEST [3] and BASICS [4] revealed the noninferiority of endovascular thrombectomy (EVT) in patients with BAO, BACHE [5] and ATTENTION [6] revealed that EVT is recommended for BAO on the basis of higher-level evidence in the latest guidelines [7].
Despite advances in endovascular therapy, the optimal management strategy remains unclear. Moreover, the impact of atrial fibrillation (AF) and the use of bridging thrombolysis on outcomes also remains unclear. AF is a well-recognized risk factor for cardioembolic stroke, which not only differs from large artery atherosclerosis in terms of its pathophysiology but may also respond differently to treatment [8]. The influence of AF on the prognosis of BAO remains underexplored, thus warranting further investigation.
In recent trials, the researchers highlighted the benefits of EVT for BAO [5, 6] and provided insights into the role of adjunctive therapies, such as intravenous thrombolysis (IVT), to EVT in patients with AF [9]. While some studies indicated that IVT before EVT improves outcomes by increasing early recanalization rates [10, 11], others revealed that its added value is questionable because of concerns about hemorrhage complications and procedural delays [12]. Understanding the interplay between AF, IVT, and EVT in patients with BAO is crucial for optimizing treatment protocols and improving patient outcomes.
The aim of this study was to evaluate the effects of AF and bridging thrombolysis on the outcomes of acute BAO and therefore provide insights into the influence of these factors on clinical decision-making. By analyzing the baseline characteristics, procedural details, and functional outcomes of a well-defined patient cohort, this research seeks to identify the key predictors of a favourable prognosis and refine treatment strategies for this challenging condition. We hypothesized that the presence of AF, along with appropriate use of bridging thrombolysis, would improve functional outcomes in patients with BAO, potentially because of differences in the etiology of stroke and response to therapy.
2. Methods
2.1. Study Design and Population
This cohort study included consecutive patients from Northern Jiangsu People’s Hospital. Patients with acute BAO who underwent EVT were enrolled from January 2018 to March 2024. The study population was divided according to the presence of comorbid AF. The inclusion criteria were as follows: patients with BAO confirmed via computed tomography angiography (CTA) or digital subtraction angiography (DSA) and an onset of symptoms within 24 hours. Patients with pre-existing severe disabilities (mRS score ≧ 3) or contraindications for endovascular therapy were excluded. All treatments were performed in accordance with the Chinese guidelines for the early management of acute ischemic stroke [13]. The study was approved by the local institutional ethics committee. Informed consent was obtained from the patients’ legal representatives before inclusion in the study.
2.2. Surgical Procedure
Bridging thrombolysis with intravenous alteplase was administered prior to EVT in eligible patients. Mechanical thrombectomy was performed using a stent retriever or aspiration catheter, with the choice of device determined by the treating neuro-interventionalist. In cases requiring additional interventions, balloon dilation or stent implantation was performed. The door-to-puncture time (DPT), time from onset to puncture (OPT), time from puncture to reperfusion (PRT), and time from onset to reperfusion (ORT) were also recorded. All patients were admitted to the neurologic intensive care unit or stroke unit and underwent CT within 24 hours after treatment.
2.3. Data Collection
The baseline characteristics included age, sex, and smoking and drinking history. Whether the patient had hypertension, diabetes mellitus, coronary artery disease or a previous ischemic stroke was recorded at the time of the medical history review. Stroke severity was assessed using the National Institutes of Health Stroke Scale (NIHSS) at admission and discharge. Imaging data, including pre-treatment ASPECT scores and thrombolysis in cerebral infarction (TICI) scores, were evaluated by independent radiologists who were blinded to patient outcomes.
2.4. Primary and Secondary Outcomes
Functional outcomes were assessed using the modified Rankin scale (mRS) at discharge and 90 days after treatment, with an mRS score of 0-2 considered indicative of a favourable outcome. The primary outcome was a good functional status at 90 days after treatment. The secondary outcome was a good functional status at discharge. The safety outcomes included in-hospital mortality, brain herniation, haemorrhage and symptomatic intracerebral haemorrhage (sICH) according to the modified safe implementation of thrombolysis in stroke-monitoring study (SITS-MOST) criteria [14].
2.5. Statistical Analysis
Continuous variables are expressed as means ± standard deviations (SDs) or medians (interquartile ranges) and were compared using Student’s t test or the Mann-Whitney U test, as appropriate. Categorical variables were compared using the chi-square test or Fisher’s exact test. Predictors were identified using the logistic regression model for functional outcomes, presented with odds ratios (ORs), 95% confidence intervals (95% CIs) and P values. An ordinal logistics analysis was performed to assess the mRS score at discharge and 90 days after treatment in patients with and without AF. A P value <0.05 indicated statistical significance. All the data were analyzed using SPSS 22.0.
