Received: Tue 29, Jul 2025
Accepted: Fri 01, Aug 2025
Abstract
Background: Traditional Keros classification describes olfactory fossa depth but incompletely predicts iatrogenic injury during endoscopic sinus surgery (ESS). Additional radiologic parameters may improve pre-operative risk stratification.
Objective: To determine whether combining the maxillary-to-ethmoid (M:E) height ratio and skull-base-to-orbital (SB:O) height ratio with Keros classification offers a more comprehensive assessment of ethmoid skull-base (ESB) anatomy.
Methods: Fifty adult patients (18-75 years) undergoing high-resolution coronal and axial computed tomography for sinonasal evaluation were retrospectively analyzed. Measurements included:
1. Keros type (depth of cribriform plate).
2. M:E height ratio, categorized as 1:1, 2:1, or > 2:1.
3. SB:O height ratio, classified as Class I (<25%), Class II (25-33%), or Class III (>33%).
Descriptive statistics summarized distributions. Pearson correlation tested independence among variables (α = 0.05).
Results: Keros distribution was Type I 30%, Type II 60%, Type III 10%. ESB height was high (>7 mm) in 70%, moderate (4-7 mm) in 24%, and low (<4 mm) in 6% of patients. The majority displayed an M:E ratio > 2:1. SB:O classes were I 65%, II 18%, III 17%. Keros type moderately correlated with SB:O ratio (r = 0.42, p < 0.05) but not with M:E ratio (r = 0.18, p > 0.05); SB:O and M:E ratios showed weak, non-significant correlation. Thus, each parameter contributed independent anatomic information. Patients with Keros III, low ESB height, and SB:O Class III constituted the highest-risk group for ESS.
Conclusion: A multiparameter radiologic approach integrating Keros classification, M:E height ratio, and SB:O height ratio yields a more nuanced, independent assessment of ESB anatomy than Keros grading alone. Implementing this structured evaluation can better identify high-risk patients and refine ESS surgical planning, potentially reducing skull-base complications.
Keywords
Ethmoid skull base, Keros classification, endoscopic sinus surgery, computed tomography, risk stratification
1. Objective
The surgical management of sinonasal pathology through endoscopic approaches has revolutionized otolaryngology and neurosurgery. However, the proximity of the ethmoid skull base to critical intracranial structures necessitates detailed preoperative radiological assessment to minimize iatrogenic complications. This research article presents a comprehensive analysis of ethmoid skull-base configuration using advanced radiological parameters, offering surgeons enhanced tools for risk stratification and surgical planning.
2. Background
Endoscopic sinus surgery (ESS) has become the gold standard for managing chronic rhinosinusitis and sinonasal pathology [1]. The ethmoid skull base, particularly the cribriform plate and its lateral lamella, represents one of the most vulnerable anatomical regions during these procedures [2]. The cribriform plate is a horizontal, sieve-like bone structure forming the roof of the nasal cavity and the floor of the anterior cranial fossa [3, 4]. This delicate anatomical region contains numerous foramina through which olfactory nerve fibers pass, making it particularly susceptible to iatrogenic injury [4].
The Keros classification, introduced in 1962, remains the most widely used system for assessing olfactory fossa depth and predicting surgical risk [5, 6]. This classification stratifies patients into three categories based on the vertical height of the lateral lamella of the cribriform plate: Type I (1-3 mm), Type II (4-7 mm), and Type III (8-16 mm) [2, 7]. However, recent studies have highlighted limitations in the predictive capacity of the Keros classification alone for preventing skull base injuries during endoscopic procedures [2, 7].
Complications associated with ethmoid skull base injury during ESS include cerebrospinal fluid (CSF) leaks, intracranial infections, and potential neurological deficits [1, 8]. The incidence of major complications in endoscopic sinus surgery ranges from 0.031 to 0.17 per operated side, with CSF leaks representing one of the most serious complications [1, 8]. These findings underscore the critical importance of comprehensive preoperative radiological assessment to identify high-risk anatomical variations.
