Received: Wed 26, Nov 2025
Accepted: Sat 13, Dec 2025
Abstract
Background: The incidence and clinicopathological features of meningioma in Manitoba were last reported over three decades ago. The purpose of our study was to assess the incidence and outcomes of surgically resected, pathologically proven meningiomas in the province of Manitoba, including features associated with recurrence.
Methods: A single-centre retrospective study was conducted to assess the incidence of surgically resected, pathologically proven meningiomas between 2011 to 2021. Eligible patients for the study were identified using the neuropathology database and other clinical, radiologic, and demographic details were obtained from clinical chart review.
Results: The total number of patients included in the study period was 521 with the calculated incidence rate of 0.038 (95% CI 0.035-0.041) cases per 1000-person year for pathologically proven meningiomas in Manitoba. Tumour features significantly associated with recurrence included spontaneous necrosis (p < 0.001), ≥4 mitoses per 10 high-power fields (p < 0.001), brain invasion (p = 0.007), and sheeting (p = 0.009).
Conclusion: This study provides valuable insights into the recent incidence rate of meningiomas in our province as well as pathological features associated with recurrence. These results emphasize the need for ongoing surveillance and characterization of meningiomas in our province and beyond, including those managed non-operatively.
Keywords
Brain tumours, meningiomas, neuro-oncology, neuropathology, neuroradiology, neurosurgery, radiation oncology
1. Introduction
Meningiomas are the most common primary central nervous system (CNS) and intracranial tumour in adults [1-3]. Meningiomas are more common with older age and in females at an approximate 2:1 ratio [1, 4-15]. Among those aged 20 and older, meningiomas represent the most common spinal cord tumour [4]. Most patients with meningiomas are asymptomatic and can be managed conservatively with observation [1, 14, 16]. However, common symptoms include headache, focal cranial nerve deficits, seizures, cognitive change, weakness, and vertigo or dizziness which may warrant intervention such as surgery or radiotherapy based on the clinical context [5, 8]. The presenting symptoms are influenced by the location of the meningioma [1]. Other indications for intervention include patient preference and radiological progression [14].
The mainstay of meningioma grading is pathological evaluation. The CNS World Health Organization (WHO) classification system is used to designate grade 1, 2, or 3, the most recent iteration of which is WHO CNS-5 2021 [3, 16, 17]. The majority of meningiomas, approximately 80%, fall under CNS WHO Grade 1 [2, 3]. Up to 18% of meningiomas are CNS WHO Grade 2, also known as atypical meningiomas, with the remaining < 2% meeting the criteria for CNS WHO Grade 3, also known as anaplastic or malignant meningiomas [2, 3]. As per the WHO CNS-5 2021 criteria, meningiomas are considered CNS WHO Grade 2 if the tumour exhibits 4-19 mitoses per 10 high-power fields (HPF), brain invasion, chordoid or clear cell histological subtype, and/or the presence of three or more of five specific histologic features [2, 3, 17]. These histologic features include sheeted architecture, small cell change with high nuclear-to-cytoplasmic ratio, prominent nucleoli, hypercellularity, or spontaneous necrosis [2, 3, 17]. CNS WHO Grade 3 meningiomas are characterized by ≥ 20 mitoses per 10 HPF, TERT promoter (pTERT) mutation, CDKN2A/B homozygous deletion, and/or frank histological anaplasia with sarcoma, carcinoma, or melanoma-like appearance [2, 3, 17]. Pediatric meningiomas are very rare, making up 0.4-2.5% of all diagnosed meningiomas, as well as having more aggressive features and presentations [18, 19].
Alongside CNS WHO Grade, the extent of surgical resection is an important prognostication factor for meningiomas; together these factors drive management decisions and predict outcomes [2, 15]. Residual tumour is commonly assessed using postoperative imaging, typically magnetic resonance imaging (MRI), though surgeons may also utilize the Simpson Grade system to provide an intraoperative evaluation. However, due to the subjectivity and reliance on visual assessment inherent to the Simpson Grade, its usage is considered controversial [15, 20]. As a result, postoperative imaging may provide a more accurate assessment by also factoring in residual tumour volume, which is then reported as gross total resection (GTR) if there is no residual tumour or subtotal resection (STR) if there is residual tumour [20]. Preoperative embolization, at the discretion of the treating team, is rarely performed as an adjunct for highly vascular or large meningiomas [21-23].
In addition to surgical intervention, radiotherapy (RT) may be used as an adjunct or even stand-alone therapy [15, 18]. This includes external beam radiotherapy (EBRT) and stereotactic radiosurgery (SRS), of which gamma knife (GK) is a common modality that is used in Manitoba [18]. Deciding between the modalities is based on the CNS WHO Grade, size, and location of the tumour. SRS is favoured for meningiomas that are low-risk and small (1-3 cm), while EBRT tends to be preferred for tumours that are recurrent, close to organs at risk such as the optic pathway, and involve multiple lesions [24, 25]. RT is strongly recommended as adjunct treatment following resection of CNS WHO Grade 2 and 3 meningiomas to improve progression-free survival and overall survival rates [1, 26-32]. On the other hand, the benefits of RT as an adjunct to surgical resection in CNS WHO Grade 1 and grossly-resected CNS WHO Grade 2 meningiomas continues to be investigated with mixed evidence [33]. When feasible, gross-total resection is the management of choice for CNS WHO Grade 1 meningiomas, though studies have suggested favourable outcomes with stereotactic radiosurgery alone [34-37]. In our centre, GK-based therapies are offered to patients when local progression of disease or a new recurrent lesion was identified. These cases were reviewed in SRS/GK rounds with the neurosurgeon and radiation oncologist.