3. Results
3.1. Patients Divided According to the Presence of Comorbid AF
A total of 124 patients were consecutively enrolled: 57 with AF and 67 without AF. The median age was 66.77 years, and 76.61% were male. There were 57 (45.97%) patients with AF, 10 (17.54%) of whom were treated with IVT. Among the 67 (54.03%) patients without AF, 8 (11.94%) underwent bridging IVT.
The median age of the patients with AF was 73 years and that of the patients without AF was 67 years (P = 0.012). The sex distribution was similar, with approximately 77% of each group being male (P = 0.956). While hypertension was more common in the non-AF patients (76.1% vs. 61.4%, P = 0.076), diabetes mellitus was significantly more common in the non-AF patients (37.3% vs. 10.5%, P = 0.001). No significant differences were detected in the ischemic cerebrovascular disease rate, coronary artery disease rate, or proportion of patients with a history of smoking or alcohol consumption. The blood pressure and stroke severity scores at admission were comparable between the groups, as were the initial imaging findings.
However, treatment approaches varied significantly. The non-AF patients had higher rates of balloon dilation (41.8% vs. 3.5%, P <0.001) and stent implantation (14.9% vs. 1.8%, P = 0.024) and greater usage of tirofiban (61.2% vs. 5.3%, P < 0.001) than the AF patients did. The AF patients were more likely to have distal basilar artery occlusion (45.6% vs. 9.0%, P < 0.001). During the procedure, the AF patients had a shorter PRT (60 min vs. 99 min, P < 0.001) than the non-AF patients did (Table 1).
Table.
1. Baseline
characteristics of acute basilar artery occlusion patients with and without
atrial fibrillation who underwent endovascular thrombectomy.
|
Variables |
AF (N = 57) |
non-AF (N = 67) |
P Value |
|
Age |
73(62.5-78.5) |
67(57-72) |
0.012 |
|
Male |
30(76.9%) |
65(76.5%) |
0.956 |
|
Hypertension |
35(61.4%) |
51(76.1%) |
0.076 |
|
Diabetes mellitus |
6(10.5%) |
25(37.3%) |
0.001 |
|
Previous stroke/TIA |
9(15.8%) |
8(11.9%) |
0.535 |
|
Coronary artery disease |
4(7.0%) |
3(4.5%) |
0.826 |
|
Smoking |
12(21.1%) |
15(22.4%) |
0.857 |
|
SBP at admission |
149.47±18.66 |
150.76±19.20 |
0.707 |
|
NIHSS score at admission |
28(16.5-37) |
24(12-33) |
0.119 |
|
Pre-treatment ASPECT |
10(8.25-10) |
10(10-10) |
0.473 |
|
DPT |
147110-209) |
148(108-243) |
0.373 |
|
OPT |
315(240-500) |
360(250-500) |
0.185 |
|
PRT |
60(48.5-95.5) |
99(80-150) |
<0.001 |
|
ORT |
388(282.5-510) |
490(355-620) |
0.12 |
|
Aspiration |
4(7.0%) |
2(3.0%) |
0.533 |
|
Balloon dilation |
2(3.5%) |
28(41.8%) |
<0.001 |
|
Stent implantation |
1(1.8%) |
10(14.9%) |
0.024 |
|
Tirofiban |
3(5.3%) |
41(61.2%) |
<0.001 |
|
BA occlusion site |
|
|
<0.001 |
|
VA-BA |
7(12.3%) |
21(31.3%) |
|
|
Proximal |
11(19.3%) |
26(38.8%) |
|
|
Middle |
13(22.8%) |
14(20.9%) |
|
|
Distal |
26(45.6%) |
6(9.0%) |
|
|
TICI 2b |
49(86.0%) |
53(79.1%) |
0.319 |
|
IVT |
10(17.5%) |
8(11.9%) |
0.377 |
TIA: Transient Ischemic Attack; SBP: Systolic
Pressure; NIHSS: National Institutes of Health Stroke Scale; ASPECTS: Alberta
Stroke Program Early CT Score; DPT: Time from Door to Puncture; OPT: Time from
Onset to Puncture; PRT: Time from Puncture to Reperfusion; ORT: Time from Onset
to Reperfusion; BA: Basilar Artery; VA: Vertebral Artery; TICI: Thrombolysis in
cerebral infarction; IVT: Intravenous Thrombolysis.
