Received: Wed 01, Oct 2025
Accepted: Tue 28, Oct 2025
Abstract
Negative- and low-pressure hydrocephalus (NegPH/LPH) is an uncommon and frequently misdiagnosed condition lacking standardised diagnostic and management protocols. We conducted a systematic review of published studies from 1980 to 2024 to synthesise evidence on aetiology, pathophysiology, and treatment. Thirty studies reporting 215 patients were identified, with antecedents including traumatic brain injury, aneurysmal subarachnoid haemorrhage, normal-pressure hydrocephalus, postoperative cases, and spontaneous presentations. Four interacting models were commonly described: viscoelastic brain change with reduced compliance, transmantle pressure gradients, poroelastic or glymphatic dysfunction, and impaired cerebrospinal fluid pulsatility. Management strategies centred on restoring compliance and cerebrospinal fluid pathways through subatmospheric external ventricular drainage, low-pressure or valveless shunting, targeted leak repair, and endoscopic third ventriculostomy. Outcomes were variable, but early recognition and carefully titrated drainage were repeatedly associated with clinical improvement, whereas delayed intervention, infection, and irreversible white-matter injury predicted poor recovery. This review highlights NegPH/LPH as a distinct neurosurgical entity and proposes a diagnostic and management framework, while emphasising the urgent need for multicentre registries with standardised definitions and outcome measures to guide evidence-based care.
Keywords
Negative-pressure hydrocephalus, low-pressure hydrocephalus, intracranial hypotension, brain compliance, cerebrospinal fluid dynamics, endoscopic third ventriculostomy, ventriculoperitoneal shunt, neurosurgical management
1. Introduction
Hydrocephalus derives from the Greek terms “hydro” (ὕδωρ), signifying “water,” and “kephalē” (κεφαλή), meaning “head,” referring to abnormal accumulation of cerebrospinal fluid (CSF) within cerebral ventricles. Traditionally associated with raised intracranial pressure (ICP), in 1965 Hakim and Adams [1] described a presentation of ventriculomegaly and a triad of clinical features without elevated ICP, later termed ‘normal pressure hydrocephalus (NPH).’ Furthermore, Pang and Altschuler [2] identified a variant with abnormally low or even negative CSF pressures, now named Negative-Pressure Hydrocephalus (NegPH) or low-pressure hydrocephalus (LPH).
Over the past three decades, several case series and studies have investigated pathophysiological mechanisms but with no resulting standard diagnostic algorithm or management protocol. A lack of these can contribute to the unique condition’s delayed recognition and can contribute to significant morbidity. This systematic review synthesises the available literature of NegPH and LPH, aiming to clarify underlying mechanisms, evaluate therapeutic intervention, and propose a unified diagnostic framework. Ultimately, improving understanding and standardising information can contribute to positive outcomes and assist clinicians with diagnosis.
2. Methods
A systematic literature search of the Scopus and Medline databases was conducted according to PRSIMA guidelines, and the review protocol was registered in the PROSPERO database (ID CRD42024626137). Studies from 1980 onwards, in English, and involving human subjects were considered. Inclusion criteria required that studies address patients diagnosed with NegPH/LPH and any interventions aimed at managing this condition, including shunt adjustments, external CSF drainage, neck wrapping, or novel treatments. Eligible studies also needed to describe ICP changes, clinical symptom resolution, complications, or mortality.
Randomised controlled trials (RCTs), cohort studies, case-control studies, case series, and case reports were included, provided they were peer-reviewed and contained sufficient methodological and outcome detail. Exclusion criteria included non-peer-reviewed literature, studies on differing hydrocephalus subtypes, and no outcome data.
Search terms were “negative pressure hydrocephalus,” “low pressure hydrocephalus,” “intracranial hypotension,” “cerebrospinal fluid drainage,” “shunt management,” “neck wrapping,” and “intracranial pressure normalization.” These were combined to create the following search strategy: (“negative pressure hydrocephalus” OR “low pressure hydrocephalus” OR “intracranial hypotension”) AND (“shunt management” OR “cerebrospinal fluid drainage” OR “neck wrapping” OR “intracranial pressure normalization”) with date and language filters (publication years >1983 and <2025, English only).
Data extraction was performed manually to include variables of author, year of publication, country of origin, study design, sample size, aim of the study, identified cause of NegPH/LPH, treatment approach, primary findings, and overall outcomes.