The last studies to broadly characterize meningiomas in Manitoba were published by Sutherland et al. (1987) and Rohringer et al. (1989) reviewing patients from 1980-1985 and 1980-1987, respectively, and reporting a crude incidence rate of 2.3 cases per 100,000 for all meningiomas [5, 38] This identifies a gap in research on the incidence of meningiomas, especially focused on those that were surgically-resected, despite being the most common CNS tumour. Our study aims to build a more recent database on meningiomas in Manitoba, allowing for the characterization of their burden on the province including incidence rates, clinical presentations, tumour features, course of treatment, and outcomes. This information can then inform the appropriate allocation of resources to their diagnosis and management.
2. Methods
The study was approved by the University of Manitoba Research Ethics Board (HS24731 (H2021:099)) and Provincial Health Research Privacy Committee. Data collection from the CancerCare Manitoba database had ethics approval from the Research Review Impact Committee (RRIC).
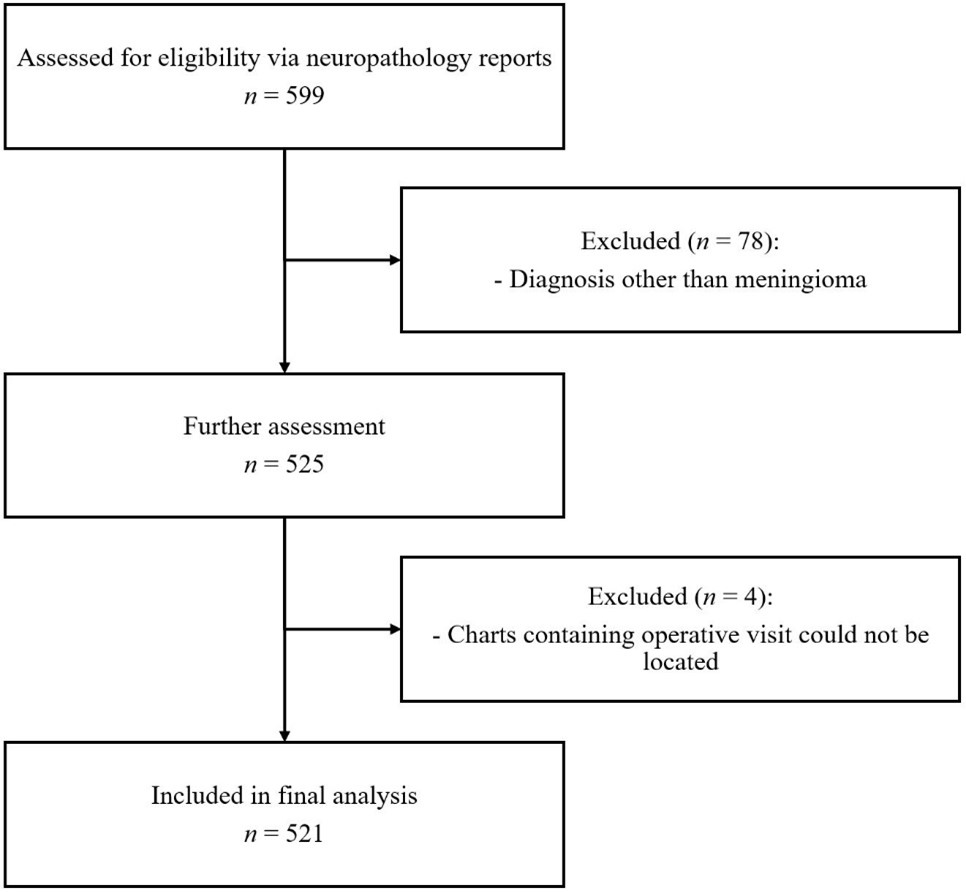
A retrospective analysis was performed on consecutive patients with surgically resected, pathologically proven meningiomas diagnosed at Health Sciences Centre (HSC) in Winnipeg, Manitoba between January 1, 2011, and June 30, 2021. HSC is the only neurosurgical centre in Manitoba, and all meningiomas in the province were surgically treated and pathologically diagnosed at HSC. Patients were identified from neuropathology database during the study period. Consecutive patients of any age group with neuropathological diagnosis of meningioma were included in the study. Patients were excluded if their clinical charts containing their operative notes were not available for review, and no patients were excluded based on incomplete imaging. Meningiomas were re-evaluated using the WHO CNS-5 2021 classification and re-graded if applicable [17]. This meant that papillary or rhabdoid morphology alone was not used as sufficient criteria to be CNS WHO Grade 3 [39].
Detailed clinical chart reviews were performed to collect data on the demographics, clinical presentations, neuropathology, surgeries, radiation treatments, and patient follow-up for recurrence. The locations of meningiomas were determined based on neuropathology, neuroradiology, and operative reports. These were categorized as convexity and parasagittal; skull base subdivided into frontobasal (planum sphenoidale, olfactory groove, tuberculum sellae, anterior clinoid, cribriform plate) and middle fossa (sphenoid ridge, cavernous sinus, petroclival); posterior fossa (subdivided into tentorium, cerebellar convexity, cerebellopontine angle, foramen magnum, and clivus); lateral ventricle; intraorbital; spinal subdivided into cervical, thoracic, and lumbar; and others which included third ventricle, pineal, and ectopic meningiomas that involved the lung and extracranial regions. A neuropathologist (NS) conducted the review of the results.
Operative reports were reviewed to collect information on the Simpson Grade, GTR vs. STR, and whether preoperative embolization was performed. If a range of Simpson Grades were provided, the grade associated with a less degree of resection was used to consider the worst-case scenario. For example, if Simpson Grade I-II was recorded, we considered it as Simpson Grade II for the sake of analysis. Postoperative imaging reports were used to evaluate whether a complete vs. partial resection was performed based on assessment for residual tumour. In cases where the interpreting radiologist could not confidently distinguish whether the enhancement around the resection cavity represented residual tumour or expected post-surgical change, the extent of resection was based on the Simpson Grade, if available. Simpson Grades I-III were considered GTR and Simpson Grade IV was considered STR. Residual tumour volume was inconsistently reported, especially in the presence of post-surgical change, and thus could not be reliably quantified and included in recurrence analyses. The CancerCare Manitoba database was accessed to collect information on radiation treatments received by patients pre- and postoperatively, including the modality and dose of radiation.