3.2. Primary and Secondary Outcomes
At 90 days after treatment, the AF patients had a higher rate of favourable outcomes than the non-AF patients did (40.4% vs. 23.9%, P = 0.049). Similarly, a favourable prognosis at discharge was more common in the AF group than in the non-AF group (38.6% vs. 19.4%, P = 0.018). The NIHSS scores at discharge did not significantly differ, indicating that the severity of stroke was similar upon discharge (P = 0.194). The rates of haemorrhage (25.0% vs. 22.2%, P = 0.727), sICH (11.5% vs. 11.1%, P = 0.943), and brain herniation (21.2% vs. 23.8%, P = 0.735) were comparable between the AF and non-AF group. Notably, in-hospital mortality occurred only in the non-AF group (6.0%), but the difference in the rate was not statistically significant (P = 0.124) (Table 2).
Table.
2. Comparison of key
outcomes between those with and without atrial fibrillation.
|
Outcomes |
AF (N = 57) |
non-AF (N = 67) |
P Value |
|
Good prognosis at 90 days |
23(40.4%) |
16(23.9%) |
0.049 |
|
Good prognosis at discharge |
22(38.6%) |
13(19.4%) |
0.018 |
|
NIHSS score at discharge |
2.5(1-39.25) |
10(3.5-37) |
0.194 |
|
ICH |
13(25.0%) |
14(22.2%) |
0.727 |
|
Symptomatic ICH |
6(11.5%) |
7(11.1%) |
0.943 |
|
Cerebral herniation |
11(21.2%) |
15(23.8%) |
0.735 |
|
In-hospital mortality |
0 |
4(6.0%) |
0.124 |
NIHSS: National Institutes of Health Stroke Scale; ICH: Intracranial Hemorrhage.
3.3. AF and IVT are Associated with Functional Outcomes at Discharge
There were 35 patients with good functional outcomes at discharge. The proportion of AF in these patients was 62.9%, higher than the rate of AF (39.3%) in patients with poor functional outcomes (P = 0.018). There were no significant differences in the proportion of patients who underwent IVT between the two groups (22.9% vs. 11.2%, P = 0.098). The NIHSS score at admission was significantly lower in the favourable prognosis group (17 vs. 30, P = 0.001). The patients with good outcomes had shorter PRT (60 vs. 90 mins, P<0.001) and shorter ORT (379 vs. 480 mins, P = 0.048). The rate of successful reperfusion (TICI 2b) was higher in the favourable prognosis group (97.1% vs. 76.4%, P = 0.014). Additionally, the post-treatment ASPECT scores were better in the favourable prognosis group (9 vs. 7, P = 0.003) (Table 3).
Table.
3. Baseline
characteristics of acute basilar artery occlusion patients divided according to
favourable outcomes at discharge.
|
Variables |
favourable outcomes (N = 35) |
Poor outcomes (N = 89) |
P Value |
|
Age |
67(55-76) |
69(58.5-75) |
0.607 |
|
Male |
27(77.1%) |
68(76.4%) |
0.93 |
|
Hypertension |
23(65.7%) |
63(70.8%) |
0.581 |
|
Diabetes mellitus |
7(20%) |
25(27%) |
0.42 |
|
Previous stroke/TIA |
5(14.3%) |
12(13.5%) |
0.907 |
|
Coronary artery disease |
2(5.7%) |
5(5.6%) |
0.983 |
|
Atrial fibrillation |
22(62.9%) |
35(39.3%) |
0.018 |
|
Smoking |
10(28.6%) |
17(19.1%) |
0.25 |
|
SBP at admission |
133(133-160) |
150(138.5-162) |
0.388 |
|
NIHSS score at admission |
17(8-30) |
30(20.5-35) |
0.001 |
|
Pre-treatment ASPECT |
10(9-10) |
10(9-10) |
0.581 |
|
DPT |
155(126-236) |
142(106-209.5) |
0.259 |
|
OPT |
320(225-420) |
315(240-500) |
0.498 |
|
PRT |
60(50-90) |
90(72.5-137.5) |
<0.001 |
|
ORT |
379(300-480) |
480(351.5-600) |
0.048 |
|
Aspiration |
1(2.9%) |
5(5.6%) |
0.857 |
|
Balloon dilation |
4(11.4%) |
26(29.2%) |
0.065 |
|
Stent implantation |
0 |
11(12.4%) |
0.033 |
|
Tirofiban |
11(31.4%) |
33(37.1%) |
0.554 |
|
BA occlusion site |
|
|
0.015 |
|
VA-BA |
5(14.3%) |
23(25.8%) |
|
|
Proximal |
9(25.7%) |
28(31.5%) |
|
|
Middle |
16(45.7%) |
16(18%) |
|
|
Distal |
5(14.3%) |
22(24.7%) |
|
|
TICI 2b |
34(97.1%) |
68(76.4%) |
0.014 |
|
IVT |
8(22.9%) |
10(11.2%) |
0.098 |
TIA: Transient Ischemic Attack; SBP: Systolic Pressure; NIHSS: National Institutes of Health Stroke Scale; ASPECTS: Alberta Stroke Program Early CT Score; DPT: Time from Door to Puncture; OPT: Time from Onset to Puncture; PRT: Time from Puncture to Reperfusion; ORT: Time from Onset to Reperfusion; BA: Basilar Artery; VA: Vertebral Artery; TICI: Thrombolysis in Cerebral Infarction; IVT: Intravenous Thrombolysis.