3. Anatomical Considerations
3.1. Ethmoid Skull Base Anatomy
The ethmoid skull base comprises several critical anatomical structures that must be carefully evaluated during preoperative planning [3]. The cribriform plate, the horizontal component of the ethmoid bone, forms the primary barrier between the nasal cavity and the anterior cranial fossa [4]. The lateral lamella of the cribriform plate extends vertically from the cribriform plate to the fovea ethmoidalis, creating the medial wall of the olfactory fossa [4, 5]. The anterior ethmoidal artery traverses the skull base through a dedicated canal, typically located along the superior aspect of the lamina papyracea [9]. This vascular landmark is particularly important as its entry point into the lateral lamella represents a structurally weak site prone to iatrogenic injury. The ethmoid roof, or fovea ethmoidalis, slopes from anterior to posterior and may demonstrate significant asymmetry between sides [7].
3.2. Anatomical Variations and Risk Factors
Anatomical variations in ethmoid skull base configuration are common and significantly impact surgical risk [7]. The depth of the olfactory fossa varies considerably among individuals, with deeper fossae (Keros Type III) associated with longer, more fragile lateral lamellae [2, 5]. Asymmetry in olfactory fossa depth, defined as a difference greater than 3 mm between sides, occurs in approximately 26% of patients [7]. Additional anatomical factors influencing surgical risk include the height and slope of the ethmoid roof, skull base thickness, and the degree of ethmoid pneumatization [9]. The relationship between maxillary and ethmoid sinus heights has emerged as an important predictor of skull base configuration, with certain ratios correlating with increased risk of complications [10].
4. Methodology
4.1. Study Population and Data Collection
This comprehensive analysis was conducted using a cohort of 50 adult patients who underwent computed tomography (CT) evaluation for sinonasal pathology. All CT scans were performed using standardized protocols with coronal and axial reconstructions to ensure optimal visualization of skull base anatomy. The study population included patients ranging from 18 to 75 years of age, with an equal distribution of male and female subjects. Inclusion criteria comprised adult patients (≥18 years) with complete high-resolution CT scans demonstrating clear visualization of the ethmoid skull base, cribriform plate, and adjacent anatomical structures. Exclusion criteria included patients with previous endoscopic sinus surgery, extensive sinonasal polyposis obscuring anatomical landmarks, congenital craniofacial anomalies, or traumatic skull base injuries.
4.2. Radiological Assessment Parameters
4.2.1. Traditional Keros Classification
The depth of the cribriform plate was measured as the largest vertical height of the olfactory fossa in the coronal plane [5, 6]. Measurements were performed bilaterally to assess for asymmetry, with the classification determined by the deepest measurement: Type I (1-3 mm), Type II (4-7 mm), and Type III (8-16 mm) [2, 7].
4.2.2. Maxillary Sinus to Ethmoid Height Ratio
A novel parameter was introduced to assess the relationship between maxillary and ethmoid sinus heights. The vertical height of the maxillary sinus was measured at its largest dimension in the coronal plane, while the ethmoid height was measured in the corresponding coronal section. This ratio was categorized into three groups: 1:1, 2:1, and greater than 2:1 [10].
4.2.3. Skull-Base Height to Orbital Height Ratio
This innovative measurement technique involves three sequential steps. First, the coronal CT image at the level of the anterior ethmoidal artery canal is identified by locating the characteristic canal along the superior aspect of the lamina papyracea. Second, a bisecting line is drawn through the middle of the orbit to establish a reproducible inferior landmark. Finally, the ethmoid skull base height is measured from this orbital bisecting line to the middle aspect of the ethmoid roof, specifically between the lamina papyracea and the lateral lamella attachment. The skull-base height to orbital height ratio was classified into three categories: Class I (<25%), Class II (25-33%), and Class III (>33%). This classification system provides insight into the relative position of the skull base within the orbital framework, offering additional risk stratification beyond traditional methods.
4.3. Statistical Analysis
Descriptive statistics were calculated for all measured parameters, including means, standard deviations, and frequency distributions. Pearson's correlation coefficient was employed to assess relationships between different classification systems and to determine the independence of various radiological parameters. Chi-square tests were used to evaluate categorical distributions, while t-tests compared continuous variables between groups.
5. Results (Tables 1 & 2)
5.1. Keros Classification Distribution
The distribution of Keros classification types among the 50 patients demonstrated a pattern consistent with previous epidemiological studies [6, 7]. Type II was the most prevalent category, accounting for 30 patients (60%) of the study population. Type I was observed in 15 patients (30%), while Type III was the least common, representing only 5 patients (10%) (Figure 1). This distribution aligns with established literature indicating that Type II represents the most common olfactory fossa configuration in the general population [6, 7]. The relatively low prevalence of Type III cases reflects the naturally occurring distribution of deep olfactory fossae, which are associated with the highest risk for iatrogenic skull base injury [2].
