Received: Wed 26, Nov 2025
Accepted: Tue 16, Dec 2025
Abstract
Objective: To evaluate the treatment outcomes, angiographic occlusion rates, and safety profiles of endovascular therapy (EVT) versus surgical intervention for intracranial dural arteriovenous fistulas (dAVFs) treated at a single institution over a 10-year period.
Methods: A retrospective review was conducted on 70 adult patients treated for intracranial dAVFs between 2014 and 2024. Inclusion criteria were patients treated with EVT, surgery or combination of both. Primary outcome was initial complete angiographic occlusion. Secondary outcomes included residual shunting, recurrence, and symptom improvement.
Results: Most patients (80%) were male, with a median age of 62 years. EVT was performed in 60%, surgery in 38.6%, and combined treatment in 1.4%. Initial angiographic occlusion was achieved in 69.6%, with no significant difference between EVT (71.4%) and surgery (69.2%). Lesions with cortical venous drainage (Borden III) showed significantly higher occlusion rates (p = 0.027). Final angiographic cure was achieved in 89.9% of patients after additional treatments or spontaneous regression. Symptomatic improvement was observed in 95.7% of patients. Treatment-related complications occurred in 18.6%, with no significant difference between modalities.
Conclusion: Both EVT and surgery are effective and safe for treating intracranial dAVFs. Angio-architectural complexity, fistula location and venous drainage patterns were significantly associated with higher treatment success.
Keywords
Intracranial dural arteriovenous fistula, endovascular treatment, surgical management, cortical venous drainage, Borden classification, Cognard classification
1. Introduction
Dural arteriovenous fistulas (dAVFs) are vascular malformations involving abnormal shunting between meningeal arteries and dural venous sinuses or bridging veins [1]. Intracranial dAVFs constitute approximately 10-15% of all intracranial arteriovenous malformations [2]. These lesions may be incidentally detected or present with pulsatile tinnitus, headache, ocular symptoms, neurological deficits, or intracranial hemorrhage, depending on location and venous drainage [3]. Their clinical significance is determined by venous hemodynamic impact, with several classification systems developed to stratify risk [4-7]. Management is based on clinical presentation, venous drainage type, and angioarchitecture [8]. Treatment options include endovascular therapy (EVT), microsurgical resection, radiosurgery, or combined approaches [9]. EVT has become increasingly favored with the advent of DMSO-based liquid embolic agents, advanced microcatheter technology, and newer imaging techniques that enhance understanding of dAVF angioarchitecture, achieving 50-76% success with low complication rates (2.2-2.5%) [10-16]. Although EVT is often first-line, surgical or combined strategies may yield better results in hemorrhagic cases or surgically accessible lesions [17]. Microsurgery achieves 69-88% obliteration, though morbidity and mortality can reach 13% [3, 18-21]. The comparative advantages of each modality, stratified by classification and clinical context, remain incompletely defined [3]. In the absence of randomized controlled trials, a tailored, patient-specific approach remains essential [3, 22].
The purpose of our study is to assess treatment efficacy, in terms of angiographic occlusion, and procedural safety in patients undergoing endovascular versus surgical intervention for intracranial dAVFs.
2. Methods
The study was approved by the institutional research ethics board. A retrospective review was conducted on all adults treated for intracranial dAVFs between 2014 and 2024. Inclusion criteria were dAVF patients treated by EVT, Surgery or combination of both. Cerebral AVMs, spinal fistulas, carotid-cavernous fistulas, and incomplete records were excluded. Clinical notes, operative reports, Computed tomography (CT), Magnetic resonance imaging (MRI), and digital subtraction angiogram (DSA) of cerebral vessels were reviewed.
Demographics (age, sex, presentation) and treatment modality (endovascular, surgical, combined) were recorded. Post-procedural data included complications, neurological status, and hospital stay. Follow-up clinical status and imaging were obtained from outpatient records and included DSA, CT angiography (CTA), and time-resolved contrast-enhanced MR angiography (MRA). Presenting symptoms were classified as benign (e.g., tinnitus, chronic headache, incidental) or aggressive (e.g., hemorrhage, seizures, focal deficits, cognitive decline) [23, 24]. CTs were reviewed for hemorrhage and mass effect, and MRI sequences (T1, T2, fluid-attenuated inversion recovery [FLAIR], susceptibility-weighted imaging [SWI], and post-contrast T1) were used to assess parenchymal changes, venous congestion, or microhemorrhages. When available, MRA and MR venography (MRV) were assessed to delineate specific vascular anatomy and venous drainage pattern. DSA was used to identify lesion classification, location, arterial supply (from external/internal carotid or vertebral arteries), and venous drainage patterns. Lesions were grouped by location and classified by Borden and Cognard systems [6, 7]
For sinus-draining dAVFs (Borden I/II; Cognard I, IIa, IIb, IIa+b), treatment depended on sinus functionality. Nonfunctional sinuses were treated with transvenous (TV) coil embolization; functional sinuses used transarterial (TA) or balloon-assisted approaches. Surgically treated sinus lesions aimed to disconnect cortical reflux. For non-sinus (Borden III; Cognard III/IV) lesions, the goal was venous outflow interruption via surgical clipping/coagulation or TA embolization with liquid agents; TV embolization was used when arterial access was limited (Figure 1). Primary outcome was initial complete angiographic occlusion. Secondary outcomes included, residual shunting, and recurrence on follow up. Angiographic cure was defined as complete absence of shunting on follow-up, including cases where residual lesions regressed spontaneously. Residual fistula referred to persistent shunting detected immediately or shortly after treatment, indicating incomplete occlusion. Recurrence was defined as reappearance of shunting at a previously obliterated site on delayed imaging after initial cure. In cases with residual lesions, the subsequent clinical course was documented specifically, those that resolved spontaneously versus those requiring additional treatment. Final obliteration rates at last follow-up were also recorded.