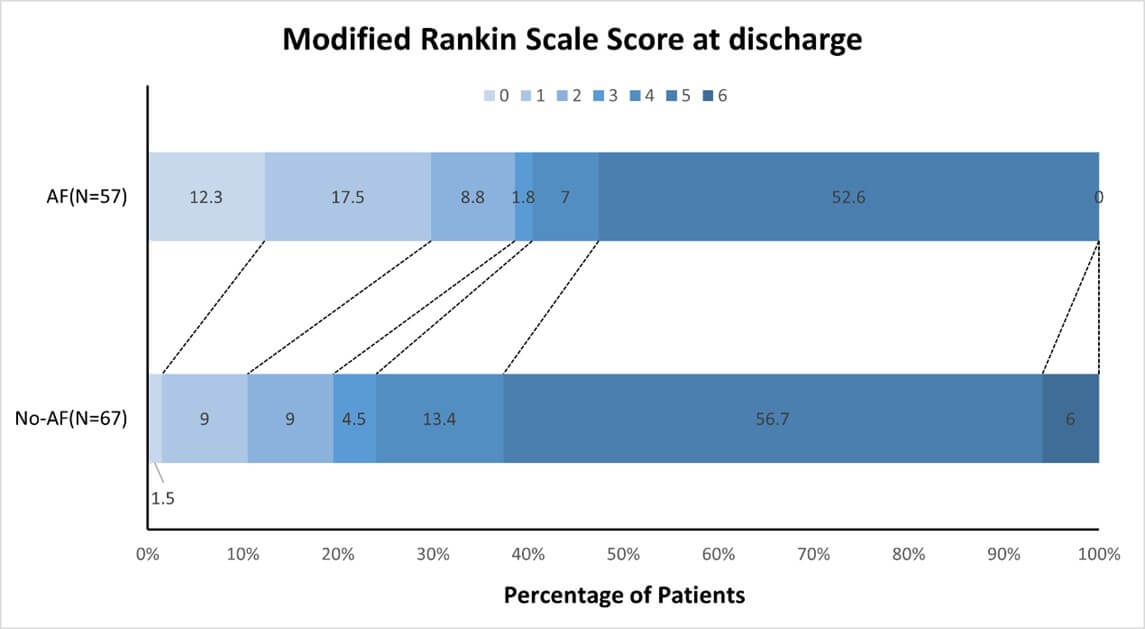
The logistic regression model revealed that AF (OR 0.236, 95% CI 0.088 to 0.629, P = 0.004), the NIHSS score at admission (OR 1.120, 95% CI 1.064-1.180, P < 0.001) and the ORT (OR 1.002, 95% CI 1.0-1.004, P = 0.024) were significant predictors of a favourable prognosis at discharge and that IVT was not (OR 0.303, 95% CI 0.086 to 1.066, P = 0.063) (Table 4). According to the ordinal shift analysis, comorbid AF was associated with a favourable shift in the mRS score at discharge (OR -1.137, 95% CI -1.869 to -0.405; P = 0.002) (Figure 1).
Table.
4. Multivariate
analysis of the association of atrial fibrillation and intravenous thrombolysis
with functional outcomes at discharge.
|
Variables |
B |
OR |
95% CI |
P Value |
|
AF |
-1.446 |
0.236 |
0.088-0.629 |
0.004 |
|
IVT |
-1.193 |
0.303 |
0.086-1.066 |
0.063 |
|
NIHSS at admission |
0.113 |
1.120 |
1.064-1.180 |
<0.001 |
|
ORT |
0.002 |
1.002 |
1.000-1.004 |
0.024 |
AF: Atrial Fibrillation; IVT: Intravenous Thrombolysis; NIHSS: National Institutes of Health Stroke Scale; ORT: Time from Onset to Reperfusion.
3.4. AF and IVT are Associated with Functional Outcomes at 90 days after Treatment
The percentage of patients with atrial fibrillation in the group with good functional outcomes at 90 days was significantly greater than that in the group with poor functional outcomes (59% vs. 40%, P = 0.049). There were more patients who underwent IVT in the group with good functional outcomes, but the difference was not significant (23.1% vs. 10.6%, P = 0.067). The NIHSS score at admission was notably lower in the good prognosis group (20 vs. 30, P = 0.007). Additionally, the patients with favourable outcomes had significantly better post-treatment ASPECT scores (9 vs. 7, P = 0.001) and a higher rate of successful reperfusion (97.4% vs. 75.3%, P = 0.003). The procedural factors also varied, with shorter PRT being associated with better outcomes (60 vs. 90 min, P < 0.001). Balloon dilation and stent implantation were less common in patients with favourable outcomes (P = 0.045 and P = 0.019, respectively) (Table 5).