2.1. Statistics
The descriptive variables were summarized and analyzed using chi-square or Fisher’s exact test for categorical variables, and t-tests or Mann-Whitney U tests for continuous variables, as appropriate. The incidence rates were calculated using the mid-period population of Manitoba in 2016, of 1,314,140 people (655,415 men, 658,725 women) [40]. The incidence rates used this value uniformly in Poisson analysis to find the 95% confidence intervals.
3. Results
In our single-centre retrospective review of patients with surgically resected, pathologically proven meningiomas, significant associations were found between higher CNS WHO Grade meningiomas and tumour location, larger tumour size, and receiving postoperative RT. Tumour features independently associated with recurrence were also identified, including spontaneous necrosis, ≥4 mitoses per 10 HPF, brain invasion, and sheeting. No significant associations were observed between recurrence and sex, age group, presenting symptoms, or preoperative tumour size.
3.1. Patient Characteristics
Over the 10.5-year study period, there were 525 patients (female: male ratio 2.4:1 [367:154]; mean age 59.14 [±1.117] years based on t-distribution, and median age 61 [20-92] years) with pathologically proven meningiomas at our institution (Table 1). Of these, 4 patients were excluded from the study as their charts could not be located for review. Using the mid-period (2016) population estimate, the calculated incidence was 50.0 patients per year and the incidence rate was 0.038 (95% CI 0.035-0.041) cases per 1000-person year for pathologically proven meningiomas in Manitoba. This can be further divided into an incidence rate of 0.023 (95% CI 0.019-0.026) cases per 1000-person year for men and 0.054 (95% CI 0.048-0.059) for women. There was no significant association between male sex and CNS WHO Grade 2/3 meningiomas (p = 0.292; Table 2). The distribution of CNS WHO Grade significantly varied across age groups (p = 0.023), with patients over 70 years of age having the highest proportion of CNS WHO Grade 2/3 meningiomas (42/120, 35%). Most meningiomas were CNS WHO Grade 1 (358/521, 68.7%), followed by CNS WHO Grade 2 (133/521, 25.5%) and CNS WHO Grade 3 (6/521, 1.2%) (Table 1). 33 patients (33/133, 24.8%) met CNS WHO Grade 2 criteria based on brain invasion alone.
Table. 1. Patient baseline characteristics (N = 521).
|
Demographics |
Value |
|
Age, median (range) |
61 (20-92) |
|
Age, mean (95% CI) |
59.14 ±1.117 |
|
Age (%) |
|
|
20-29 |
13 (2.5%) |
|
30-39 |
31 (6.0%) |
|
40-49 |
74 (14.2%) |
|
50-59 |
126 (24.2%) |
|
60-69 |
139 (26.7%) |
|
70-79 |
104 (20.0%) |
|
80-89 |
31 (6.0%) |
|
90-99 |
3 (0.6%) |
|
Sex (%) |
|
|
Male |
154 (29.6%) |
|
Female |
367 (70.4%) |
|
CNS WHO Grade (%) |
|
|
1 |
358 (68.7%) |
|
2 |
133 (25.5%) |
|
3 |
6 (1.2%) |
|
None (ungraded) |
24 (4.6%) |
|
Laterality (%), n =
445 |
|
|
Left |
229 (51.5%) |
|
Right |
183 (41.1%) |
|
Midline |
33 (7.4%) |
|
Location (%) |
|
|
Convexity, Parasagittal |
161 (30.9%) |
|
Skull base |
230 (44.1%) |
|
Frontobasal* |
140 (26.9%) |
|
Middle cranial fossa** |
89 (17.1%) |
|
Posterior fossa*** |
69 (13.2%) |
|
Lateral ventricle |
4 (0.8%) |
|
Intraorbital |
5 (1.0%) |
|
Other |
6 (1.2%) |
|
Spinal cord |
46 (8.7%) |
|
Cervical |
10 (2.0%) |
|
Thoracic |
33 (6.3%) |
|
Lumbar |
2 (0.4%) |
*Frontobasal includes
planum sphenoidale, olfactory groove, tuberculum sellae, anterior clinoid, and
cribriform plate.
**Middle fossa includes
sphenoid ridge, cavernous sinus, and petroclival.
***Posterior fossa includes tentorium, cerebellar convexity, cerebellopontine angle, foramen magnum, and clivus.
A CNS WHO Grade was not assigned for 24 patients (24/521, 4.6%). These cases were excluded from grade-based comparative analyses but included in incidence calculations. A reason for not assigning a grade included radiologically suspected brain invasion that was unconfirmed pathologically because of insufficient brain tissue specimen. Another reason was the presence of worrisome features suggestive of a higher grade, despite the tumour overall meeting only the criteria of CNS WHO Grade 1. Examples of worrisome features included focal areas with increased Ki-67 proliferation rate, focal areas meeting subjective criteria, local growth from a CNS WHO Grade 2 meningioma but CNS WHO Grade 1 based strictly on the specimen provided, and measurements of aggressive growth behavior such as elevated Ki-67 labeling index in the absence of elevated mitoses.
3.2. Tumour Characteristics
In our study, meningiomas were most commonly located in the skull base (230/521, 44.1%), including frontobasal (140/521, 26.9%) and middle fossa (89/521, 17.1%); convexity and parasagittal (161/521, 30.9%); and posterior fossa (69/521, 13.2%) regions (Table 1). There was a significant association between higher CNS WHO Grade and location (p < 0.001; Table 2). Meningiomas resected from the posterior fossa and spine tend to be CNS WHO Grade 1, compared to skull base and convexity/parasagittal tumours often presenting with CNS WHO Grade 2/3.