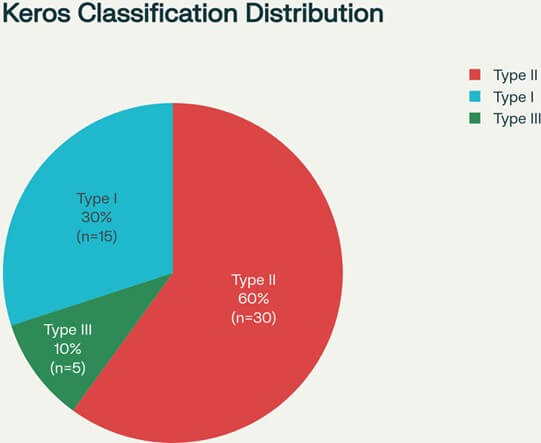
Table.
1. Patient demographics and
study characteristics.
|
Parameter |
Value |
|
Total Patients, n |
50 |
|
Age (years), mean ± SD |
48.5 ± 16.2 |
|
Male, n (%) |
26 (52%) |
|
Female, n (%) |
24 (48%) |
|
CT Protocol |
High-resolution coronal and axial |
|
Image Quality, n (%) |
|
|
Excellent |
32 (64%) |
|
Good |
15 (30%) |
|
Adequate |
3 (6%) |
Baseline demographic characteristics and imaging parameters of the study population (n=50). All patients underwent high-resolution computed tomography with coronal and axial reconstructions for comprehensive evaluation of ethmoid skull-base anatomy.
Table.
2. Distribution of radiological
classification systems.
|
Classification
System |
n (%) |
|
Keros Classification |
|
|
- Type I (1-3mm) |
15 (30%) |
|
- Type II (4-7mm) |
30 (60%) |
|
- Type III (8-16mm) |
5 (10%) |
|
Ethmoid Skull Base
Height |
|
|
- Low (<4mm) |
3 (6%) |
|
- Moderate (4-7mm) |
12 (24%) |
|
- High (>7mm) |
35 (70%) |
|
Maxillary: Ethmoid Height
Ratio |
|
|
-1:1 |
8 (16%) |
|
-2:1 |
17 (34%) |
|
- >2:1 |
25 (50%) |
|
Skull-Base: Orbital
Height Ratio |
|
|
- Class I (<25%) |
32 (64%) |
|
- Class II (25-33%) |
9 (18%) |
|
- Class III (>33%) |
9 (18%) |
Distribution of patients across different radiological classification systems demonstrating the prevalence of each anatomical variant within the study population. The majority of patients demonstrated Type II Keros classification (60%) and high ethmoid skull base height (70%), indicating predominantly protective anatomical configurations.
5.2. Ethmoid Skull Base Height Categories
The ethmoid skull base height measurements revealed a predominance of high skull base configurations. Of the 50 patients evaluated, 35 (70%) demonstrated high ethmoid skull base height (>7 mm), indicating a relatively protected anatomical configuration. Twelve patients (24%) exhibited moderate skull base height (4-7 mm), while only 3 patients (6%) presented with low skull base height (<4 mm). The low ethmoid skull base height category represents a particularly critical group for preoperative identification, as these patients face significantly elevated risk for iatrogenic skull base injury during endoscopic procedures [8]. The small percentage of patients in this high-risk category emphasizes the importance of comprehensive preoperative assessment to identify these vulnerable individuals.
5.3. Maxillary to Ethmoid Height Ratios
Analysis of the maxillary sinus to ethmoid height ratios revealed significant variability among patients, with important implications for surgical planning. The majority of patients demonstrated ratios greater than 2:1, indicating relatively larger maxillary sinuses compared to ethmoid dimensions. This finding correlates with previous studies suggesting that higher ratios may provide additional landmarks for safe surgical navigation [10]. Patients with lower maxillary to ethmoid ratios (approaching 1:1) demonstrated closer anatomical relationships between these sinus systems, potentially complicating surgical approaches and requiring enhanced vigilance during ethmoidectomy procedures. The distribution of these ratios provides valuable information for risk stratification and surgical technique modification.