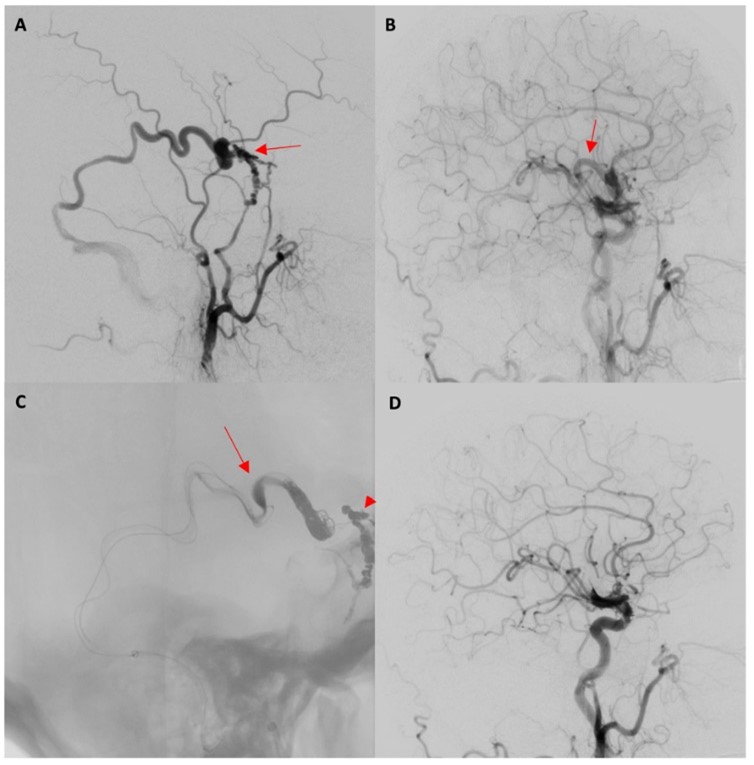
CCA: Common Carotid Artery; DSA: Digital Subtraction Angiogram; ECA: External Carotid Artery; MMA: Middle Meningeal Artery.
Factors influencing treatment selection (surgery vs. EVT) were analyzed for lesion location, clinical presentation, urgency, age, and sex. Risk factors for incomplete occlusion were analyzed, including age, sex, location, Borden/Cognard grade, number of feeders (≤ 3 vs. > 3), unilateral or bilateral feeders, and clinical presentation type. For EVT cases, number of arterial pedicles, use of middle meningeal artery, and embolic material (liquid vs. coils) were examined. Safety outcomes included complications such as dissection, stroke, hemorrhage, venous thrombosis, cranial nerve palsies, and catheter entrapment in EVT cases; and stroke, infection, hemorrhage, and nerve deficits in surgical cases. Mortality was defined as death within 30 days related to disease or treatment.
All statistical analyses were conducted using R (version 4.4.2 in RStudio). Demographic and patient characteristic data were analyzed by using descriptive summary statistics. Categorical variables such as sex, past medical history, etiology, and clinical presentation were reported with frequency distributions and percentages. Numerical variables, such as age, follow-up duration and hospital stay duration were reported as median and interquartile ranges. The Pearson test and Fisher’s exact test was used to identify significant association between occlusion type and categorical variables, as well as between treatment modality preferences and categorical variables.
3. Results
A total of 70 adult patients (median age- 62 yrs; 80% male) with intracranial dAVFs, were identified and included in the study. The detailed demographics and clinical characteristics are summarized in (Table 1). The majority (88.6%) of dAVFs were idiopathic. The median follow-up duration was 24.8 months (IQR 8.8-38.6) and the median hospital stay was 3.5 days (IQR 1-11). Patients with benign symptoms comprised 25.7% who presented incidentally and 20% who presented with flow-related symptoms such as tinnitus. Aggressive presentations were observed in 27.1% of patients who exhibited neurological deficits, and in another 27.1% who presented with intracranial hemorrhage. According to the Borden classification, 84.3% of the lesions were Type III. Under the Cognard classification, 50% were categorized as Type III and 34.3% as Type IV.
Table. 1. Descriptive Statistics of DAVF Patient Cohort.
|
Demographics |
|
|
Age,
median (IQR), years |
62
(51–70) |
|
Sex |
n (%) |
|
Male
|
56
(80%) |
|
Female |
14(20%) |
|
Etiology |
n (%) |
|
Idiopathic |
62
(88.6) |
|
Trauma
/ cranial surgery |
3
(4.3) |
|
Cerebral
venous thrombosis |
5
(7.1) |
|
Pregnancy |
1
(1.4) |
|
Infection |
0
(0) |
|
Medical Comorbidities |
n (%) |
|
Hypertension |
43
(61.4) |
|
Diabetes
mellitus |
17
(24.3) |
|
Coronary
artery disease |
5
(7.1) |
|
Smoking |
16
(22.9) |
|
Prior
stroke |
5
(7.1) |
|
Prior
TIA |
4
(5.7) |
|
Malignancy |
10
(14.3) |
|
Clinical Presentation |
n (%) |
|
Incidental |
18
(25.7) |
|
Flow-related
symptoms |
14
(20.0) |
|
NHND
(focal or global) |
19
(27.1) |
|
Hemorrhage |
19
(27.1) |
|
Lesion Location |
n (%) |
|
Sinus draining |
|
|
Transverse–sigmoid
sinus |
8
(11.4) |
|
Jugular
bulb |
3
(4.3) |
|
Superior Sagittal
Sinus |
0 (0%) |
|
Torcula |
0 (0%) |
|
Non-Sinus draining |
|
|
Parasagittal |
15
(21.4) |
|
Convexity |
15
(21.4) |
|
Cribriform |
8
(11.4) |
|
Tentorial
/ petrous |
12
(17.1) |
|
Sphenoid |
3
(4.3) |
|
Posterior
fossa |
4
(5.7) |
|
Marginal |
2
(2.9) |
|
Angiographic
Classification |
n (%) |
|
Borden |
|
|
Type
I |
4
(5.7) |
|
Type
II |
7
(10.0) |
|
Type
III |
59
(84.3) |
|
Cognard |
n (%) |
|
Type I |
2 (2.9) |
|
Type IIA |
2 (2.9) |
|
Type IIB |
1 (1.4) |
|
Type IIA+B |
6 (8.6) |
|
Type III |
35 (50) |
|
Type IV |
24 (34.3) |
|