Five patients underwent resection of multiple meningiomas, all of whom were female patients with CNS WHO Grade 1 meningiomas. Of note, one of these patients had two spinal meningiomas at different levels of the thoracic spine. Of the 46 (46/521, 8.8%) spinally located meningiomas, thoracic was the most common with 33 (33/46, 71.7%) identified. The median tumour size for CNS WHO Grade 2/3 meningiomas was 4.70 cm [3.45-6.30], which was significantly larger (p < 0.001) than CNS WHO Grade 1 meningiomas which were 3.70 cm [2.30-4.90] (Table 2).
Table. 2. Comparison of Patient Characteristics and Tumour Features Between WHO
Grade 1 and Grade 2/3 Meningiomas.
|
Tumour Grade 1 |
Tumour Grade 2/3 |
p-value |
|
|
Sex |
|||
|
Female |
256 |
92 |
0.292 |
|
Male |
102 |
47 |
|
|
Age Group |
|||
|
<50 |
77 |
37 |
0.023 |
|
50-70 |
203 |
60 |
|
|
>70 |
78 |
42 |
|
|
Headache (N = 496) |
|||
|
Yes |
136 |
55 |
0.841 |
|
No |
221 |
84 |
|
|
Brain Invasion (N = 226) |
|||
|
Yes |
0 (0) |
58 (61) |
- |
|
No |
131 |
37 |
|
|
Spontaneous Necrosis (N = 492) |
|||
|
Yes |
37 |
72 |
< 0.001 |
|
No |
316 |
67 |
|
|
Mitoses ≥4/10 HPF |
|||
|
Yes |
0 |
83 |
- |
|
No |
358 |
56 |
|
|
Prominent Nucleoli (N = 496) |
|||
|
Yes |
18 |
34 |
< 0.001 |
|
No |
339 |
105 |
|
|
Hypercellularity |
|||
|
Yes |
89 |
64 |
< 0.001 |
|
No |
269 |
75 |
|
|
Small-Cell Changes |
|||
|
Yes |
45 |
48 |
< 0.001 |
|
No |
313 |
91 |
|
|
Sheeting |
|||
|
Yes |
13 |
27 |
< 0.001 |
|
No |
345 |
112 |
|
|
Postoperative Radiation |
|||
|
Yes |
56 |
42 |
< 0.001 |
|
No |
302 |
97 |
|
|
Radiation Modality |
|||
|
EBRT |
13 |
18 |
< 0.001 |
|
GK |
39 |
15 |
|
|
Both |
5 |
9 |
|
|
None |
301 |
97 |
|
|
Location |
|||
|
Convexity,
Parasagittal |
101 |
48 |
< 0.001 |
|
Skull Base |
148 |
75 |
|
|
Posterior Fossa |
62 |
6 |
|
|
Spine |
40 |
6 |
|
|
Other |
7 |
4 |
|
|
Tumour Size (N = 496) |
|||
|
median [IQR] |
3.70 [2.30-4.90] |
4.70 [3.45-6.30] |
< 0.001 |
3.3. Surgical Characteristics
The Simpson Grade was not consistently denoted in the surgical operative reports. The Simpson Grade was reported in 115 patients (115/521, 22.1%), with a distribution as follows: Simpson Grade I (48/115, 41.7%), Grade II (47/115, 40.9%), Grade III (11/115, 9.6%), Grade IV (9/115, 7.8%), and Grade V (0/115, 0.0%). There were 337 (337/521, 64.7%) patients with GTRs and 176 (176/521, 33.8%) with STRs, evaluated using postoperative imaging and further informed by the operative report and Simpson Grade, when provided. Preoperative embolization was performed for 10 patients (10/521, 1.9%). Peri-operative complications of any type were reported for 176 patients (176/521, 33.8%).
Table. 3. Comparison of Patient Characteristics and Tumour Features by Tumour
Recurrence Status.
|
Recurrence |
No Recurrence |
p-value |
||
|
Sex (N = 268) |
|
|||
|
Female |
26 |
164 |
0.661 |
|
|
Male |
13 |
65 |
|
|
|
Age Group (N = 268) |
|
|||
|
<50 |
8 |
50 |
0.951 |
|
|
50-70 |
22 |
131 |
|
|
|
>70 |
9 |
48 |
|
|
|
Headache (N = 268) |
|
|||
|
Yes |
15 |
83 |
0.932 |
|
|
No |
24 |
146 |
|
|
|
Brain Invasion (N = 120) |
|
|||
|
Yes |
13 |
30 |
0.007 |
|
|
No |
8 |
69 |
|
|
|
Spontaneous Necrosis (N = 267) |
|
|||
|
Yes |
14 |
45 |
< 0.001 |
|
|
No |
25 |
183 |
|
|
|
Mitoses ≥4/10 HPF (N = 268) |
|
|||
|
Yes |
17 |
42 |
< 0.001 |
|
|
No |
22 |
187 |
|
|
|
Prominent Nucleoli (N = 268) |
|
|||
|
Yes |
6 |
30 |
0.894 |
|
|
No |
33 |
199 |
|
|
|
Hypercellularity (N = 268) |
|
|||
|
Yes |
10 |
69 |
0.705 |
|
|
No |
29 |
160 |
|
|
|
Small-Cell Changes (N = 268) |
|
|||
|
Yes |
6 |
56 |
0.300 |
|
|
No |
33 |
173 |
|
|
|
Sheeting (N = 268) |
|
|||
|
Yes |
8 |
15 |
0.009 |
|
|
No |
31 |
214 |
|
|
|
Tumour Size (N = 267) |
|
|||
|
median [IQR] |
4.00 [3.25-4.85] |
3.90 [2.80-5.20] |
0.670 |
|
3.4. Pathological Characteristics
Tumour features on pathology that had a significant association with recurrence included spontaneous necrosis (p < 0.001), ≥ 4 mitoses per 10 HPF (p < 0.001), brain invasion (p = 0.007), and sheeting (p = 0.009) (Table 3). Histological subtypes were reported in a minority of neuropathological reports. There were 2 meningothelial, 4 angiomatous, 3 microcystic, 4 mixed angiomatous/microcystic, 4 fibrous, 5 chordoid, and 4 of clear cell subtype meningiomas identified on the neuropathology reports. As a result, histological subtypes were not used for further analysis.