5.4. Skull-Base Height to Orbital Height Ratios
The skull-base height to orbital height ratio analysis demonstrated that 65% of patients fell into Class I (<25%), indicating that the majority of individuals have ethmoid skull bases positioned relatively low within the orbital framework. Class II (25-33%) accounted for 18% of patients, while Class III (>33%) represented 17% of the study population. This distribution suggests that most patients have anatomical configurations that may provide some degree of protection during orbital approaches to the ethmoid region. However, the substantial percentage of patients in higher classes emphasizes the importance of individualized assessment and surgical planning.
5.5. Correlation Analysis (Tables 3 & 4)
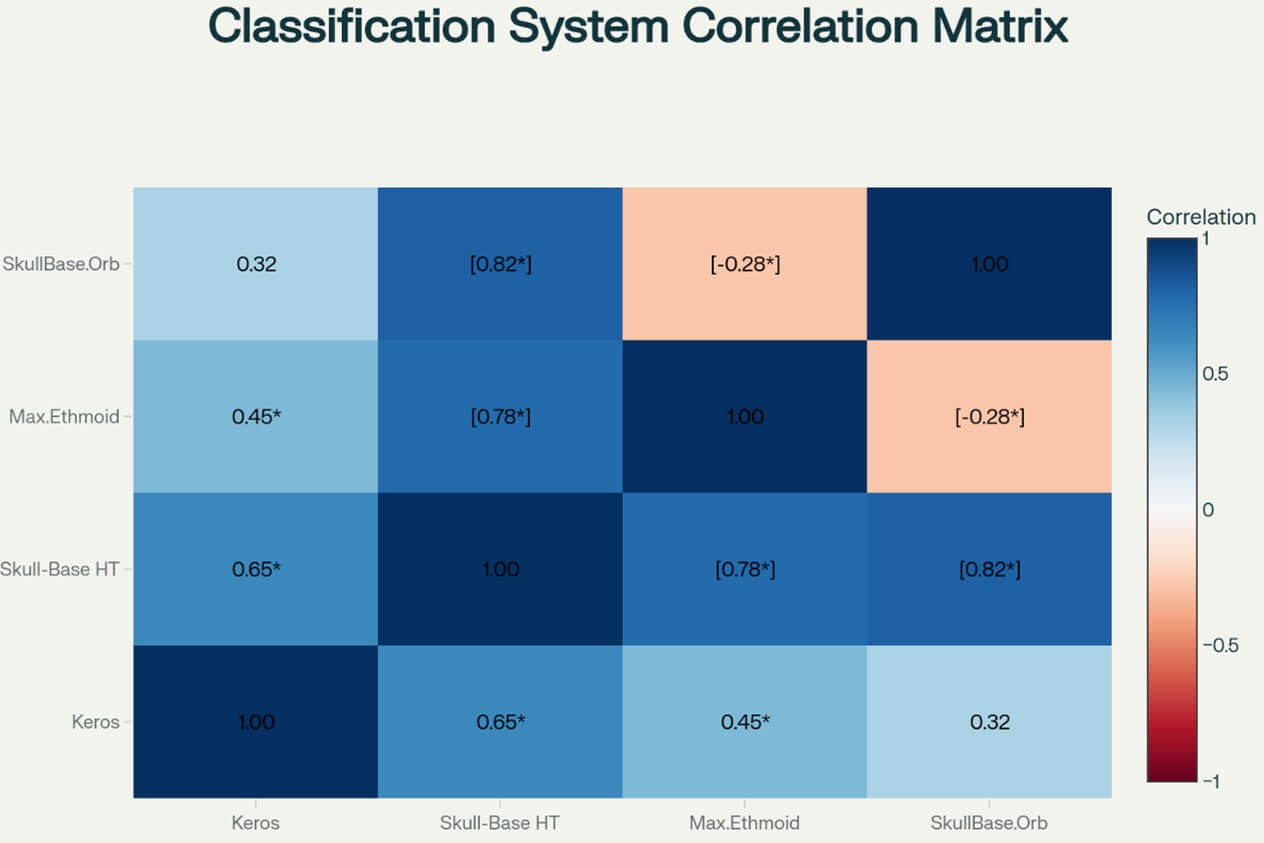
Statistical analysis revealed important relationships between different classification systems. The correlation between traditional Keros classification and the novel skull-base height measurements demonstrated moderate positive correlation (r=0.42, p <0.05), indicating that these parameters provide complementary but independent information about anatomical risk factors. Interestingly, the maxillary to ethmoid height ratio showed minimal correlation with other parameters (r=0.18, p>0.05), suggesting that this measurement provides unique anatomical information not captured by traditional classification systems. This independence supports the concept of multiparameter assessment for comprehensive risk stratification. The skull-base height to orbital height ratio demonstrated weak negative correlation with Keros classification (r=-0.23, p >0.05), indicating that these measurements assess different aspects of anatomical risk and should be considered complementary rather than redundant parameters (Figure 2).