Primary Treatment
Modality |
n (%) |
|
Endovascular |
42
(60.9) |
|
Surgery |
27
(39.1) |
|
Combined |
1
(1.4) |
|
Endovascular Approach |
n (%) |
|
Transarterial (TA) |
36 (51.4) |
|
Transvenous (TV) |
2 (2.9) |
|
Transarterial + Transvenous (TA/TV) |
4 (5.7) |
|
Transarterial in Combined |
1 (1.4) |
|
Surgery Type |
n (%) |
|
Ligation of Bridging Vein |
24 (34.3) |
|
Ligation of Bridging Vein, Combined |
1 (1.4) |
|
Disconnection of Cortical venous reflux |
3 (4.3) |
|
Clinical Outcomes |
n (%) |
|
Complete
symptom improvement |
48
(68.6) |
|
Partial
improvement |
19
(27.1) |
|
No
improvement |
1
(1.4) |
|
Angiographic Outcomes |
n (%) |
|
Initial
Angiographic occlusion |
48
(69.6) |
|
Residual
shunting |
21
(30.4) |
|
Spontaneous
regression |
4
(19.0 of residuals) |
|
Additional
treatment required |
11
(52.3 of residuals) |
|
Recurrence |
1
(1.4) |
|
Final
Angiographic cure |
51(73.9) |
|
Mortality, n (%) |
2
(2.8) |
|
Follow-up duration, Median (IQR), months |
24.8
(8.8–38.6) |
|
Hospital stay, days |
Median (IQR) |
|
Overall |
3.5
(1-11) |
|
EVT
- elective |
1
(1–2) |
|
EVT
- emergency |
9.5
(6–13.2) |
|
Surgery
- elective |
2
(2–3.2) |
|
Surgery
- emergency |
20
(6.8–54.5) |
Treatment was primarily endovascular in 60% of patients, surgical in 38.6%, and combined in 1.4% (Table 2). EVT was mostly TA (51.4%), with 4 cases managed using a combined TA/TV approach. Surgical management largely consisted of disconnection of the bridging vein by clipping or coagulation (34.3%), with a smaller subset undergoing disconnection of the refluxing cortical veins. Treatment decisions appeared to be influenced by lesion location and accessibility. For example, all cribriform dAVFs were treated surgically, whereas convexity and parasagittal fistulas were more commonly managed via endovascular routes. Initial angiographic complete occlusion was achieved in 69.6% (48/69) of all treated patients. No significant difference (p = 0.427) was seen in the occlusion rates between the endovascular (71.4%) and surgical (69.2%) groups. One patient (1.4%) was lost to follow-up. Lesions located at the convexity demonstrated a higher likelihood of complete occlusion (p = 0.034), as did dAVFs with fewer than three arterial feeders (p < 0.001) (Table 3).
Table. 2. Initial
Angiographic Occlusion Rates and complete obliteration rate at final follow up
by Treatment Modality and Type of lesion. Overall initial complete angiographic
occlusion for surgical vs. endovascular treatment: 69.2 vs. 71.4 %, p = 0.427.
|
Type
|
Treatment |
Initial
Complete angiographic occlusion |
Residual
flow |
Spontaneous
regression |
Angiographic
cure on follow up |
|
Borden
I, II & Cognard I, IIa, IIb, IIa+b |
Endovascular
(Overall) |
4/8
(50%) |
4/8
(50%) |
|
4/8
(50%) |
|
TA |
2/5
(40%) |
3/5
(60%) |
|
2/5
(40%) |
|
|
TA/TV |
0/1
(0%) |
1/1
(100%) |
|
0/1
(0%) |
|
|
TV |
2/2
(100%) |
0/2
(0%) |
|
2/2
(100%) |
|
|
Surgery |
0/3
(0%) |
3/3
(100%) |
1/3 |
1/3
(33.3%) |
|
|
Combined |
- |
- |
|
- |
|
|
All
treatments |
4/11(36.4%) |
7/11
(63.6%) |
1/7 |
5/11
(45.5%) |
|
|
Borden
III & Cognard III,IV |
Endovascular
(Overall) |
26/34
(76.5%) |
8/34
(23.5%) |
1/8 |
27/34
(79.4%) |
|
TA |
23/31
(74.2%) |
8/31
(25.8%) |
1/8 |
24/31
(77.4%) |
|
|
TA/TV |
3/3(100%) |
0/3
(0%)) |
|
3/3
(100%) |
|
|
TV |
- |
- |
|
- |
|
|
Surgery |
18/23
(78.3%) |
5/23
(21.7%) |
2/5 |
19/23
(82.6%) |
|
|
Combined |
0/1
(0%) |
1/1
(100%) |
|
0/1
(0%) |
|
|
|
All
treatments |
44/58
(75.9%) |
14/58
(24.1%) |
3/14 |
46/58
(79.3%) |
|
All
grades |
Endovascular
(Overall) |
30/42(71.4%) |
12/42(28.6%) |
1/12 |
31/42
(73.8%) |
|
|
Surgery |
18/26
(69.2%) |
8/26
(30.8%) |
3/8 |
20/26(76.9%) |
|
|
Combined |
0/1
(0%) |
1/1
(100%) |
|
0/1
(0%) |
|
Overall
Cohort |
All
treatments |
48/69
(69.6%) |
21/69(30.4%) |
4/21 |
51/69
(73.9%) |
TA: Transarterial; TV: Transvenous.
Table. 3. Predictive Factors Associated with Complete Occlusion.