RCTs were assessed using the Cochrane Risk of Bias Tool, observational studies were evaluated with the Newcastle–Ottawa Scale, and all other studies (case reports, case series, cohort studies etc.) were appraised using JBI-SUMARI appraisal tools. Two independent reviewers performed these assessments, and any discrepancies were resolved through discussion and, if necessary, consultation with a third reviewer. This process ensured a consistent approach to appraising issues such as selection bias, measurement bias, and confounding variables, thereby enhancing the overall rigour and reliability of the synthesised findings.
A narrative synthesis and thematic analysis were conducted, identifying common themes and recurring trends, enabling a structured overview despite variations. Heterogeneity was assessed by comparing methodological elements across studies. Marked differences in design or data findings were integrated descriptively, ensuring accurate representation.
3. Results
3.1. Study Selection Process
An initial search identified 474 publications (254 from Scopus and 220 from Medline). After removing 25 duplicates, 449 abstracts were screened using JBI SUMARI by two independent reviewers, resulting in 383 exclusions. Of the 66 full texts assessed, 9 conflicts between reviewers were resolved after discussion and debate. A total of 36 full texts were excluded for reasons including irrelevance to NegPH/LPH (24), reviews/editorials (6), commentaries (3), poor translation (1), or non-English language (2). Thirty publications proceeded to the final appraisal, with 6 reviewer conflicts resolved through consensus. The PRISMA flow chart illustrating this selection process is presented in (Figure 1). PRISMA checklist included in appendix.
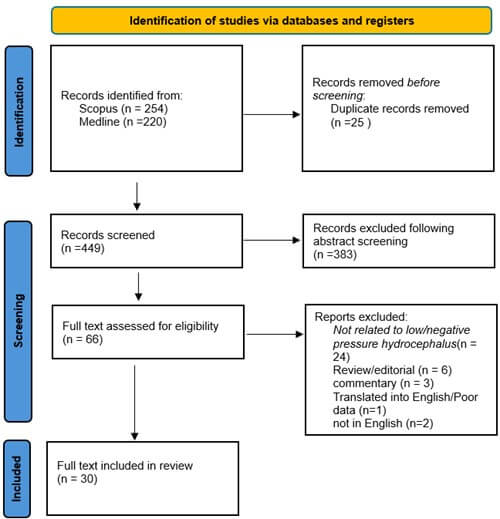
Database searching yielded 474 records (Scopus 254; MEDLINE 220). Following deduplication (25 records), 449 unique records were screened, and 383 were excluded at title/abstract. Sixty-six reports underwent full-text review; 36 were excluded with reasons (not related to low/negative-pressure hydrocephalus 24; review/editorial 6; commentary 3; translated into English/poor data 1; not in English 2). Thirty studies met inclusion criteria. “Records” denote database entries; “reports” denote full-text articles. Where multiple reasons applied, a single primary reason was assigned for counting. Prepared and reported using PRISMA 2020 guidance.
3.2. Study Characteristics
Of the 30 publications assessed, most were case reports (n=12) and case series (n=11), reflecting the rarity and clinical complexity of NegPH/LPH. The remainder included four retrospective case series, one consecutive series, and two retrospective cohort studies. The low number of studies speaks to the difficult nature of neurological studies. Study designs, sample sizes, and outcomes varied widely which limited direct comparison due to the heterogeneity (Table 1).
Table. 1. Distribution of study designs in the evidence base.
|
Type of study |
Number |
|
Case reports |
12 |
|
Case series |
11 |
|
Retrospective case series |
4 |
|
Retrospective cohort study |
2 |
|
Consecutive series |
1 |
The figure summarizes the design classification for all included studies (N = 30): case reports (12/30, 40%), case series (11/30, 36.7%), retrospective case series (4/30, 13.3%), retrospective cohort (2/30, 6.7%) and, consecutive series (1/30, 3.3%). Design labels follow the terminology reported by the original authors; where multiple characteristics were present, the primary design term in the report was used for categorization.
The publications reported on 215 cases, with traumatic brain injury (TBI) emerging as an antecedent, followed by aneurysmal subarachnoid haemorrhage (SAH) and NPH. A substantial portion of cases lacked a definitive preceding insult, highlighting the multifactorial nature of NegPH/LPH (Graph 1).
Treatment strategies varied widely, including sub-atmospheric and titrated external ventricular drainage (EVD), low-pressure valve systems, valveless shunts, blood patch applications, and endoscopic third ventriculostomy (ETV). Complex cases sometimes had multiple interventions. This heterogeneity reinforces the need for standardised treatment protocols.