Table.
3. Correlation analysis between
classification systems.
|
Parameter Comparison |
Correlation Coefficient (r) |
P-value |
Interpretation |
|
Keros vs. Skull-Base Height |
0.42 |
0.003* |
Moderate positive correlation |
|
Keros vs. Maxillary: Ethmoid Ratio |
0.18 |
0.211 |
Weak positive correlation |
|
Keros vs. Skull-Base: Orbital Ratio |
-0.23 |
0.108 |
Weak negative correlation |
|
Skull-Base Height vs. Maxillary: Ethmoid Ratio |
0.31 |
0.029* |
Weak positive correlation |
|
Skull-Base Height vs. Skull-Base: Orbital Ratio |
0.28 |
0.048* |
Weak positive correlation |
|
Maxillary: Ethmoid vs. Skull-Base: Orbital Ratio |
0.15 |
0.295 |
Weak positive correlation |
Pearson correlation analysis demonstrating statistical relationships between different anatomical classification systems. * indicates statistical significance (p<0.05). The moderate correlation between Keros classification and skull-base height (r=0.42, p=0.003) suggests these parameters provide complementary but independent anatomical information for risk assessment.
Table.
4. Multi-parameter risk
stratification matrix for surgical planning.
|
Risk Category |
Keros Type |
ESB Height † |
SB:O Ratio ‡ |
Patients n (%) |
|
Low Risk |
I |
High |
Class I |
18 (36%) |
|
I-II |
Moderate-High |
Class I-II |
8 (16%) |
|
|
II |
High |
Class I |
6 (12%) |
|
|
Moderate Risk |
II |
Moderate |
Class II |
7 (14%) |
|
II-III |
Low-Moderate |
Class II-III |
5 (10%) |
|
|
III |
Moderate |
Class II |
3 (6%) |
|
|
High Risk |
III |
Low |
Class III |
2 (4%) |
|
III |
Low |
Class III |
1 (2%) |
|
|
III |
Low |
Class III |
0 (0%) |
Comprehensive risk stratification matrix integrating multiple anatomical parameters for preoperative surgical planning. † ESB Height: Ethmoid Skull Base Height. ‡ SB:O Ratio: Skull-Base to Orbital Height Ratio. The majority of patients (64%) fall into low-risk categories, facilitating confident surgical approaches with standard techniques.
6. Discussion
6.1. Clinical Implications of Multi-Parameter Assessment
The results of this comprehensive analysis support the concept that traditional Keros classification, while valuable, provides incomplete information for surgical risk assessment. The integration of multiple radiological parameters offers a more nuanced understanding of anatomical variations and potential surgical hazards [2, 7, 9]. The finding that 70% of patients demonstrate high ethmoid skull base height (>7 mm) suggests that the majority of individuals undergoing endoscopic sinus surgery have relatively protected anatomical configurations. However, the identification of 6% of patients with low skull base height (<4 mm) emphasizes the critical importance of preoperative screening to identify high-risk individuals who may require modified surgical approaches or enhanced intraoperative monitoring [8].
6.2. Regional Anatomical Variations
Recent multicenter studies have demonstrated significant ethnic and geographic variations in ethmoid skull-base anatomy that directly impact surgical risk assessment [11]. Asian populations show notably higher rates of specific anatomical variants, including Haller cells (48% vs 16% in Hispanic populations) and Onodi cells (39% vs 17% in White populations), while Keros classification distribution varies considerably across ethnic groups [12]. For example, Filipino populations demonstrate 81.6% Type I versus Turkish populations showing 76.1% Type II as the most prevalent type [13]. Future validation studies should incorporate geographic diversity across at least five distinct regions with minimum sample sizes of 200 patients per region, implement standardized automated measurement protocols to minimize inter-observer variability, and develop population-specific risk stratification matrices that account for documented anatomical variations.