|
Characteristic |
Complete Occlusion |
Incomplete Occlusion |
p-value |
|
|
|
|
|
|
Sex |
|
|
|
|
Female |
9 |
4 |
1 |
|
Male |
39 |
17 |
|
|
Age Group |
|
|
|
|
<50 |
11 |
3 |
0.732 |
|
>70 |
11 |
6 |
|
|
50-70 |
26 |
12 |
|
|
Location |
|
|
|
|
Convexity |
14 |
1 |
0.034* |
|
Cribriform |
6 |
1 |
|
|
Jugular |
1 |
2 |
|
|
Other Bridging Vein |
15 |
6 |
|
|
Parasagittal |
9 |
6 |
|
|
Transverse-Sigmoid |
3 |
5 |
|
|
Borden Classification |
|
|
|
|
Borden 1/2 |
4 |
7 |
0.027* |
|
Borden 3 |
44 |
14 |
|
|
Cognard Classification |
|
|
|
|
Cognard 1-2 |
4 |
7 |
0.027* |
|
Cognard 3-4 |
44 |
14 |
|
|
Number of Arterial Feeders |
|
|
|
|
>3 feeders |
14 |
16 |
<0.001*** |
|
≤3 feeders |
34 |
5 |
|
|
Laterality |
|
|
|
|
Bilateral |
19 |
13 |
0.147 |
|
Unilateral |
29 |
8 |
|
|
Clinical Presentation |
|
|
|
|
Flow-related |
8 |
6 |
0.440 |
|
Hemorrhage |
12 |
6 |
|
|
Incidental |
15 |
3 |
|
|
NHND |
13 |
6 |
|
|
EVT: Arterial Pedicles
Treateda |
|
|
|
|
>1 pedicles |
4 |
5 |
0.102 |
|
1 pedicle |
23 |
7 |
|
|
EVT: Endovascular Approacha |
|
|
|
|
MMA |
27 |
10 |
0.209 |
|
Other arterial pedicles |
1 |
2 |
|
The type of venous drainage significantly influenced occlusion rates (Tables 2 and 3). Lesions with non-sinus drainage (i.e., Borden Type III or Cognard Types III/IV) demonstrated significantly higher (75.9% vs 36.4%; p = 0.027) initial and final angiographic occlusion compared to fistulas draining into the dural sinuses. Among the 3 surgically treated patients with dural sinus drainage (all Borden Type II), residual fistulas (Type 1 in nature) were noted postoperatively in all cases, with one lesion regressing spontaneously. Eight patients underwent EVT (4 Borden Type I and 4 Borden Type II); 2 patients achieved complete occlusion with TV embolization of nonfunctional sinuses. Among the 6 patients treated with sinus-preserving strategies—5 underwent TA Embolisation and 1 underwent a balloon-assisted TA/TV approach. Initial angiographic cure was achieved in 2 patients (33.3%) and improved to 50% after staged treatment in one additional patient.
In contrast, Borden Type III fistulas showed excellent outcomes (Table 2). Surgical treatment yielded 78.3% (18/23) initial occlusion, with 2 residual fistulas regressing spontaneously and 2 more fistulas cured with adjunctive treatment by EVT. Endovascularly treated Borden III lesions had 76.5% (26/34) initial occlusion. All residual cases (n=8) were eventually cured, 1 by spontaneous regression and 7 following additional surgery. These results underscore the higher treatment efficacy in high-grade (Borden III) lesions and the technical challenges and therapeutic intent associated with sinus-preserving treatment in lower-grade fistulas. Among the 48 patients (69.6%) who achieved complete occlusion on initial post-treatment angiography, 1 patient (2.1% of initially cured) experienced a recurrence. This patient underwent a subsequent endovascular procedure for complete cure.
Of the 21 patients (30.4%) with residual lesions following initial treatment, 11 (52.4% of those with residuals) underwent a total of 13 retreatment procedures, 4 by endovascular means and 9 by surgery, with 2 patients requiring re-operation. Among these 11 patients, 10 (90.9% of retreated) ultimately achieved complete occlusion. Four of the 21 residual lesions (19.0% of residuals) showed spontaneous regression on follow-up imaging. Six patients (28.6% of residuals) with residual disease did not receive further treatment. Of these, 1 patient (1.4% overall) died within 1 month, 1 patient (1.4%) declined additional therapy, and the remaining four (5.7%) had low-grade or stable lesions and are currently being monitored with serial imaging. Overall, at final follow-up, 62 out of 69 patients (89.9%) had achieved complete angiographic cure. Symptoms resolved completely in 48 patients (68.6%), with partial improvement in 19 patients (27.1%) and no improvement in 1 patient (1.4%).
Treatment-related complications (Table 4) were seen in 18.6% of all patients and were not significantly different (p = 0.342) between the endovascular group (21.4%) and the surgical group (14.8%). In the EVT group, complications included arterial dissection (of the middle meningeal artery) (7.1%), stroke (7.1%), catheter-related complications, and cranial nerve injuries. Surgical complications primarily involved hemorrhage (7.4%), stroke (3.7%) and wound infection (3.7%). No major long-term deficits were reported, underscoring the safety of both approaches when appropriately selected.
Table. 4. Patient
based Treatment-Related Complications Summary.
|
Treatment |
Complication |
n (%) |
|
Endovascular |
Overall |
9/42(21.4%)* |
|
Dissection |
3
(7.1%) |
|
|
Catheter
trap |
1
(2.4%) |
|
|
Stroke |
3
(7.1%) |
|
|
CVT |
2
(4.8%) |
|
|
Hemorrhage |
1
(2.4%) |
|
|
Temporary
cranial nerve palsy |
0
(0%) |
|
|
Permanent
cranial nerve palsy |
1
(2.4%) |
|
|
Temporary
motor weakness/aphasia |
2
(4.8%) |
|
|
Permanent
motor weakness/aphasia |
0
(0%) |
|
|
Surgical |
Overall |
4/27(14.8%)* |
|
Stroke |
1
(3.7%) |
|
|
CVT |
0
(0%) |
|
|
Hemorrhage |
2
(7.4%) |
|
|
Temporary
cranial nerve palsy |
0
(0%) |
|
|
Permanent
cranial nerve palsy |
0
(0%) |
|
|
Temporary
motor weakness/aphasia |
0
(0%) |
|
|
Permanent
motor weakness/aphasia |
0
(0%) |
|
|
Wound
infection |
1
(3.7%) |
|
|
Bone
flap necrosis |
0
(0%) |
Table. 5. Treatment
Modality Preferences by Patient Characteristics.