Outcome reporting was also varied. Certain authors employed well-recognised metrics, such as the modified Rankin Scale, Glasgow Outcome Scale Extended, or Karnofsky Performance Status, many studies documented mortality at differing time points, normalisation of ICP, return to baseline function, or discharge destination. Others simply indicated that patients were “well” at follow-up. This lack of uniform outcome measures, combined with the high degree of methodological variation among studies, limits capacity for definitive conclusions. Study characteristics are summarised in (Table 2). No RCT or observational studies were identified in the included studies therefore Cochrane Risk of Bias Tool and Newcastle–Ottawa Scale could not be used, all studies included were appraised by 2 reviewers using JBI-SUMARI appraisal tools, results included in (Appendix & Tables 1-3).
Table. 2. Study characteristics and management strategies in low/negative-pressure
hydrocephalus (LPH/NegPH).
|
Study (author) |
Study
(Year) |
Country of
origin |
Research
method or design |
Number of
cases |
Purpose |
Associated
pathology |
method of
treatment |
Main
finding |
outcome |
|
Pang and Altschuler [2] |
1994 |
USA |
Case Series |
12 |
To report experiences
of LPH |
Multiple underlying
diagnosis |
Sub atmospheric EVD +
low pressure VP shunt |
LPH is related to
alteration of the viscoelastic modulus of the brain |
Recovered back to
baseline |
|
Vassilyadi
[27] |
1995 |
Canada |
Case Series |
2 |
To report experiences of LPH in spinal arachnoid
cystopleural shunting |
Multiple underlying diagnosis |
Removal of shunt |
Communication between the arachnoid cyst and the
subarachnoid space exist |
Both patients recovered |
|
Dias [28] |
1999 |
USA |
Case Series |
2 |
To report experience of
'low-pressure state' followed a lumbar puncture (LP) |
LP |
Blood patch |
Lumbar CSF leak can
result in LPH and VPS failure |
Complete recovery
without further surgery |
|
Owler [22] |
2001 |
Australia |
Case Series |
5 |
To report experiences of LPH |
Multiple underlying diagnosis |
Valveless or low pressure valve inserted |
Symptomatic hydrocephalus can occur in normal or low ICP states |
At 2 year follow up 4 showed marked improvement and 1 no change |
|
Clarke [29] |
2006 |
USA |
Case Series |
2 |
To report experiences of VLPH |
SAH,TBI |
Sub atmospheric EVD + low pressure VP
shunt |
VLPH may be a separate entity and
need further study to improve outcome |
Case 1 discharged to nursing home GCS
E4V1M5 Case 2 bedridden |
|
Filippidis
[11] |
2011 |
USA |
Case Series |
3 |
To report experiences of NegPH |
CSF leaks at craniotomy sites |
CSF leak repair + Sub atmospheric EVD + low pressure VP shunting +/-
ETV |
Clinical suspicion of NegPH or LPH should be high on repeated
"shunt failures" |
Varies outcome; case 1- discharged home with multiple re presentations
with shunt malfunction case 2 - death case 3- partial reversal of clinical
picture |
|
Akins [8] |
2011 |
USA |
Retrospective case series |
9 |
To report successful treatment of LPH |
Multiple underlying diagnosis |
Sub atmospheric EVD + VP/VPl shunt with or without valve |
Sub atmospheric drainage of CSF helps
improve clinical outcome in LPH patients |
MRS 3 or better in all patients |
|
Hamilton
[5] |
2012 |
Canada |
consecutive series |
20 |
To report successful treatment of LPH with ETV |
Multiple underlying diagnosis |
ETV |
ETV can reestablish communication with SAS leading to symptom
improvement |
ICP normalised in all patients in group 2 (with ETV) |
|
Kalani [23] |
2013 |
USA |
Case Series |
2 |
To describe the development of a new
negative pressure shunt system |
SAH and CSF leak |
Pumping negative pressure shunt
system |
Actively pumping CSF thought shunt
resulted in good outcome |
Patient 1; 12 month follow up showed
back to baseline with shunt working without pumping. Patient 2; shunt system