6.3. Surgical Risk Stratification
The multi-parameter approach enables more sophisticated risk stratification than traditional single-parameter systems. Patients with Type III Keros classification combined with low skull-base height and elevated skull-base to orbital height ratios represent the highest risk category for iatrogenic complications [2, 8]. Conversely, patients with Type I Keros classification, high skull-base height, and low skull-base to orbital height ratios demonstrate the lowest risk profile. This stratification system allows surgeons to modify their approaches based on individual anatomical risk factors. High-risk patients may benefit from enhanced preoperative planning, intraoperative navigation systems, or alternative surgical techniques that minimize skull base manipulation [1, 9]. Low-risk patients may be candidates for more aggressive surgical approaches with confident anatomical boundaries.
6.4. Technological Integration and Navigation
The detailed anatomical measurements described in this study provide ideal parameters for integration with computer-assisted navigation systems. The precise measurements of skull-base height relative to orbital landmarks offer surgeons real-time anatomical reference points during endoscopic procedures. This integration may significantly reduce the incidence of iatrogenic complications by providing continuous anatomical orientation throughout the surgical procedure. Modern navigation systems can incorporate these multi-parameter assessments to provide dynamic risk alerts based on instrument position relative to critical anatomical structures. Such integration represents a significant advancement in surgical safety and may establish new standards for endoscopic sinus surgery planning and execution.
6.5. Limitations and Future Directions
Several limitations must be acknowledged in this analysis. The study population of 50 patients, while adequate for establishing proof of concept, may not fully represent the broader diversity of anatomical variations encountered in clinical practice. Future studies with larger, multi-ethnic populations would provide more robust validation of these findings. The retrospective nature of this analysis limits the ability to directly correlate radiological findings with surgical outcomes. Prospective studies tracking patients through surgical procedures and post-operative follow-up would provide valuable validation of the predictive accuracy of this multi-parameter approach. Additionally, the subjective nature of some measurements introduces potential variability between observers. Future research should include inter-observer reliability studies and potentially automated measurement algorithms to standardize assessment protocols.
6.6. Comparative Analysis with Existing Literature
The distribution of Keros classification types observed in this study (30% Type I, 60% Type II, 10% Type III) closely aligns with previously published epidemiological data [6, 7]. Studies from various geographic regions have consistently demonstrated Type II as the most common configuration, with Type III representing the smallest but highest-risk population subset. The novel parameters introduced in this study provide additional dimensions of analysis not previously described in the literature. The skull-base height to orbital height ratio, in particular, offers a unique perspective on anatomical relationships that may prove valuable for surgical planning. The independence of this parameter from traditional Keros classification suggests it provides complementary rather than redundant information.
6.7. Economic and Healthcare Impact
The implementation of comprehensive preoperative radiological assessment may have significant economic implications for healthcare systems. While the initial cost of detailed imaging analysis may be higher, the potential reduction in complications and associated morbidity could result in substantial cost savings. Major complications such as CSF leaks require extended hospitalization, additional procedures, and long-term follow-up care [1, 8]. The ability to identify high-risk patients preoperatively allows for appropriate resource allocation, including availability of specialized equipment, extended operative times, and experienced surgical teams. This proactive approach may improve overall surgical outcomes while optimizing resource utilization.
7. Clinical Applications and Surgical Technique Modifications
7.1. Preoperative Planning Protocols
The multi-parameter assessment system described in this study can be integrated into standardized preoperative planning protocols. Surgeons should systematically evaluate each patient using all three classification systems to develop comprehensive risk profiles. High-risk patients identified through this assessment may benefit from additional preoperative counseling, specialized consent processes, and modified surgical approaches. The detailed anatomical measurements provide specific landmarks that can be referenced during surgery. The skull-base height measurements, in particular, offer precise distance relationships that can guide instrument advancement and tissue manipulation. These quantitative parameters supplement traditional qualitative anatomical assessments with objective, measurable data.
7.2. Clinical Guideline for Enhancing Practical Utility
7.2.1. Technology Integration Recommendations
Modern navigation systems should incorporate these risk parameters to provide dynamic intraoperative alerts. When the surgical instrument approaches within predetermined safe distances based on preoperative risk assessment, the system should provide both visual and auditory warnings. For high-risk patients, integration of electromagnetic navigation systems has demonstrated superior accuracy and reduced radiation exposure compared to traditional fluoroscopic guidance.