|
Characteristic |
Endovascular n (%) |
Surgery n (%) |
p-value |
|
Location |
|
|
|
|
Convexity |
10
(66.7%) |
5
(33.3%) |
0.002** |
|
Cribriform |
0
(0%) |
8
(100%) |
|
|
Jugular |
2
(66.7%) |
1
(33.3%) |
|
|
Other
Bridging Vein |
12
(57.1%) |
9
(42.9%) |
|
|
Parasagittal |
12
(85.7%) |
2
(14.3%) |
|
|
Transverse-Sigmoid |
6
(75%) |
2
(25%) |
|
|
Presenting Symptoms |
|
|
|
|
Flow-related |
10
(71.4%) |
4
(28.6%) |
0.250 |
|
Hemorrhage |
8
(44.4%) |
10
(55.6%) |
|
|
Incidental |
10
(55.6%) |
8
(44.4%) |
|
|
NHND |
14
(73.7%) |
5
(26.3%) |
|
|
Emergency vs. Elective |
|
|
|
|
Elective |
26
(65%) |
14
(35%) |
0.565 |
|
Emergency/Urgent |
16
(55.2%) |
13
(44.8%) |
|
|
Age Group |
|
|
|
|
<50 |
9
(60%) |
6
(40%) |
0.891 |
|
>70 |
9
(56.2%) |
7
(43.8%) |
|
|
50-70 |
24
(63.2%) |
14
(36.8%) |
|
|
Sex |
|
|
|
|
Female |
9
(64.3%) |
5
(35.7%) |
1 |
|
Male |
33
(60%) |
22
(40%) |
|
***P<.001, **P<.01,
*P<.05. Overall treatment distribution: Endovascular 42 (60.9%), Surgery 27
(39.1%) of 69 patients.
4. Discussion
In our retrospective cohort study, the overall initial complete angiographic occlusion was seen in 69.6% (48/69) of patients, with similar (p = 0.47) outcomes between the endovascular (71.4%) and surgical (69.2%) groups. Angioarchitectural complexity (p =< 0.001), fistula location (p = 0.034) and venous drainage patterns (p = 0.027) significantly influenced treatment success in our series. Previous studies have demonstrated that multiple arterial feeders particularly when small, tortuous, or difficult to access, are associated with lower incidence of complete obliteration [25]. Consistent with these findings, lesions with three or fewer arterial feeders in our cohort were associated with higher incidence of complete occlusion compared to those with more than three feeders. Lesions over the convexity or laterally along the transverse or sigmoid sinus often achieve better occlusion, especially when supplied by a single dominant feeder and draining into a single vein or non-functional sinus [25, 26]. In contrast, midline or paramedian fistulas involving the superior sagittal sinus or torcula (Figures 2 & 3) tend to have lower obliteration rates due to complex bilateral arterial supply [25, 26]. These lesions often require bilateral embolization or disconnection to achieve cure, as unilateral approaches are typically inadequate [27]. In our series, location was significantly associated with treatment outcome (p = 0.034), with convexity lesions demonstrating higher initial occlusion rates than other locations. Although higher rates of residual fistula were noted in bilaterally supplied lesions in our series, this difference did not reach statistical significance when compared to unilaterally supplied cases.
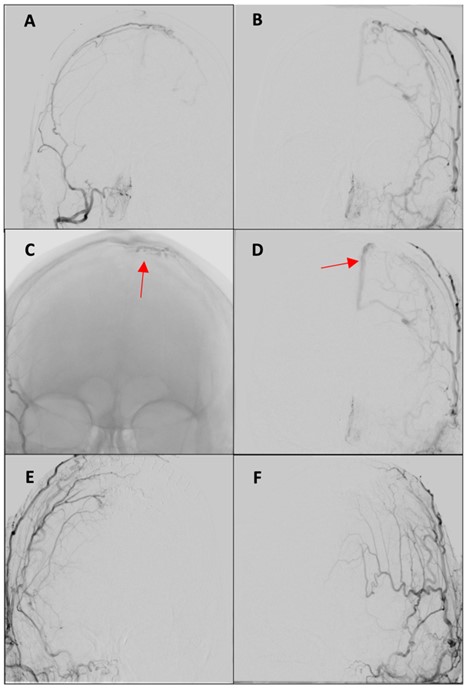
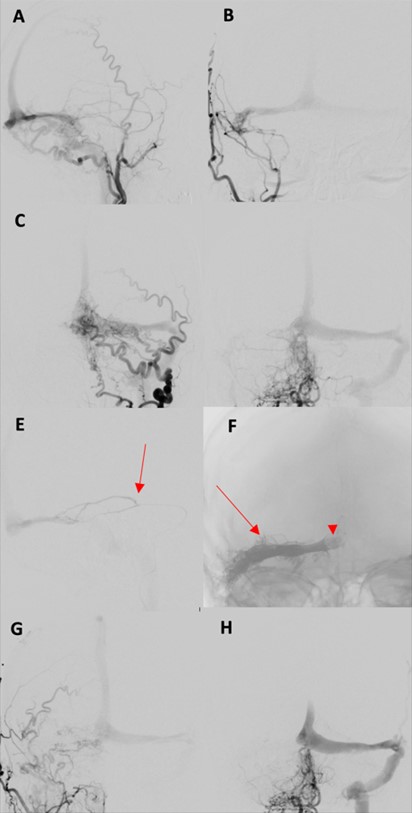
AP: Anteroposterior; DSA: Digital Subtraction Angiogram; ECA: External Carotid Artery; MMA: Middle Meningeal Artery; STA: Superficial Temporal Artery.
Clinical outcomes were largely influenced by the type of venous drainage, with significant differences observed between lesions draining into dural sinuses and those draining directly into cortical (non-sinus) veins [6, 7]. In our series, lesions with non-sinus drainage patterns demonstrated significantly higher angiographic occlusions (p = 0.027). Surgical disconnection is remains the most definitive approach for non-sinus draining fistulas, particularly when the lesion drains into a single leptomeningeal vein. In our cohort, Initial angiographic occlusion was achieved in 78.3% (18/23) with cure in 87% (20/23) of patients following surgery for non-sinus draining fistulas. Targeted interruption of the draining vein can achieve high cure rates, with reported series demonstrating obliteration rates approaching 95-100%. [26, 28-30]. The slightly lower rate observed in our series, may be attributed to anatomical complexity such as multiple draining veins, deep-seated fistulas, or proximity to eloquent cortical areas limiting surgical exposure and safe access [31, 32].
EVT has emerged as an effective treatment option in non-sinus draining lesions.Initial angiographic occlusion following EVT was achieved in 76.5% (26/34) with cure in 79.4% (27/34) of patients in our series. Obliteration rates for dural arteriovenous fistulas (dAVFs) have historically ranged from 50% to 80%, influenced by factors such as microcatheter navigability, distal positioning within the fistulous segment, and the number of arterial pedicles embolized [27]. Early endovascular efforts primarily used N-butyl cyanoacrylate (NBCA) glue, yielding occlusion in only 30-50% of cases [10, 33]. The subsequent development of DMSO-based liquid embolic agents substantially improved outcomes, allowing effective treatment through a single arterial pedicle in many instances and expanding the proportion of dAVFs amenable to a transarterial approach [13, 21]. These technical advancements, combined with ongoing refinements in microcatheter design, contributed to the higher success rates observed in contemporary series [34].