worked however died of fungal infection at 9 months post op |
|
Galbarriata
[26] |
2013 |
Spain |
Case Series |
4 |
To report experience of treating LPH with custom-made catheter |
Multiple underlying diagnosis |
Sub atmospheric EVD + Valveless VP shunt + custom-made peritoneal
catheter |
1.9 mm peritoneal catheter may be a good option in mx of LPH |
6 months-3 year follow up- 1 death (@ 6 months), 1 partially
dependent, 2 complete recovery |
|
Hunn [19] |
2014 |
Australia |
Case Series |
8 |
To report successful treatment of
NegPH |
Multiple underlying diagnosis |
Titrated EVD + low-pressure VP
shunting |
Early recognition and prompt
treatment associated with good outcome |
1 death at 48 days (no cause found);
7 alive at >1year (4 return to premorbid function 3had sequalae of primary
disease) |
|
Lin [25] |
2016 |
China |
Case report |
1 |
To describe the treatment of LPH following skull base surgery |
Skull base surgery-without CSF leak |
Sub atmospheric EVD + ETV |
Concurrent EVD at sub atmospheric drainage and ETV help clinical
improvement |
Recovered to normal ventricle size and "clear consciousness" |
|
Foster [24] |
2016 |
USA |
Cohort study |
16 |
To Describe the role of ETV in
treatment of LPH |
Multiple underlying diagnosis |
sub atmospheric EVD + ETV +/- Shunt |
ETV can be used successfully to
manage refractory LPH by opening CSAS to ventricles |
Those who underwent ETV 2 out of 6
avoided the need for VPS |
|
Olivero [7] |
2016 |
USA |
Case report |
1 |
To report on the role of MRE in diagnosis low brain stiffness in LPH |
Aquaductal stenosis |
Sub atmospheric EVD + low pressure VP shunt |
First direct observation of extremely low brain stiffness in NPH |
"Patient did well" |
|
Strand [10] |
2017 |
USA |
Case report |
1 |
Report a case of LPH and up-to-date
summary of the condition |
Post fossa tumour resection |
ETV + Shunt placement |
ETV + Shunt placement may play a role
in definitive management |
Well at 3 months follow up |
|
Smalley
[16] |
2017 |
USA |
Case Series |
29 |
To report experiences of LPH |
Multiple underlying diagnosis |
Multiple treatment methods |
Early diagnosis and continuous CSF drainage resulted in good outcomes |
All except 1 (death) made full recovery back to baseline |
|
Pandey [13] |
2017 |
China |
Case report |
1 |
To report experiences of NegPH |
Post fossa tumour resection |
Titrated EVD+ hyponatremia +
low-pressure VP shunting |
Titrated EVD drainage and possibly
hyponatremia helps in mx of NegPH |
Clinical improvement at 2 months
(able to ambulate independently) |
|
Michael
[30] |
2017 |
USA |
Case report |
1 |
To report experiences of LPH |
Aquaductal stenosis + chronic shunting |
Titrated EVD +valveless VP shunting |
Chronically shunted patients are at higher risk of LPH |
5 year follow up- ICP normalised, ongoing chronic headaches |
|
Cheng [17] |
2017 |
China |
Retrospective case series |
7 |
To report successful treatment of LPH |
Multiple underlying diagnosis |
Sub atmospheric EVD + low pressure VP
shunt +/- regular pumping of valve |
Sub atmospheric drainage via EVD, lop
pressure EVP and regular pumping of valve resulted in good outcomes |
At 12 months Mean GOS score was 4.57
and Karnofsky score was 84.28 |
|
Badran [14] |
2018 |
UK |
Case report |
1 |
To describe the treatment of LPH in this case |
Post fossa tumour resection |
Sub atmospheric EVD + Valveless VP shunt |
Negative pressure shunting needed for long term management |
Clinical and radiological improvement beyond 1 year follow up |
|
Houlden [18] |
2018 |
Canada |
Retrospective case series |
4 |
To report experiences of VLPH and
predictors of good outcome |
Multiple underlying diagnosis |
Titrated EVD drainage |
Early recognition and treatment of
CSF leak coupled with restoration of FHR results in better outcomes |
1 patient showed good long-term
outcome (discharged home) 2 deaths, 1 poor neurological outcome |
|
Diaz-Romero
[31] |
2019 |
Spain |
Case report |