3.5. Radiation Therapy
There was a significant association between higher CNS WHO Grade meningiomas and the administration of postoperative RT (p < 0.001; Table 2). CNS WHO Grade 2/3 meningiomas were more likely to receive postoperative RT, including 30.2% (42/139) of CNS WHO Grade 2/3 compared to 15.6% (56/358) of CNS WHO Grade 1. Meningiomas that received RT at any point, including any length of time prior to or after surgical resection, included 17.6% (63/358) of CNS WHO Grade 1 meningiomas and 35.3% (49/139) of CNS WHO Grade 2/3 meningiomas.
RT modality significantly differed between the groups as well (p < 0.001; Table 2). CNS WHO Grade 2/3 meningiomas had a relatively higher use of EBRT (18/139, 12.9% of all CNS WHO Grade 2/3; 13/358, 3.6% of all CNS WHO Grade 1) and combination therapies (9/139, 6.5% of all CNS WHO Grade 2/3; 5/358, 1.4% of all CNS WHO Grade 1). In terms of RT modalities among patients of each CNS WHO Grade receiving RT, 44 (44/358, 12.3%) CNS WHO Grade 1 meningiomas received GK and 18 (18/358, 5.0%) received EBRT. In comparison, of the CNS WHO Grade 2/3 meningiomas receiving RT, 24 (24/139, 17.3%) received GK and 27 (27/139, 19.4%) received EBRT.
4. Discussion
The incidence rate from our study was greater than that of Rohringer et al. [5] This was despite the Rohringer et al. study including several meningiomas that did not undergo surgical biopsy nor resection [5]. One potential explanation for this is the aging population [1, 7, 14, 15]. An important contributing factor is the general trend of increasing accessibility and usage of modern imaging modalities such as computed tomography and magnetic resonance imaging, which may subsequently increase the rate of incidental meningioma findings [7, 15, 18]. Despite up to 20% of new diagnoses of meningiomas being incidentally-discovered and asymptomatic, it can be distressing for patients [7]. The Manitoban incidence rate from our study was comparable to that of other study centres in New Zealand and Germany, although both studies included patients from earlier time periods of 2000 to the early-mid 2010s [41, 42].
The distribution of sex (female: male ratio 2.4:1) and mean age of patients in our study (59.14 years), with most patients being aged 60-69 years, resembled that of other studies on surgically resected meningiomas [8-12]. Interestingly, no pediatric patients were identified, with the youngest patient being 20 years of age at the time of surgery. This is despite our study capturing all meningiomas surgically resected and pathologically evaluated for diagnosis in Manitoba.
The proportions of CNS WHO Grade 1, 2, and 3 surgically resected meningiomas were similar to that of other studies [13, 41, 42]. Despite the introduction of the CNS WHO 2016 criteria during the study period, this has been shown to have minimal impact on the distribution of meningioma grades [39]. However, the cIMPACT-NOW update 8 provides new evidence that the meningiomas with solely brain invasion should no longer be classified as CNS WHO Grade 2 [43]. If incorporated into updated grading criteria, this would reassign 33 (33/133, 24.8%) CNS WHO Grade 2 meningiomas as CNS WHO Grade 1, further increasing the proportion of benign meningiomas resected in the study period.
There was a greater proportion of skull base meningiomas among our population and decreased proportion of convexity and parasagittal meningiomas as compared to other studies [8, 10, 11, 13]. Left-sided meningiomas were also more common than right-sided meningiomas in other studies, though right sided meningiomas being more common has also been observed [11-13]. Spinal meningiomas most commonly presented in the thoracic region, in line with the literature [44].
Our study identified tumour features significantly associated with recurrence including spontaneous necrosis (p < 0.001), ≥4 mitoses per 10 HPF (p < 0.001), brain invasion (p = 0.007), and sheeting (p = 0.009) (Table 3). Despite the individual histologic criteria holding equal weight in grading meningiomas, spontaneous necrosis and sheeting demonstrated significant associations with recurrence in our study. However, there is mixed evidence in the literature to support our findings, possibly attributed to the interobserver and sampling variability inherent to the histologic minor criteria [45-48].
Patients in our study with higher grade meningiomas were more likely to undergo adjuvant radiotherapy and be treated with EBRT than those with lower grade meningiomas. However, of the 139 patients with higher grade meningiomas, only 42 patients received radiotherapy because observation is offered as an option to those with CNS WHO Grade 2 meningiomas. These cases integrated patient preference and goals of care alongside the clinical recommendations of the neurosurgeon and radiation oncologist in SRS/GK rounds.
4.1. Limitations
Our study was limited by its retrospective single-centre design, selection bias towards surgically resected meningiomas, and lack of molecular profiling data. Only including meningiomas that underwent surgical resection and subsequent pathological evaluation may significantly underestimate the incidence rate of all meningiomas in the province, especially considering that most meningiomas are managed conservatively [1, 7, 14]. Furthermore, recurrence may be underestimated due to the exclusion of patients with missing charts or incomplete postoperative imaging and follow-up.