The management of dAVFs that drain into dural sinuses (Borden type I and II) is fundamentally distinct and requires careful consideration of both the abnormal arteriovenous shunt and the normal venous outflow of the surrounding brain. In such lesions, particularly in the absence of cortical venous reflux, the primary treatment objective is often symptomatic relief rather than complete angiographic cure [28, 29, 35, 36]. Surgical treatment in these fistulas is complex and less commonly employed due to its invasive nature and potential morbidity [32, 37]. In our series, only 3 patients with Borden Type II dAVFs underwent surgery, primarily to disconnect cortical venous reflux. While all 3 had residual fistulas, 1 patient experienced complete symptomatic relief and 2 reported partial improvement. EVT is the preferred first-line approach for dAVFs that drain into sinus [35, 38]. When the involved sinus is non-functional, TVE can achieve high cure rates (80-90%) [21, 35]. In our series, 2 patients with nonfunctional sinus underwent successful TVE, achieving angiographic cure with partial symptomatic improvement. When the sinus is functional, TVE is often contraindicated and TA embolization or sinus-preserving techniques, such as balloon-assisted protection are employed (Figure 3) [39-41]. Cure rates are lower (60-75%) depending on the arterial anatomy, and technique used [35]. Among 6 patients treated with sinus-preserving EVT in our series, angiographic cure was achieved in two (33.3%). Nonetheless, five (83.3%) experienced symptomatic improvement, emphasizing the role of partial flow modification in achieving clinical benefit even without complete angiographic cure.
While EVT is widely considered as the first-line treatment for most dAVFs, treatment strategies must be individualized, especially when arterial access is limited or hazardous, or when venous routes are unfavorable [9, 22, 27, 34, 42]. In our cohort, notably all anterior cranial fossa lesions were treated by microsurgery, whereas most parasagittal (85.7%), convexity (66.7%), and sigmoid sinus (75%) fistulas were treated endovascularly. Clinical urgency also influences treatment choice with patients presenting with hemorrhage or rapidly progressive deficits often requiring immediate intervention, favoring approaches that ensures prompt and definitive occlusion [3]. In some cases, failed EVT may compromise further endovascular access by occluding key arterial pedicles. In such instances, adjunctive microsurgery can achieve high cure rates [34, 43]. In our series, 7 patients with residual fistulas after initial EVT were successfully treated with surgery, highlighting the crucial complementary role of microsurgical intervention in complex or refractory cases (Figure 2).
The overall complication rate following endovascular treatment in our series was 21.4% (9/42 patients), compared to 14.8% (4/27 patients) in the surgical group. Reported endovascular complication rates range from 9% to 21%, often related to cranial nerve palsies or motor deficits resulting from embolic migration via external-to-internal carotid anastomoses, especially when skull base feeders are targeted [27, 35, 39]. Transvenous embolization may also cause venous infarction or cranial nerve dysfunction secondary to sinus thrombosis [44]. In our cohort, despite the 21.4% complication rate, only 1 patient had a permanent deficit, with 2 others experiencing transient symptoms, a similar trend as seen in other studies as well [35]. Surgical complication rates, range from 4% to 15% and are primarily related to craniotomy and manipulation near eloquent cortical or venous structures [25, 45]. Overall, both treatment modalities demonstrated acceptable safety profiles.
This study has several limitations. Its retrospective, single-center design introduces potential selection and information biases, and limits generalizability. Treatment allocation was non-randomized, based on clinical judgment, which may confound outcome comparisons. The small sample size, particularly within subgroups, restricted statistical power. Additionally, only treated patients were included, excluding data on conservatively managed low-grade fistulas. Finally, functional outcomes were based on clinical records, without standardized patient-reported outcome measures. However our results showing equivalence between EVT and surgical management of dAVF may facilitate more EVT approaches for these complex intracranial vascular malformation in the future.
5. Conclusion
There was no significant difference in the cure rate of dAVF and associated complications between EVT and surgical treatment. Angio-architectural complexity, fistula location and venous drainage patterns were significantly associated with higher treatment success in our series.
Author Contributions
SA collected the data and wrote the first draft of the manuscript. JJSS conceptualized the study, oversaw the conduct of the study and statistical analysis and finalized the final version of the manuscript, YL did the statistical analysis; NK, MA and JAS reviewed the final draft.
Funding
None.
Conflicts of Interest
JJSS-has received grant funding from Medtronic Canada for EMMA Can study. Other authors have no conflicts to disclose.
Abbreviations
CT: Computed Tomography
CTA: CT Angiography
DSA: Digital Subtraction Angiography
DMSO: Dimethyl Sulfoxide
dAVF: Dural arteriovenous Fistula
EVT: Endovascular Therapy
FLAIR: Fluid-Attenuated Inversion Recovery
MRI: Magnetic Resonance Imaging
MRA: MR Angiography
MRV: MR Venography
TA: Transarterial
TV: Transvenous
TVE: Transvenous Embolization
REFERENCES
[1] W Houser, H L Baker Jr, A
L Rhoton Jr, et al. “Intracranial dural arteriovenous malformations.” Radiology,
vol. 105, no. 1, pp. 55-64, 1972. View at: PubMed
[2] D Gandhi, J Chen, M Pearl,
et al. “Intracranial dural arteriovenous fistulas: classification, imaging
findings, and treatment.” AJNR Am J Neuroradiol, vol. 33, no. 6, pp.
1007-1013, 2012. View at: Publisher
Site | PubMed
[3] Wilhelm Sorteberg,
Angelika Sorteberg, Eva Astrid Jacobsen, et al. “Endovascular versus surgical
treatment of cranial dural arteriovenous fistulas: a single center 8-year
experience.” Acta Neurochir (Wien), vol. 164, no. 1, pp. 151-161, 2022.