1 |
To report experiences of NegPH |
Sylvian fissure arachnoid cyst shunting |
Multiple underlying diagnosis |
Transmental pressure gradient results in ventriculomegaly and NegPH |
Radiological and clinical improvement when shunt tied off |
|
Wu [6] |
2019 |
China |
Case Series |
39 |
To report on new hydrocephalus
classification based on ventricular pressure |
Multiple underlying diagnosis |
EVD placement + VPS placement when
pressure normalised |
NegPH diagnosis was associated with
higher rates of infection and lower GOSe |
8 deaths, 4 vegetative state, 15
sever disability |
|
Funnell [4] |
2020 |
UK |
Retrospective case series |
19 |
To describe an ultra-low-pressure state in NPH |
NPH |
Adjustable gravitational valve |
Further CSF drainage of a subgroup of NPH patients may lead to
improvement of symptoms |
58% showed improvement in walking 78% reported subjective improvement |
|
Vilela [15] |
2020 |
Brazil |
Case report |
1 |
To report experience of treating LPH
following LP with blood patch |
CSF leak following LP |
Blood patch |
Lumbar CSF leak can result in LPH and
VPS failure |
Rapid improvement to baseline at day
3 post blood patch |
|
Suzuki [20] |
2021 |
Japan |
Case report |
1 |
To report experience of treating NegPH with blood patch |
CSF leak at lumbar drain site |
Blood patch |
Identifying CSF leak point can help treat NegPH |
Clinical and radiological improvement following blood patch no long
term follow up |
|
Olivero [9] |
2021 |
USA |
Case report |
1 |
To describe that low brain stiffness
may be the cause of LPH |
Aquaductal stenosis + chronic
shunting |
Sub atmospheric EVD + low pressure VP
shunt |
Some evidence suggesting loss of
brain stiffness is likely the cause of LPH |
Brain stiffness normalised when
clinical and radiological symptoms improved |
|
Ozeki [32] |
2022 |
Japan |
Case report |
1 |
To report experiences of LPH post Lumbar drain in SAH patient |
Lumbar Drain |
Removal of lumbar drain |
LPH has good prognosis if diagnosed and treated early |
Discharged home 3 weeks post admission |
|
Czorlich [33] |
2023 |
Germany |
Retrospective cohort study |
15 |
To investigate aneurysmal SAH
associated acute low pressure hydrocephalus
|
Aneurysmal SAH |
EVD placement + VPS placement when
pressure normalised |
Forced CSF drainage is required for
favourable outcome |
7 day outcome showed decrease in
vents or increase in GCS with forced drainage |
|
Godoy
Hurtado [21] |
2023 |
Spain |
Case Series |
6 |
To report successful treatment of NegPH |
Multiple underlying diagnosis |
Sub atmospheric EVD + low pressure VP shunt |
Condition can develop in any context, large bore catheters and
negative pressure drainage resulted in good outcomes |
MRS 3 or better in 5 out of 6 patients |
Data are aggregated
from 30 studies (215 cases) published 1994–2023 across nine countries.
Treatments were grouped as sub-atmospheric external ventricular drainage (EVD),
ventriculoperitoneal/pleural shunting (VP/VPl; valveless or low-pressure
valves), endoscopic third ventriculostomy (ETV), epidural blood patch, and
other strategies. Outcomes are presented as reported by the original authors
(e.g., mRS, GOS/GOSE, discharge status); where percentages are shown,
denominators reflect non-missing data for that item.
EVD: External
Ventricular Drain; VP: Ventriculoperitoneal; VPl: Ventriculopleural; ETV: Endoscopic
Third Ventriculostomy; LP: Lumbar Puncture; SAH: Subarachnoid Haemorrhage; TBI:
Traumatic Brain Injury; mRS: Modified Rankin Scale; GOS/GOSE: Glasgow Outcome
Scale/Extended.
Source: Study-level dataset compiled for this review.
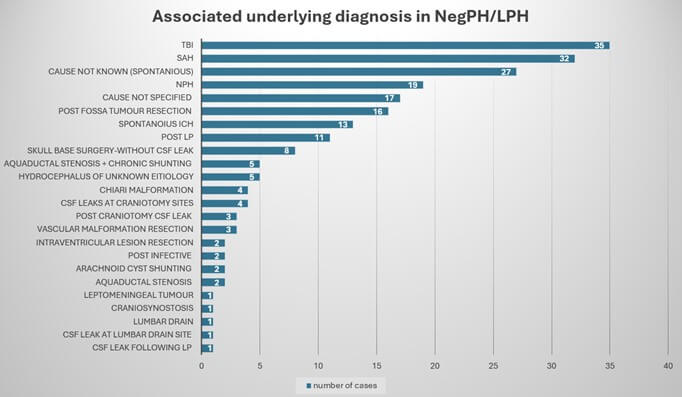
LPH: Low-Pressure Hydrocephalus; NegPH: Negative-Pressure Hydrocephalus; TBI: Traumatic Brain Injury; SAH: Subarachnoid Haemorrhage; ICH: Intracerebral Haemorrhage; NPH: Normal-Pressure Hydrocephalus; LP: Lumbar Puncture; CSF: Cerebrospinal Fluid; N: Total Number of Cases.