Inconsistencies of reporting symptoms, location, histological subtypes, and Simpson Grade limited our ability to perform robust data collection and analysis of these variables. Missing and incomplete data such as ungraded meningiomas and lack of reported residual tumour volume may lead to the underestimation of recurrence risk in patients with higher-grade features or incomplete resections. Furthermore, the lack of standardized location classification poses a conflict in comparing the locations of cranial meningiomas to other studies [8, 10, 11, 13]. For the purposes of future studies into meningiomas in Manitoba and other institutions, establishing consistent reporting guidelines for these features would strengthen future studies as well as the ease of data collection.
Lastly, our study did not include the molecular profiles of meningiomas, including pTERT and CDKN2A/B [2, 3, 17]. This may affect the distribution of lower-grade meningiomas in our study.
5. Conclusion
The incidence rate from our study of 0.038 (95% CI 0.035-0.041) cases per 1000-person year for pathologically proven meningiomas demonstrates an interval increase. The CNS WHO Grade of meningioma was associated with several tumour features as well as recurrence. These findings will help optimize the appropriate population-based allocation of resources for the diagnosis, treatment, and follow-up of meningiomas and emphasizes the need for ongoing surveillance and characterization of meningiomas in our province and beyond.
Disclosure
JS is principal investigator of EMMA Can study funded by Medtronic Canada. The other authors have no conflicts of interest to report.
Author Contributions
Jaydon Gan: Investigation, Data curation, Formal analysis, Visualization, Writing - Original draft. Nikunj Patil: Investigation, Data curation, Writing - review & editing. Yan Sin Leung: Formal analysis, Visualization, Writing - review & editing. Susan Alcock: Resources, Writing - review & editing. Saranya Kakumanu: Conceptualization, Supervision, Writing - review & editing. Jai Shankar: Conceptualization, Supervision, Writing - review & editing. Namita Sinha: Conceptualization, Resources, Supervision, Writing - review & editing.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Abbreviations
CNS: Central Nervous System
WHO: World Health Organization
HPF: High-Power Field(s)
TERT: Telomerase Reverse Transcriptase
pTERT: Telomerase Reverse Transcriptase Promoter Region
CDKN2A/B: Cyclin-Dependent Kinase Inhibitor 2A/B
MRI: Magnetic Resonance Imaging
GTR: Gross Total Resection
STR: Subtotal Resection
RT: Radiotherapy
EBRT: External Beam Radiotherapy
SRS: Stereotactic Radiosurgery
GK: Gamma Knife
RRIC: Research Review Impact Committee
HSC: Health Sciences Centre
CI: Confidence Interval
IQR: Interquartile Range
cIMPACT-NOW: Consortium to Inform Molecular and Practical Approaches to CNS Tumor Taxonomy
REFERENCES
[1] Christian Ogasawara,
Brandon D Philbrick, D Cory Adamson “A Review of Epidemiology, Pathology,
Diagnosis, Treatment, and Future Directions.” Biomedicines, vol. 9, no.
3, pp. 319, 2021. View at: Publisher
Site | PubMed
[2] William C Chen, Haley K
Perlow, Abrar Choudhury, et al. “Radiotherapy for meningiomas.” J Neurooncol,
vol. 160, no. 2, pp. 505-515, 2022. View at: Publisher Site | PubMed
[3] Elena Marastoni, Valeria
Barresi “Meningioma Grading beyond Histopathology: Relevance of Epigenetic and
Genetic Features to Predict Clinical Outcome.” Cancers, vol. 15, no. 11,
pp. 2945, 2023. View at: Publisher
Site | PubMed
[4] Quinn T Ostrom, Mackenzie
Price, Corey Neff, et al. “CBTRUS Statistical Report: Primary Brain and Other
Central Nervous System Tumors Diagnosed in the United States in 2016—2020.” Neuro
Oncol, vol. 25, no. 12 Suppl 2, pp. iv1-iv99, 2023. View at: Publisher Site | PubMed
[5] M Rohringer, G R
Sutherland, D F Louw, et al. “Incidence and clinicopathological features of
meningioma.” J Neurosurg, vol. 71, no. 5 Pt 1, pp. 665-672, 1989. View
at: Publisher
Site | PubMed
[6] Junguo Cao, Weijia Yan,
Guihong Li, et al. “Incidence and survival of benign, borderline, and malignant
meningioma patients in the United States from 2004 to 2018.” Int J Cancer,
vol. 151, no. 11, pp. 1874-1888, 2022. View at: Publisher Site | PubMed
[7] Abdurrahman I Islim,
Christopher P Millward, Samantha J Mills, et al. “The management of incidental
meningioma: An unresolved clinical conundrum.” Neurooncol Adv, vol. 5,
no. Suppl 1, pp. i26-i34, 2023. View at: Publisher Site | PubMed
[8] Stephen T. Magill, Jacob
S. Young, Ricky Chae, et al. “Relationship between tumor location, size, and
WHO grade in meningioma.” Neurosurg Focus, vol. 44, no. 4, pp. E4, 2018.
View at: Publisher
Site
[9] Ari J Kane, Michael E
Sughrue, Martin J Rutkowski, et al. “Anatomic location is a risk factor for
atypical and malignant meningiomas.” Cancer, vol. 117, no. 6, pp.
1272-1278, 2011. View at: Publisher
Site | PubMed
[10] Chongran Sun, Zhangqi Dou,
Jiawei Wu, et al. “The Preferred Locations of Meningioma According to Different
Biological Characteristics Based on Voxel-Wise Analysis.” Front Oncol,
vol. 10, pp. 1412, 2020. View at: Publisher Site | PubMed
[11] Maria Kamenova, Raphael
Guzman, Jehuda Soleman, et al. “Demographics and outcome of histologically
confirmed intracranial meningiomas.” Clin Transl Neurosci, vol. 3, pp.