View at: Publisher
Site | PubMed
[4] D L Barrow, R H Spector, I
F Braun, et al. “Classification and treatment of spontaneous carotid-cavernous
fistulas.” J Neurosurg, vol. 62, no. 2, pp. 248-256, 1985. View at: Publisher Site | PubMed
[5] R Djindjian, J J Merland,
A Rey, et al. “Super-Selective Arteriography of the External Carotid Artery.” Neurochirurgie,
pp. 165-171, 1978. View at: PubMed
[6] C Cognard, Y P Gobin, L
Pierot, et al. “Cerebral dural arteriovenous fistulas: clinical and
angiographic correlation with a revised classification of venous drainage.” Radiology,
vol. 194, no. 3, pp. 671-680, 1995. View at: Publisher Site | PubMed
[7] J A Borden, J K Wu, W A
Shucart “A proposed classification for spinal and cranial dural arteriovenous
fistulous malformations and implications for treatment.” J Neurosurg,
vol. 82, no. 2, pp. 166-179, 1995. View at: Publisher Site | PubMed
[8] Humain Baharvahdat, Yinn
Cher Ooi, Wi Jin Kim, et al. U”pdates in the management of cranial dural
arteriovenous fistula.” Stroke Vasc Neurol, vol. 5, no. 1, pp. 50-58,
2019. View at: Publisher
Site | PubMed
[9] Sun J Chung, Jong S Kim,
Jong C Kim, et al. “Intracranial dural arteriovenous fistulas: analysis of 60
patients.” Cerebrovasc Dis, vol. 13, no. 2, pp. 79-88, 2002. View at: Publisher Site | PubMed
[10] Dong Joon Kim, Robert A
Willinsky, Timo Krings, et al. “Intracranial dural arteriovenous shunts:
transarterial glue embolization—experience in 115 consecutive patients.” Radiology,
vol. 258, no. 2, pp. 554-561, 2011. View at: Publisher Site | PubMed
[11] Pierre Guedin, Stephan
Gaillard, Anne Boulin, et al. “Therapeutic management of intracranial dural
arteriovenous shunts with leptomeningeal venous drainage: report of 53
consecutive patients with emphasis on transarterial embolization with acrylic
glue.” J Neurosurg, vol. 112, no. 3, pp. 603-610, 2010. View at: Publisher Site | PubMed
[12] Adam Kobayashi, Rustam
Al-Shahi Salman “Prognosis and treatment of intracranial dural arteriovenous
fistulae: a systematic review and meta-analysis.” Int J Stroke, vol. 9,
no. 6, pp. 670-677, 2014. View at: Publisher
Site | PubMed
[13] Douglas M Choo, Jai Jai
Shiva Shankar “Onyx versus nBCA and coils in the treatment of intracranial
dural arteriovenous fistulas.” Interv Neuroradiol, vol. 22, no. 2, pp.
212-216, 2016. View at: Publisher
Site | PubMed
[14] Mark Nassar, Nazukjit Kaur
Nijjar, Zul Kaderali, et al. “Transvenous embolization of dural arteriovenous
fistulas with cortical venous drainage.” Can J Neurol Sci, vol. 50, no.
2, pp. 274-277, 2023. View at: Publisher
Site | PubMed
[15] Alex Botsford, Jai Jai
Shiva Shankar “Digital subtraction angiography-Dynavision in pretreatment
planning for embolization of dural arterio-venous fistulas.” J Neuroimaging,
vol. 28, no. 1, pp. 112-117, 2018. View at: Publisher Site | PubMed
[16] Alex Botsford, Jai Jai
Shiva Shankar “DSA-Dynavision in pretreatment planning for coil embolization of
indirect carotid-cavernous fistula.” J Neurointerv Surg, vol. 9, no. 4,
pp. 389-393, 2017. View at: Publisher Site | PubMed
[17] C P Lucas, J M Zabramski,
R F Spetzler, et al. “Treatment for intracranial dural arteriovenous
malformations: a meta-analysis from the English language literature.” Neurosurgery,
vol. 40, no. 6, pp. 1119-1130, 1997. View at: Publisher Site | PubMed
[18] Anna Piippo, Mika Niemelä,
Jouke van Popta, et al. “Characteristics and long-term outcome of 251 patients
with dural arteriovenous fistulas in a defined population.” J Neurosurg,
vol. 118, no. 5, pp. 923-934, 2013. View at: Publisher Site | PubMed
[19] F Signorelli, G M Della
Pepa, G Sabatino, et al. “Diagnosis and management of dural arteriovenous
fistulas: a 10-year single-center experience.” Clin Neurol Neurosurg,
vol. 128, pp. 123-129, 2015. View at: Publisher Site | PubMed
[20] Udaya K Kakarla, Vivek R
Deshmukh, Joseph M Zabramski, et al. “Surgical treatment of high-risk
intracranial dural arteriovenous fistula: clinical outcomes and avoidance of
complications.” Neurosurgery, vol. 61, no. 3, pp. 447-459, 2007. View
at: Publisher
Site | PubMed
[21] Bradley A Gross, Felipe C
Albuquerque, Karam Moon, et al. “Evolution of treatment and a detailed analysis
of occlusion, recurrence, and clinical outcomes in an endovascular library of
260 dural arteriovenous fistulas.” J Neurosurg, vol. 126, no. 6, pp.
1884-1893, 2017. View at: Publisher
Site | PubMed
[22] Young Sill Kang, Won-Sang
Cho, Sung Ho Lee, et al. “Role of surgery in management of intracranial dural
arteriovenous fistulas.” J Cerebrovasc Endovasc Neurosurg, vol. 25, no.
2, pp. 117-131, 2023. View at: Publisher Site | PubMed
[23] G Bavinzski, B Richling, M
Killer, et al. “Evolution of different therapeutic strategies in the treatment
of cranial dural arteriovenous fistulas—report of 30 cases.” Acta Neurochir
(Wien), vol. 138, no. 2, pp. 132-138, 1996. View at: Publisher Site | PubMed
[24] P Lasjaunias, M Chiu, K
ter Brugge, et al. “Neurological manifestations of intracranial dural
arteriovenous malformations.” J Neurosurg, vol. 64, no. 5, pp. 724-730,
1986. View at: Publisher
Site | PubMed
[25] Michael F Stiefel, Felipe
C Albuquerque, Min S Park, et al. “Endovascular treatment of intracranial dural
arteriovenous fistulae using Onyx: a case series.” Oper Neurosurg, vol.