3.2.1. Associated Pathologies, Diagnostic Criteria and Advances in Diagnostic Accuracy/Predicting LPH/NegPH
Multiple theories have been proposed to explain the pathophysiology of NegPH/LPH. TBI and SAH were frequently identified, followed closely by NPH and cases classified as spontaneous NegPH/LPH. Several authors described instances of NegPH/LPH occurring after lumbar puncture (LP), lumbar drain placements, or surgical procedures such as posterior fossa craniotomies and skull base surgeries (endonasal or open approaches).
Clarke et al. [3] introduced the concept of very-low-pressure hydrocephalus (VLPH), describing cases requiring sub-atmospheric drainage (up to-40cm H2O). Despite intervention, outcomes were poor, suggesting VLPH may represent a refractory variant. Ultra-low-pressure states in NPH were described by Funnell et al. [4] and SILPASH by Hamilton et al. [5] These show the clinical variability and current poor understanding of low ICP states, suggesting individuality in each management approach. Pathophysiologically, Funnell et al. [4] did not observe a unique difference but did suggest a potential for impaired brain compliance.
Pang and Altschuler [2], proposed several core features of NegPH/LPH, including normal or low ICP in the context of ventriculomegaly, clinical deterioration with medium-pressure CSF diversion, and clinical plus radiological improvement following sub-atmospheric drainage. Wu et al. [47] expanded these concepts through additional imaging findings and aetiology such as TBI, SAH, infection, and posterior fossa surgery into a new classification based on ventricular pressure. They set a threshold of <70 mmH2O for low-pressure hydrocephalus in their proposed scheme (Figure 4), emphasising CT imaging in diagnosing secondary NegPH/LPH. Figures 2 & 3 from Wu et al. [6] illustrate both the updated diagnostic framework and the ventricular pressure-based classification of hydrocephalus.
Advancements in CT-usage as a diagnostic modality has improved, but an inability to quantify brain compliance or stiffness remains. Olivero et al. [7] successfully employed magnetic resonance elastography (MRE) to measure significantly low brain stiffness (1.62 kPa) in a patient with LPH, where a normal value is approximately 3.0 kPa. This stiffness did not improve despite clinical and radiological improvement, suggesting that low compliance may not be transient. This contrasts with earlier MRE studies of NPH, which reported conflicting data regarding brain stiffness. Olivero et al. [7] concluded that while LPH patients exhibit low stiffness it may not be antecedent to symptoms and is unimpacted by treatment. Their observations also imply that the mechanics of brain tissue may undergo permanent change in some cases of NegPH/LPH.


3.2.2. Hypothesis and Theories Surrounding Pathophysiology of NegPH/LPH
Multiple theories attempt to explain the pathophysiology of NegPH/LPH, but no model has yet accounted for all presentations. Instead, current literature suggests an interplay between four principal models that have emerged: the viscoelasticity theory, the transmantle pressure gradient theory, the poroelastic model of the brain (including the glymphatic system), and the pulsatile vector theory. At present, these frameworks should be viewed as theories which may work independently or in combination across different subtypes.
The viscoelasticity theory, initially from Pang and Altschuler [2], links increased brain compliance with ventriculomegaly in low or normal ICP ranges. They attribute this loss to histological changes such as gliosis, atrophy, and loss of structural proteins which may stem from cerebral insults (such as SAH or diffuse axonal injury (DAI)). These changes microscopically weaken brain recoil. A number of authors [7-10] employed MRE to measure parenchymal stiffness, revealing reductions in brain stiffness of up to 46% compared to healthy populations. This reduction could be causative for ventriculomegaly with even minor shifts in CSF pressure. However, no certain risk profile is apparent. Olivero et al. [9] observed decreased parenchymal stiffness beyond clinical and radiological resolution of symptoms, raising the question of irreversible structural changes. It is unclear if these changes evolve with time. Targeted interventions to restore compliance may be a therapeutic avenue.