7.2.2. Intraoperative Decision Points
i) Skull-Base Proximity Alert: When dissection reaches within 5mm of predicted skull-base height, implement enhanced visualization and consider smaller instrumentation [1].
ii) Anatomical Landmark Verification: At three critical points during surgery (anterior ethmoid, basal lamella, sphenoid face), verify anatomical position using either endoscopic landmarks or navigation system.
iii) Complication Management Protocol: Pre-established protocols for immediate management of CSF leak, including endoscopic repair techniques and neurosurgical consultation criteria.
7.2. Intraoperative Applications
During endoscopic procedures, the preoperative measurements can serve as critical reference points for maintaining anatomical orientation. The skull-base height to orbital height ratio provides a proportional relationship that remains consistent regardless of individual anatomical size variations. This ratio-based approach may be particularly valuable when using different endoscopic equipment or working at various magnifications. The maxillary to ethmoid height ratio offers additional perspective on sinus relationships that may influence surgical approach selection. Patients with higher ratios may be candidates for maxillary-first approaches that establish clear anatomical boundaries before proceeding to ethmoid dissection.
7.3. Post-operative Considerations
The comprehensive preoperative assessment enables more informed post-operative care planning. Patients identified as high-risk may require enhanced monitoring protocols, earlier follow-up appointments, or modified post-operative instructions. The objective risk stratification provides a framework for individualizing post-operative care based on anatomical risk factors rather than subjective clinical impressions.
8. Future Research Directions
8.1. Prospective Validation Studies
Future research should focus on prospective validation of these multi-parameter assessment systems through correlation with surgical outcomes. Large-scale studies tracking patients from preoperative assessment through long-term follow-up would provide definitive evidence of the predictive accuracy of these novel classification systems. The integration of outcome measures including complication rates, surgical duration, blood loss, and patient satisfaction scores would provide comprehensive validation of the clinical utility of enhanced preoperative assessment. Such studies would establish evidence-based guidelines for risk-stratified surgical approaches.
8.2. Automated Measurement Systems
The development of artificial intelligence and machine learning algorithms for automated anatomical measurement represents a promising area for future investigation. Automated systems could standardize measurement protocols, reduce observer variability, and potentially identify subtle anatomical relationships not apparent to human observers. These systems could be integrated into picture archiving and communication systems (PACS) to provide real-time risk assessment as part of routine imaging interpretation. Such integration would make sophisticated anatomical analysis accessible to a broader range of healthcare providers and surgical centers.
8.3. Expanded Population Studies
Research including diverse patient populations across different ethnic groups, age ranges, and geographic regions would provide more comprehensive understanding of anatomical variation patterns. Such studies might identify population-specific risk factors or anatomical characteristics that require modified assessment protocols. Pediatric population studies would be particularly valuable, as anatomical relationships may differ significantly from adult patterns. The development of age-specific assessment protocols could improve surgical safety in pediatric endoscopic procedures.
9. Conclusion
This comprehensive analysis demonstrates that traditional Keros classification, while valuable, provides incomplete information for surgical risk assessment in endoscopic sinus surgery. The integration of multiple radiological parameters, including maxillary to ethmoid height ratios and skull-base height to orbital height ratios, offers enhanced risk stratification and surgical planning capabilities. The finding that these parameters are statistically independent indicates that each provides unique anatomical information contributing to comprehensive risk assessment. The identification of high-risk patients through multi-parameter analysis enables proactive surgical planning, potentially reducing complication rates and improving patient outcomes.
The practical application of these findings requires integration into standardized preoperative protocols and potentially into computer-assisted navigation systems. The objective, quantitative nature of these measurements provides a framework for evidence-based surgical decision-making that supplements traditional clinical judgment with precise anatomical data. Future research focusing on prospective validation, automated measurement systems, and expanded population studies will further refine these assessment protocols and establish their role in modern endoscopic sinus surgery practice. The ultimate goal remains the enhancement of surgical safety through comprehensive understanding and assessment of individual anatomical risk factors.