65, no. 6, pp. ons132-ons140, 2009. View at: Publisher
Site | PubMed
[26] Sabareesh K Natarajan,
Basavaraj Ghodke, Louis J Kim, et al. “Multimodality treatment of intracranial
dural arteriovenous fistulas in the Onyx era: a single center experience.” World
Neurosurg, vol. 73, no. 4, pp. 365-379, 2010. View at: Publisher Site | PubMed
[27] C Cognard, A C Januel, N A
Silva Jr, et al. “Endovascular treatment of intracranial dural arteriovenous
fistulas with cortical venous drainage: new management using Onyx.” AJNR Am
J Neuroradiol, vol. 29, no. 2, pp. 235-241, 2008. View at: Publisher Site | PubMed
[28] Michael Söderman, Ladislav
Pavic, Göran Edner, et al. “Natural history of dural arteriovenous shunts.” Stroke,
vol. 39, no. 6, pp. 1735-1739, 2008. View at: Publisher Site | PubMed
[29] Gerasimos Baltsavias,
Anton Valavanis, Luca Regli, et al. “Cranial dural arteriovenous shunts:
selection of the ideal lesion for surgical occlusion according to the
classification system.” Acta Neurochir (Wien), vol. 161, no. 9, pp.
1775-1781, 2019. View at: Publisher
Site | PubMed
[30] M Collice, G D'Aliberti, G
Talamonti, et al. “Surgical interruption of leptomeningeal drainage as
treatment for intracranial dural arteriovenous fistulas without dural sinus
drainage.” J Neurosurg, vol. 84, no. 5, pp. 810-817, 1996. View at: Publisher Site | PubMed
[31] Bradley A Gross, Rose Du
“Surgical treatment of high-grade dural arteriovenous fistulae.” J Clin
Neurosci, vol. 20, no. 11, pp. 1527-1532, 2013. View at: Publisher Site | PubMed
[32] Ramsey Ashour R, Morcos JJ
“Surgical management of cerebral dural arteriovenous fistulae. In: Spetzler RF,
Kondziolka D, Higashida RT, Kalani MYS, eds. Comprehensive Management of
Arteriovenous Malformations of the Brain and Spine.” Cambridge University
Press, pp. 144-171, 2015.
[33] C W Kerber, L D Cromwell,
P E Sheptak “Intraarterial cyanoacrylate: an adjunct in the treatment of
spinal/paraspinal arteriovenous malformations.” AJR Am J Roentgenol,
vol. 130, no. 1, pp. 99-103, 1978. View at: Publisher Site | PubMed
[34] Yosuke Akamatsu, Santiago
Gomez-Paz, David Vergara-Garcia, et al. “Role of surgical intervention for
intracranial dural arteriovenous fistulas with cortical venous drainage in an
endovascular era: a case series.” Oper Neurosurg, vol. 20, no. 4, pp.
364-372, 2021. View at: Publisher
Site | PubMed
[35] Tobias Rossmann, Michael
Veldeman, Elias Oulasvirta, et al. “Long-term treatment outcomes and natural
course of low-grade intracranial dural arteriovenous fistulas.” Neurosurg
Focus, vol. 56, no. 3, pp. E2, 2024. View at: Publisher Site | PubMed
[36] Bradley A Gross, Rose Du
“The natural history of cerebral dural arteriovenous fistulae.” Neurosurgery,
vol. 71, no. 3, pp. 602-603, 2012. View at: Publisher Site | PubMed
[37] T M Sundt Jr, D G Piepgras
“The surgical approach to arteriovenous malformations of the lateral and
sigmoid dural sinuses.” J Neurosurg, vol. 59, no. 1, pp. 32-39, 1983.
View at: Publisher
Site | PubMed
[38] Pearse M. Cranial dural
vascular malformations. In: Practical Neuroangiography. Lippincott Williams
& Wilkins; pp. 322-350, 2013.
[39] D F Vollherbst, C Ulfert
1, U Neuberger, et al. “Endovascular treatment of dural arteriovenous fistulas
using transarterial liquid embolization in combination with transvenous
balloon-assisted protection of the venous sinus.” AJNR Am J Neuroradiol,
vol. 39, no. 7, pp. 1296-1302, 2018. View at: Publisher Site | PubMed
[40] J O Zamponi Jr, F P
Trivelato 1, M T S Rezende, et al. “Transarterial treatment of cranial dural
arteriovenous fistulas: the role of transarterial and transvenous
balloon-assisted embolization.” AJNR Am J Neuroradiol, vol. 41, no. 11,
pp. 2100-2106, 2020. View at: Publisher
Site | PubMed
[41] Hajime Yabuzaki, Tomoaki
Terada, Tomoyuki Tsumoto, et al. “Transarterial embolization in dural
arteriovenous fistulas under sinus balloon protection using the SHOURYU
supercompliant balloon.” Interdiscip Neurosurg, vol. 27, pp. 101412,
2022. View at: Publisher
Site
[42] R Webster Crowley, Avery J
Evans, Mary E Jensen, et al. “Combined surgical/endovascular treatment of a
complex dural arteriovenous fistula in a 21-month-old: technical note.” J
Neurosurg Pediatr, vol. 3, no. 6, pp. 501-506, 2009. View at: Publisher Site | PubMed
[43] Dimitri Benner, Christoph
J Griessenauer “Commentary: role of surgical intervention for intracranial
dural arteriovenous fistulae with cortical drainage in an endovascular era: a
case series.” Oper Neurosurg, vol. 20, no. 5, pp. E391-E392, 2021. View
at: Publisher Site | PubMed
[44] M Kirsch, T Liebig, D
Kühne, et al. “Endovascular management of dural arteriovenous fistulas of the
transverse and sigmoid sinus in 150 patients.” Neuroradiology, vol. 51,
no. 7, pp. 477-483, 2009. View at: Publisher Site | PubMed
[45] Michael T Lawton, Rene O Sanchez-Mejia, Diep Pham, et al. “Tentorial dural arteriovenous fistulae: operative strategies and microsurgical results for six types.” Neurosurgery, vol. 62, no. 3, pp. 110-125, 2008. View at: Publisher Site | PubMed