Table.
5. Baseline
characteristics of acute basilar artery occlusion patients divided according to
favourable outcomes at 90 days.
|
Variables |
favourable outcomes (N = 39) |
Poor outcomes (N = 85) |
P value |
|
Age |
64.05±13.91 |
68.01±10.81 |
0.121 |
|
Male |
30(76.9%) |
65(76.5%) |
0.956 |
|
Hypertension |
25(64.1%) |
61(71.8%) |
0.39 |
|
Diabetes mellitus |
7(17.9%) |
24(28.2%) |
0.219 |
|
Previous stroke/TIA |
6(15.4%) |
11(12.9%) |
0.713 |
|
Coronary artery disease |
2(5.1%) |
5(5.9%) |
1 |
|
Atrial fibrillation |
23(59%) |
34(40%) |
0.049 |
|
Smoking |
10(25.6%) |
17(20%) |
0.48 |
|
SBP at admission |
148.36±17.92 |
151±19.36 |
0.472 |
|
NIHSS score at admission |
20(10-31) |
30(19.5-35) |
0.007 |
|
Pre-treatment ASPECT |
10(9-10) |
10(9-10) |
0.633 |
|
DPT |
155(124-236) |
142(106-209.5) |
0.379 |
|
OPT |
319(215-420) |
320(245-515) |
0.27 |
|
PRT |
60(55-90) |
90(72.5-137.5) |
<0.001 |
|
ORT |
379(300-480) |
480(351.5-600) |
0.27 |
|
Aspiration |
1(2.6%) |
5(5.9%) |
0.727 |
|
Balloon dilation |
5(12.8%) |
25(29.4%) |
0.045 |
|
Stent implantation |
0 |
11(12.9%) |
0.019 |
|
Tirofiban |
14(35.9%) |
30(35.3%) |
0.948 |
|
BA occlusion site |
|
|
0.015 |
|
VA-BA |
6(15.4%) |
22(25.9%) |
|
|
Proximal |
11(28.2%) |
26(30.6%) |
|
|
Middle |
17(43.6%) |
15(17.6%) |
|
|
Distal |
5(12.8%) |
22(25.9%) |
|
|
TICI 2b |
38(97.4%) |
64(75.3%) |
0.003 |
|
IVT |
9(23.1%) |
9(10.6%) |
0.067 |
TIA: Transient Ischemic Attack; SBP: Systolic Pressure; NIHSS: National Institutes of Health Stroke Scale; ASPECTS: Alberta Stroke Program Early CT Score; DPT: Time from Door to Puncture; OPT: Time from Onset to Puncture; PRT: Time from Puncture to Reperfusion; ORT: Time from Onset to Reperfusion; BA: Basilar Artery; VA: Vertebral Artery; TICI: Thrombolysis in Cerebral Infarction; IVT: Intravenous Thrombolysis.
Logistic regression analysis revealed that AF (OR 0.346, 95% CI 0.143 to 0.841, P = 0.019), the NIHSS score at admission (OR 1.083, 95% CI 1.037-1.131, P < 0.001) and the PRT (OR 1.019, 95% CI 1.007-1.032, P = 0.002) were significant factors associated with a favourable prognosis at 90 days after treatment, whereas IVT (OR 0.325, 95% CI 0.101-1.045, P = 0.059) had a less definitive association (Table 6). The ordinal shift analysis revealed that AF was not significantly associated with the mRS score at 90 days after treatment (OR -0.557, 95% CI -1.235 to 0.120, P = 0.107) (Figure 2).