2514183X19894945, 2019. View at: Publisher Site
[12] Alba Corell, Erik Thurin,
Thomas Skoglund, et al. “Neurosurgical treatment and outcome patterns of
meningioma in Sweden: a nationwide registry-based study.” Acta Neurochir,
vol. 161, pp. 333-341, 2019. View at: Publisher Site
[13] Sayied Abdol Mohieb
Hosainey, David Bouget, Ingerid Reinertsen, et al. “Are there predilection
sites for intracranial meningioma? A population-based atlas.” Neurosurg Rev,
vol. 45, pp. 1543-1552, 2022. View at: Publisher Site
[14] Olivia
Näslund, Thomas Skoglund, Dan Farahmand, et al.. “Indications and outcome in
surgically treated asymptomatic meningiomas: a single-center case-control
study.” Acta Neurochir (Wien), vol. 162, no. 9, pp. 2155-2163, 2020. View
at: Publisher Site | PubMed
[15] Alper Dincer, Saul F.
Morales-Valero, Stephanie M. Robert, et al. “Surgical strategies for
intracranial meningioma in the molecular era.” J Neurooncol, vol. 162,
pp. 253-265, 2023. View at: Publisher
Site
[16] Varun Yarabarla, Amrutha
Mylarapu, Tatiana J Han, et al. “Intracranial meningiomas: an update of the
2021 World Health Organization classifications and review of management with a
focus on radiation therapy.” Front Oncol, vol. 13, pp. 1137849, 2023.
View at: Publisher
Site | PubMed
[17] Pouya Jamshidi, Daniel J
Brat “The 2021 WHO classification of central nervous system tumors: what
neurologists need to know.” Curr Opin Neurol, vol. 35, no. 6, pp.
764-771, 2022. View at: Publisher
Site | PubMed
[18] Justin Z Wang, Alexander P
Landry, David R Raleigh, et al. “Meningioma: International Consortium on
Meningiomas consensus review on scientific advances and treatment paradigms for
clinicians, researchers, and patients.” Neuro Oncol, vol. 26, no. 10,
pp. 1742-1780, 2024. View at: Publisher
Site | PubMed
[19] Angus Toland, Samantha N
McNulty, Melike Pekmezci, et al. “Pediatric meningioma: a clinicopathologic and
molecular study with potential grading implications.” Brain Pathol, vol.
30, no. 6, pp. 1134-1143, 2020. View at: Publisher Site | PubMed
[20] Silky Chotai, Theodore H
Schwartz “The Simpson Grading: Is It Still Valid?” Cancers (Basel), vol.
14, no. 8, pp. 2007, 2022. View at: Publisher Site | PubMed
[21] F Trivelatto, G S Nakiri,
M Manisor, et al. “Preoperative Onyx Embolization of Meningiomas Fed by the
Ophthalmic Artery: A Case Series.” AJNR Am J Neuroradiol, vol. 32, no.
9, pp. 1762-1766, 2011. View at: Publisher
Site | PubMed
[22] D M S Raper, R M Starke, F
Henderson Jr, et al. “Preoperative Embolization of Intracranial Meningiomas:
Efficacy, Technical Considerations, and Complications.” AJNR Am J
Neuroradiol, vol. 35, no. 9, pp. 1798-1804, 2014. View at: Publisher Site | PubMed
[23] M Iacobucci, L Danieli, E
Visconti, et al. “Preoperative embolization of meningiomas with polyvinyl
alcohol particles: The benefits are not outweighed by risks.” Diagn Interv
Imaging, vol. 98, no. 4, pp. 307-314, 2017. View at: Publisher Site | PubMed
[24] Caroline
Apra, Matthieu Peyre, Michel Kalamarides “Current treatment options for
meningioma.” Expert Rev Neurother, vol. 18, no. 3, pp. 241-249, 2018. View
at: Publisher Site | PubMed
[25] Matthew
S. Susko, David R. Raleigh “Radiotherapy for Meningioma.” In: Zadeh G,
Goldbrunner R, Krischek B, Nassiri F, eds. Biological and Clinical Landscape
of Meningiomas. Springer International Publishing; pp. 95-106, 2023. View
at: Publisher Site
[26] C Leland Rogers, Minhee
Won, Michael A Vogelbaum, et al. “High-risk Meningioma: Initial Outcomes From
NRG Oncology/RTOG 0539.” Int J Radiat Oncol Biol Phys, vol. 106, no. 4,
pp. 790-799, 2020. View at: Publisher Site | PubMed
[27] Damien C Weber, Carmen
Ares, Salvador Villa, et al. “Adjuvant postoperative high-dose radiotherapy for
atypical and malignant meningioma: A phase-II parallel non-randomized and
observation study (EORTC 22042-26042).” Radiother Oncol, vol. 128, no.
2, pp. 260-265, 2018. View at: Publisher Site | PubMed
[28] Gabriel Verly, Lucas
Bresciani, Thiffany Delfino, et al. “Surgery alone versus surgery plus adjuvant
radiotherapy for WHO grade 2 meningioma: meta-analysis of reconstructed
time-to-event data.” Neurosurg Rev, vol. 47, no. 1, pp.702, 2024. View
at: Publisher
Site | PubMed
[29] Caryn Wujanto, Tabitha Y
Chan, Yu Yang Soon, et al. “Should adjuvant radiotherapy be used in atypical
meningioma (WHO grade 2) following gross total resection? A systematic review
and Meta-analysis.” Acta Oncol, vol. 61, no. 9, pp. 1075-1083, 2022.