The transmantle pressure gradient theory suggests disrupted CSF flow between ventricles and the cortical subarachnoid spaces (CSAS) creates a differential pressure across brain parenchyma, which drives ventriculomegaly despite a low ICP. Filippidis [11] and others post-LP, base of skull surgery, and trauma changes to CSAS pressures from assumed blockage or scarring, trapping CSF intraventricularly. This establishes a pressure gradient, with the ventricles becoming the higher-pressure compartment. Even if the measured ICP remains within normal or mildly reduced ranges, this differential can be sufficient to drive ventricular enlargement. Clinical improvement post intervention, such as ETV, supports this theory. Measuring regional ICP differences remains technically difficult, complicating further confirmation. In certain studies, evidence of transependymal CSF movement remains despite blockages, suggesting partial communication may remain. Also, the model fails to explain differences in outcomes across similar aetiologies, and suggests that brain compliance or other overlapping mechanisms likely influence disease expression.
The poroelastic model depicts brain tissue as composite of both solid and fluid phases with various porosity, permitting exchange under differing physiological or pathological conditions. Initially proposed from Hakim [12], that the brain resembles an “open-cell sponge” and expanded by Pandey [13] and others suggesting approximately 80% is fluid and 20% is cellular matrix In NegPH/LPH, this balance may be disrupted, leading to a “low fluid state” that increases brain compliance. Other studies report fluid accumulation rather than depletion, possibly due to inflammation or altered absorption. Badran [14] and Atkin [8] observed inflammatory processes in patients with haemorrhagic or traumatic injuries that may influence local fluid dynamics. The poroelastic model suggests the brain’s mechanical response depends not only on the integrity of its cellular matrix but also on fluid exchange across intra-, extra-, and intraventricular compartments. The glymphatic system, a periarterial conduit that facilitates CSF flow and waste clearance, is theorised to be a key factor in this. Haemorrhage or inflammation may impair this system, reducing parenchymal fluid transport and viscoelasticity, leading to increased susceptibility to low-pressure hydrocephalus. Although nearly half of adult LPH cases involve prior haemorrhage, direct evidence linking glymphatic dysfunction to NegPH/LPH remains limited. Further research could be aimed at establishing predictive imaging or molecular markers of these processes, assisting early detection.
The pulsatile vector theory attributes cerebrospinal fluid movement to cyclical arterial and venous pulsations. Under normal conditions three directional forces act together to distribute cerebrospinal fluid throughout the cranial vault. These forces are centripetal interstitial shock waves, centrifugal brain expansion and fronto-occipital subarachnoid shock waves. When cerebral perfusion is reduced, as in severe illness or after surgery, these forces weaken, and ventricular cerebrospinal fluid outflow is impaired. In chronically shunted patients, who often have low intracranial pressure due to continuous drainage, the reduced pulsatile waveform may fail to open shunt valves, allowing ventricles to enlarge despite low pressure. Case reports and small series describe a mismatch between cerebrospinal fluid production, absorption and pulsatile flow that supports this theory. However, it does not explain why some cases of NegPH/LPH resolve spontaneously while others remain resistant to treatment, nor why many chronically shunted patients never develop the condition. Additional factors such as local inflammation, baseline brain compliance or partial compartmental obstructions may influence these pulsatile dynamics.
The range of theories posited highlight the multifactorial and unknown nature of NegPH/LPH’s pathophysiology. Each theory, including brain compliance, altered pressure gradients, poroelastic fluid exchange, and impaired pulsatile flow, all reflects features seen in clinical, radiological, and pathological studies. None, however, explain the diversity of causes (TBI, SAH, Iatrogenic complications) nor the variation in disease course and treatment response. This lends itself to potential multifactorial causes, or overlap in current theories, for example: a haemorrhagic event could disrupt the glymphatic and subarachnoid pathways, elevating compliance and creating a transmantle gradient, producing a persistent NegPH/LPH state.
The scarcity of robust data is a challenge in understanding NegPH/LPH. Much of current literature consists of case reports, small series, or retrospective analyses, leaving gaps in the epidemiology and pathophysiology. However, the rarity and often acute presentations make large RCT’s or prospective studies difficult. Even-so, significant hurdles persist in measuring dynamic parameters such as compartment-specific ICP, precise brain compliance, and real-time glymphatic flow. While techniques such as MRE and functional MRI may offer valuable insight into compliance, fluid dynamics, and inflammation, they remain limited in availability due to equipment demands, time constraint, and patient instability.