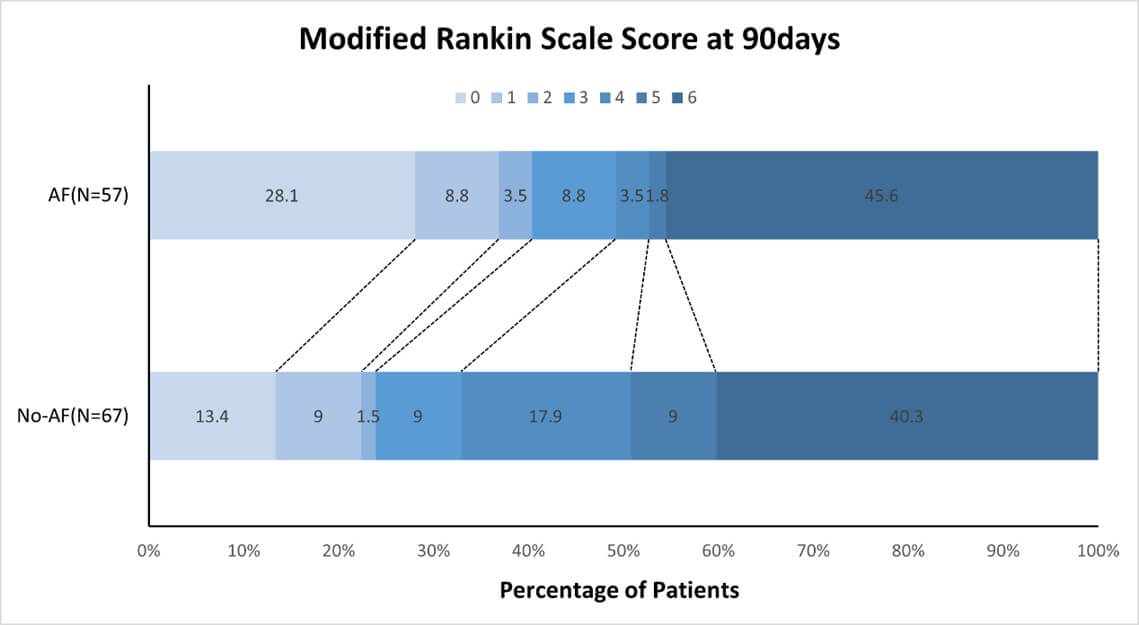
Table.
6. Multivariate
analysis of the association of atrial fibrillation and intravenous thrombolysis
with functional outcomes at 90 days.
|
Variables |
B |
OR |
95% CI |
P value |
|
AF |
-1.060 |
0.346 |
0.143-0.841 |
0.019 |
|
IVT |
-1.125 |
0.325 |
0.101-1.045 |
0.059 |
|
NIHSS score at admission |
0.090 |
1.095 |
1.045-1.146 |
<0.001 |
|
ORT |
0.002 |
1.002 |
1.000-1.003 |
0.028 |
AF: Atrial Fibrillation; IVT: Intravenous Thrombolysis; NIHSS: National Institutes of Health Stroke Scale; ORT: Time from Onset to Reperfusion.
4. Discussion
The major findings of this study indicate that AF patients with acute BAO tend to be older and have a more favourable neurological status than non-AF patients do. Specifically, the AF patients had higher rates of good prognosis at discharge and 90 days after treatment, despite similar overall stroke severity scores and complication rates during hospitalization. The key predictors of favourable outcomes included AF, lower admission NIHSS score and shorter ORT, in addition to IVT. Additionally, procedural differences, such as the less frequent use of balloon dilation and stent implantation in AF patients, were notable. These findings underscore the importance of early and effective interventions and highlight distinct management strategies that could optimize recovery and long-term prognosis in this patient population.
The results of our study align with and differ from those of previous studies on acute basilar artery occlusion in patients with AF. As in our study, prior studies have revealed that patients with AF are typically older and have a lower prevalence of vascular risk factors, such as hypertension and diabetes [15]. However, our study indicated that AF patients had significantly better outcomes, as measured by favourable prognosis at discharge and 90 days after treatment, which contrasts with some earlier reports suggesting worse outcomes in AF patients due to greater risk of hemorrhage transformation [16]. The higher rates of successful reperfusion (TICI 2b) in AF patients in our study might explain this discrepancy, as timely and effective reperfusion has been shown to significantly improve outcomes [17].
Additionally, the predictive value of lower admission NIHSS scores and shorter ORT aligns with literature emphasizing the importance of rapid and efficient treatment for improving stroke outcomes [18]. Our findings regarding procedural differences, such as less frequent balloon dilation and stent implantation in AF patients, add a novel aspect to the current understanding and suggest the need for tailored interventional strategies. These similarities and differences highlight the evolving landscape of stroke management in patients with AF and underscore the importance of continuous evaluation and adaptation of treatment protocols to optimize patient outcomes [19, 20].
A unique aspect of our study is the comprehensive evaluation of procedural characteristics and their impact on patient outcomes, particularly focusing on the differences in treatment modalities between AF patients and non-AF patients with acute basilar artery occlusion. Our analysis included detailed assessments of the number of stent retrieval attempts, PRT, and use of specific interventional techniques, such as balloon dilation and stent implantation. Notably, we found that AF patients, despite being older, had better functional outcomes, which can be attributed to a higher rate of successful reperfusion (TICI 2b) and shorter PRT [18].