View at: Publisher
Site | PubMed
[30] Se-Woong Chun, Kyung Min
Kim, Min-Sung Kim, et al. “Adjuvant radiotherapy versus observation following
gross total resection for atypical meningioma: a systematic review and
meta-analysis.” Radiat Oncol, vol. 16, no. 1, pp. 34, 2021. View at: Publisher Site | PubMed
[31] Leland Rogers, Peixin
Zhang, Michael A Vogelbaum, et al. “Intermediate-risk meningioma: initial
outcomes from NRG Oncology RTOG 0539.” J Neurosurg, vol. 129, no. 1, pp.
35-47, 2018. View at: Publisher
Site | PubMed
[32] Amon Bergner, Andrea
Daniela Maier, Christian Mirian, et al. “Adjuvant radiotherapy and stereotactic
radiosurgery in grade 3 meningiomas — a systematic review and meta-analysis.” Neurosurg
Rev, vol. 45, no. 4, pp. 2639-2658, 2022. View at: Publisher Site | PubMed
[33] Mohammed Nadeem, Abhijit
Goyal-Honavar, Palavalasa Sravya, et al. “Prognostic Factors and Outcomes in
World Health Organization Grade 1 and Grade 2 Intracranial Meningiomas—5-Year
Institutional Experience.” World Neurosurg, vol. 187, pp. e331-e339,
2024. View at: Publisher
Site | PubMed
[34] Maximilian I Ruge, Juman
Tutunji, Daniel Rueß, et al. “Stereotactic radiosurgery for treating
meningiomas eligible for complete resection.” Radiat Oncol, vol. 16, no.
1, pp. 22, 2021. View at: Publisher
Site | PubMed
[35] Fernando De Nigris
Vasconcellos, Pavel Pichardo-Rojas, Augusto Muller Fieldler, et al. “Upfront
stereotactic radiosurgery versus adjuvant radiosurgery for parasagittal and
parafalcine meningiomas: a systematic review and meta-analysis.” Neurosurg
Rev, vol. 47, no. 1, pp. 127, 2024. View at: Publisher Site | PubMed
[36] Valentina Pinzi, Marcello
Marchetti, Anna Viola, et al. “Hypofractionated Radiosurgery for Large or in
Critical-Site Intracranial Meningioma: Results of a Phase 2 Prospective Study.”
Int J Radiat Oncol Biol Phys, vol. 115, no. 1, pp. 153-163, 2023. View
at: Publisher
Site | PubMed
[37] Marcello Marchetti, Arjun
Sahgal, Antonio A F De Salles, et al. “Stereotactic Radiosurgery for
Intracranial Noncavernous Sinus Benign Meningioma: International Stereotactic
Radiosurgery Society Systematic Review, Meta-Analysis and Practice Guideline.” Neurosurgery,
vol. 87, no. 5, pp. 879-890, 2020. View at: Publisher Site | PubMed
[38] G R Sutherland, R Florell,
D Louw, et al. “Epidemiology of primary intracranial neoplasms in Manitoba,
Canada.” Can J Neurol Sci, vol. 14, no. 4, pp. 586-592, 1987. View at: PubMed
[39] Alexander D Rebchuk,
Bradley M Chaharyn, Armaghan Alam, et al. “The impact of brain invasion
criteria on the incidence and distribution of WHO grade 1, 2, and 3
meningiomas.” Neuro Oncol, vol. 24, no. 9, pp. 1524-1532, 2022. View at:
Publisher Site | PubMed
[40] Statistics Canada. Table
17-10-0005-01 Population estimates on July 1, by age and gender. 2024. View at:
Publisher Site
[41] Bernd Holleczek, Daniel
Zampella, Steffi Urbschat, et al. “Incidence, mortality and outcome of
meningiomas: A population-based study from Germany.” Cancer Epidemiol,
vol. 62, pp. 101562, 2019. View at: Publisher Site | PubMed
[42] Clinton P Turner, Bert van
der Werf, Andrew J J Law, et al. “The epidemiology of patients undergoing
meningioma resection in Auckland, New Zealand, 2002 to 2011.” J Clin
Neurosci, vol. 80, pp. 324-330, 2020. View at: Publisher Site | PubMed
[43] Felix Sahm, Kenneth D
Aldape, Priscilla K Brastianos, et al. “cIMPACT-NOW update 8: Clarifications on
molecular risk parameters and recommendations for WHO grading of meningiomas.” Neuro
Oncol, vol. 27, no. 2, pp. 319-330, 2025. View at: Publisher Site | PubMed
[44] Christoph Hohenberger,
Peter Hau, Karl-Michael Schebesch, et al. “Spinal meningiomas.” Neurooncol
Adv, vol. 5, no. Suppl 1, pp. i112-i121, 2023. View at: Publisher Site | PubMed
[45] Andrej Vranic, Mara
Popovic, Andrej Cör, et al. “Mitotic Count, Brain Invasion, and Location Are
Independent Predictors of Recurrence-Free Survival in Primary Atypical and
Malignant Meningiomas: A Study of 86 Patients.” Neurosurgery, vol. 67,
no. 4, pp. 1124, 2010. View at: Publisher Site | PubMed
[46] Keitaro CHIBA, Takashi
SUGAWARA, Daisuke KOBAYASHI, et al. “Atypical Histological Features as Risk
Factors for Recurrence in Newly Diagnosed WHO Grade I Meningioma.” Neurol
Med Chir (Tokyo), vol. 61, pp. 647-651, 2021. View at: Publisher Site
[47] Valeria Barresi, Simona
Lionti, Samuel Caliri, et al. “Histopathological features to define atypical
meningioma: What does really matter for prognosis?” Brain Tumor Pathol,
vol. 35, no. 3, pp. 168-180, 2018. View at: Publisher Site | PubMed
[48] Pinar Karabagli, Hakan Karabagli, Zekiye Mavi, et al. “Histopathological and Clinical Features as Prognostic Factors of Atypical Meningiomas.” Turk Neurosurg, vol. 30, no. 5, pp. 746-575, 2020. View at: Publisher Site | PubMed