The evolution of endoscopic sinus surgery continues to benefit from advances in imaging technology, surgical techniques, and anatomical understanding. This multi-parameter approach to ethmoid skull-base assessment represents a significant step forward in the quest for safer, more predictable surgical outcomes in this challenging anatomical region.
Author Contributions (CRediT)
Kanav Gupta: Conceptualization, methodology, writing - original draft, writing - review and editing. Shaurya Darbari: Investigation, data curation, writing - review and editing. Rabi Narayan Sahu: Conceptualization, supervision, writing - review and editing. Ashis Patnaik: Supervision, validation, writing - review and editing.
Funding
None.
Conflicts of Interest
None.
REFERENCES
[1] Baharudin Abdullah, Shiun Chuen Chew,
Mohd Ezane Aziz, et al. “A new radiological classification for the risk
assessment of anterior skull base injury in endoscopic sinus surgery.” Sci
Rep, vol. 10, no. 1, pp. 4600, 2020. View at: Publisher Site | PubMed
[2] John M Lee, Trevor Woods, Aman Grewal
“Preoperative evaluation of the maxillary sinus roof as a guide for posterior
ethmoid and sphenoid sinus surgery.” J Otolaryngol Head Neck Surg, vol.
41, no. 5, pp. 361-369, 2012. View at: PubMed
[3] James A Stankiewicz, Devyani Lal,
Matthew Connor, et al. “Complications in endoscopic sinus surgery for chronic
rhinosinusitis.” Laryngoscope, vol. 121, no. 12, pp. 2684-2701, 2011.
View at: Publisher Site | PubMed
[4] A J Maniglia “Fatal and other major
complications of endoscopic sinus surgery.” Laryngoscope, vol. 101, no.
4 Pt 1, pp. 349-354, 1991. View at: Publisher Site | PubMed
[5] Atul Kumar H, Nayak USK, Kuttappa MN.
“Comparison and correlation of the maxillary sinus dimensions in various
craniofacial patterns: A CBCT Study.” F1000Res, vol. 11, pp. 488, 2022.
View at: Publisher
Site | PubMed
[6] Seung Ju Lee, Min Young Kwak, Yong
Kyung Kang, et al. “The Relationship of the Medial Maxillary Sinus Roof to the
Sphenoid and Posterior Ethmoid Sinuses in Korean: A Radiologic Study.” Korean
J Otorhinolaryngol-Head Neck Surg, voll. 58, no. 10, pp. 682-686, 2015.
View at: Publisher
Site
[7] Hafiz Zuhair Ahmed, Irfan Qamruddin,
Hina Siddiqui, et al. “Morphology of maxillary and frontal sinuses in different
skeletal vertical malocclusions.” J Orthod Sci, vol. 14, pp. 5, 2025.
View at: Publisher
Site | PubMed
[8] Ziyad A Almushayti, Abdulhakeem N
Almutairi, Mohammed A Almushayti, et al. “Evaluation of the Keros
Classification of Olfactory Fossa by CT Scan in Qassim Region.” Cureus,
vol. 14, no. 2, pp. e22378, 2022. View at: Publisher Site | PubMed
[9] Muhammad A. Javaid, María Aguirre
“Cribriform plate.” e-Anatomy, - IMAIOS [Internet]. [cited 2025 July
29].
[10] J
Pinsolle, F San-Galli, F Siberchicot, et al. “Modified Approach for Ethmoid and
Anterior Skull Base Surgery Arch
Otolaryngol Head Neck Surg, vol. 117, no. 7, pp. 779-782, 1991.
View at: Publisher Site | PubMed
[11] Venkata Joga Prasanth Mokhasanavisu,
Rohit Singh, R Balakrishnan, et al. “Ethnic Variation of Sinonasal Anatomy on
CT Scan and Volumetric Analysis.” Indian J Otolaryngol Head Neck Surg, vol.
71, no. 3, pp. 2157-2164, 2019. View at: Publisher Site | PubMed
[12] Marta Kulich, Ryan Long, Francis
Reyes Orozco, et al. “Racial, Ethnic, and Gender Variations in Sinonasal
Anatomy.” Ann Otol Rhinol Laryngol, vol. 132, no. 9, pp. 996-1004, 2023. View
at: Publisher Site | PubMed
[13] J Earwaker “Anatomic variants in sinonasal CT.” RadioGraphics, vol. 13, no. 2, 1993. View at: Publisher Site