Additionally, the study identified that AF patients were less likely to undergo balloon dilation and stent implantation than non-AF patients were, suggesting that simpler and potentially less invasive procedures might be sufficient for achieving favourable outcomes in this subgroup. This finding is consistent with those of recent studies that emphasize the importance of minimizing procedural complexity to reduce the risk of complications and improve recovery [21]. Furthermore, our logistic regression analysis identified lower NIHSS scores at admission and shorter PRT were significant predictors of favourable prognosis, underscoring the importance of rapid intervention and initial assessment of stroke severity in determining patient outcomes [20, 21]. These methodological insights contribute to the growing body of evidence supporting the optimization of endovascular procedures tailored to patient-specific factors, ultimately enhancing the effectiveness of stroke management strategies.
Despite these significant findings, our study has several limitations that must be acknowledged. First, the study’s retrospective design may introduce selection bias and limit the generalizability of the results. The relatively small sample size, particularly when stratified into AF and non-AF groups, may reduce the statistical power and reliability of some findings. Additionally, the single-centre nature of the study limits the applicability of the results to broader, more diverse populations. Furthermore, potential confounding factors, such as variations in prehospital care and in-hospital treatment protocols, were not fully controlled, which could influence the outcomes.
5. Conclusion
This study highlights the significant impact of atrial fibrillation on the functional outcomes of patients with acute basilar artery occlusion, emphasizing the need for tailored treatment strategies. Bridging thrombolysis did not affect the functional outcomes of BAO patients, regardless of whether BAO was comorbid with AF. The need for rapid and effective reperfusion therapies is clear, yet the study's single-centre nature present limitations. Future research should focus on validating these results in larger, multicentre cohorts to increase their generalizability. Prospective studies with extended follow-up periods are essential for evaluating the long-term benefits and risks of various treatment modalities. Additionally, the integration of advanced imaging techniques and novel interventional strategies should be explored to optimize patient outcomes. Addressing potential confounding factors and variations in prehospital and in-hospital care will be crucial for a comprehensive understanding of optimal management strategies. Through these efforts, we aim to improve the quality of care and outcomes for patients with this severe form of stroke, ultimately contributing to better clinical practices and patient prognoses.
Conflicts of Interest
None.
Funding
This work was supported by the Fund for Natural Science Foundation of Jiangsu Province (BK20221280), National Natural Science Foundation of China (No.82371336, No.82172603), the Chinese Postdoctoral Science Foundation (2022M711426), the Collaborative Research Program of Chinese and Western Medicine for Major Difffcult Diseases in Northern Jiangsu People’s Hospital (ZXXTGG2022B02) , the Youth Research Program of Northern Jiangsu People’s Hospital (SBQN23004), the Special Fund for Social Key Research and Development Plan of Yangzhou City (YZ2024068), Social Development Fund of Yangzhou Science and Technology Plan (YZ2024091) and Natural Science Foundation of Jiangsu province for Excellent Young Scholars (BK20231536).
REFERENCES
[1] Heinrich
P Mattle, Marcel Arnold, Perttu J Lindsberg, et al. “Basilar artery occlusion.”
Lancet Neurol, vol. 10, no. 11, pp. 1002-1014, 2011. View at: Publisher Site | PubMed
[2] Nitin
Goyal, Georgios Tsivgoulis, Chris Nickele, et al. “Posterior circulation CT
angiography collaterals predict outcome of endovascular acute ischemic stroke
therapy for basilar artery occlusion.” J Neurointerv Surg, vol. 8, no.
8, pp. 783-786, 2016. View at: Publisher Site | PubMed
[3] Xinfeng
Liu, Qiliang Dai, Ruidong Ye, et al. “Endovascular treatment versus standard
medical treatment for vertebrobasilar artery occlusion (BEST): a randomised
controlled trial.” Lancet Neurol, vol. 19, no. 12, pp. 1152-1160, 2020.
View at: Publisher Site | PubMed
[4] Lucianne
C M Langezaal, Erik J R J van der Hoeven, Francisco J A Mont'Alverne, et al.
“Endovascular therapy for stroke due to basilar-artery occlusion.” N Engl J
Med, vol. 384, no. 20, pp. 1910-1920, 2021. View at: Publisher
Site | PubMed
[5] Tudor
G Jovin, Chuanhui Li, Longfei Wu, et al. “Trial of Thrombectomy 6 to 24 Hours
after Stroke Due to Basilar-Artery Occlusion.” N Engl J Med, vol. 387,
no. 15, pp. 1373-1384, 2022. View at: Publisher Site | PubMed
[6] Chunrong
Tao, Raul G Nogueira, Yuyou Zhu, et al. “Trial of endovascular treatment of
acute basilar-artery occlusion.” N Engl J Med, vol. 387, no. 15, pp.
1361-1372, 2022. View at: Publisher Site | PubMed
[7] Chinese
Society of Neurology, Chinese Stroke Society “Chinese guidelines for diagnosis
and treatment of acute ischemic stroke 2023.” Chinese Journal of Neurology,
vol. 57, no. 6, pp. 523-559, 2024.
[8] Semih
Giray, Ozcan Ozdemir, Demet F Baş, et al. “Does
stroke etiology play a role in predicting outcome of acute stroke patients who
underwent endovascular treatment with stent retrievers?” J Neurol Sci, vol. 372, pp. 104-109, 2017.
View at: Publisher Site | PubMed
[9] Lihong
Wen, Song Zhang, Kunzhen Wan, et al. “Risk factors of haemorrhagic
transformation for acute ischemic stroke in Chinese patients receiving
intravenous thrombolysis: a meta analysis.” Medicine (Baltimore), vol.
99, no. 7, pp. e18995, 2020. View at: Publisher Site | PubMed
[10]
Julian Maingard, Yasmin Shvarts,
Ronan Motyer, et al. “Outcomes of endovascular thrombectomy with and without
bridging thrombolysis for acute large vessel occlusion ischemic stroke.” Intern
Med J, vol. 49, no. 3, pp. 345-351, 2019. View at: Publisher
Site | PubMed
[11]
Keng Siang Lee, Isabel Siow, John Jy
Zhang, et al. “Bridging thrombolysis improves survival rates at 90 days
compared with direct mechanical thrombectomy alone in acute ischemic stroke due
to basilar artery occlusion: a systematic review and meta-analysis of 1096
patients.” J Neurointerv Surg, vol. 15, no. 10, pp. 1039-1045, 2023.
View at: Publisher Site | PubMed
[12]
Jonathan M Coutinho, David S
Liebeskind, Lee-Anne Slater, et al. “Combined intravenous thrombolysis and
thrombectomy vs thrombectomy alone for acute ischemic stroke: a pooled analysis
of the SWIFT and STAR studies.” JAMA Neurol, vol. 74, no. 3, pp.
268-274, 2017. View at: Publisher Site | PubMed
[13]
Chinese Society of Neurology, Chinese
Stroke Society “Chinese guidelines for diagnosis and treatment of acute
ischemic stroke 2018.” Chinese Journal of Neurology, vol. 51, no. 9, pp.
666-682, 2018. View at: Publisher
Site | PubMed
[14]
S Lorenzano, N Ahmed, A Rosselli, et
al. “Safe implementation of thrombolysis in stroke-monitoring study in Italy.” Eur
J Neurol, vol. 17, no. 1, pp. 163-167, 2010. View at: Publisher Site | PubMed
[15]
Benedikt Frank, Rachael Fulton,
Christian Weimar, et al. “Impact of atrial fibrillation on outcome in
thrombolyzed patients with stroke: evidence from the Virtual International
Stroke Trials Archive (VISTA).” Stroke, vol. 43, no. 7, pp. 1872-1877,
2012. View at: Publisher Site | PubMed
[16]
Gustavo Saposnik, David Gladstone,
Roula Raptis, et al. “Atrial fibrillation in ischemic stroke: predicting
response to thrombolysis and clinical outcomes.” Stroke, vol. 44, no. 1,
pp. 99-104, 2013. View at: Publisher Site | PubMed
[17]
Chenhao Zhao, Weidong Luo, Xing Liu,
et al. “Effect of atrial fibrillation on outcomes after mechanical thrombectomy
and long-term ischemic recurrence in patients with acute basilar artery
occlusion.” Front Neurol, vol. 13, pp. 909677, 2022. View at: Publisher Site | PubMed
[18]
Jeffrey L Saver, Mayank Goyal, Aad
van der Lugt, et al. “Time to treatment with endovascular thrombectomy and
outcomes from ischemic stroke: a meta-analysis.” JAMA, vol. 316, no. 12,
pp. 1279-1288, 2016. View at: Publisher Site | PubMed
[19]
Giulia Frauenfelder, Francesco Diana,
Renato Saponiero, et al. “A direct aspiration first-pass technique (ADAPT) for
acute ischemic stroke thrombectomy: Indications, technique, and emerging
devices.” Neuroradiol J, vol. 19, pp. 19714009241303063, 2024. View at: Publisher Site | PubMed
[20]
William J Powers, Alejandro A
Rabinstein, Teri Ackerson, et al. “2018 Guidelines for the early management of
patients with acute ischemic stroke: a guideline for healthcare professionals
from the American Heart Association/American Stroke Association.” Stroke,
vol. 49, no. 3, pp. e46-e110, 2018. View at: Publisher Site | PubMed
[21]
Marc Ribo, Alan Flores,
Marta Rubiera, et al. “Difficult catheter access to the occluded vessel during
endovascular treatment of acute ischemic stroke is associated with worse
clinical outcome.” J Neurointerventional Surg, vol. 5 Suppl 1, pp.
i70-i73, 2013. View at: Publisher Site | PubMed