Given these limitations, the need for collaboration across centres to capture data and standardise outcome measures is paramount. Without consistent definitions, a shared data dictionary, and agreed end points, pooling cohorts remains difficult and important signals remain unstudied. A coordinated registry that records baseline anatomy, physiological measures, imaging, and treatment sequences would add statistical power and external validity. Future studies should explore how inflammatory profiles, endothelial function, and vascular variants may influence the risk of developing NegPH/LPH. Mapping of the condition, using serial imaging and bedside measurements, could reveal windows in which specific therapies are most effective.
Current hypotheses suggest that NegPH/LPH arise from interacting disturbances in brain mechanics and circulation. The viscoelasticity model, the transmantle pressure gradient model, the poroelastic and glymphatic framework, and the pulsatile vector concept each explain part of the syndrome. The task is to integrate these views into a framework that accounts for variation in presentation, prognosis, and response to treatment. Meaningful progress will require advanced imaging, precise physiological measurement, and multi-centre data pooling to support evidence-based diagnosis, management, and prevention.
3.2.3. Treatment Approaches and Outcomes
Management of NegPH/LPH aims to restore brain compliance and thereby improve neurological function. Clinical recovery often follows reduction of ventriculomegaly using conservative measures, subatmospheric external drainage, repair of cerebrospinal fluid leaks, or permanent diversion; failure to normalise intracranial pressure or persistent ventricular dilatation correlates with poorer outcomes. The need for multiple modalities reflects multifactorial aetiology.
Filippidis and colleagues [11] describe four pathways by which impaired compliance produces deficits: parenchymal distortion, reduced cerebral perfusion, incipient herniation from expanding ventricles, and pressure differentials between ventricles and cortical subarachnoid space. These mechanisms map to motor, cognitive, and behavioural change. Enlargement of the frontal horns can affect corticospinal pathways with gait disturbance and weakness; paediatric diffusion tensor imaging shows partial reversal after restoration of drainage. Expansion around the third ventricle is linked to memory impairment. Raised radial pressure on cortical surfaces can restrict regional blood flow and drive encephalopathy, from confusion to psychomotor slowing and reduced consciousness. Marked pressure gradients may generate acquired tonsillar descent, mimicking a secondary Chiari picture. The breadth of deficits underscores the urgency of restoring compliance.
Leak repair is a low-risk first step, particularly after cranial or spinal procedures. Reports by Vilela [15] and Smalley [16] document reversal of NegPH/LPH after sealing iatrogenic leaks; epidural blood patch is commonly used. Smalley’s [16] series of eleven post LP cases, two recovered with a single patch by six months; four also received patches with variable outcomes; three improved conservatively. Filippidis [11] notes mixed postoperative trajectories and transient benefit from neck wrapping to augment turgor. Case level variability, comorbidity, and inconsistent outcome measures produce divergent success rates, indicating that leak closure may be necessary yet insufficient when compliance remains abnormal.
External ventricular drainage is the main acute intervention, but NegPH/LPH typically requires subatmospheric settings to drive cerebrospinal fluid outflow, reduce ventricular size, and allow parenchymal re-expansion. Across Atkin [8], Badran [14], Cheng [17], Hamilton [5], Houlden [18], Hunn [19], Pandey [13] and Suzuki [20] protocols vary in titration and monitoring. Hunn [19] targeted 120 to 360 ml per day in eight patients, with discharge Glasgow Outcome Scale scores from 1 to 4 and seven survivors at follow up beyond one month to more than four years: four resumed work or prior activities, supporting carefully moderated negative pressure as a means to restore compliance. Houlden [18] associated sustained reduction in the frontal horn ratio with stabilisation. Atkin [8] raised the drainage threshold stepwise by 3 cm H2O every 3 to 5 days once symptoms and imaging improved, then proceeded to shunting: in nine patients most achieved modified Rankin Scale scores 0 to 3 at six months to three years. These approaches are resource intensive, often require repeated imaging, and show inter patient variability; larger cohorts and continuous intracranial pressure telemetry could refine increments and reduce radiation exposure.
Post stability, many patients receive shunts, most commonly ventriculo-peritoneal, with ventriculo-pleural diversion used selectively. Configurations include adjustable valves (Houlden [18], Hunn [19], Wu [6]), low opening pressure or gravity assisted systems used by Godoy Hurtado [21] (Miethke ProSA, M Blue, proGAV), and valveless systems in series such as Owler’s [22]. The goal is to accommodate low operating pressures without overdrainage. Conventional valves may not open at NegPH/LPH pressures, whereas very low thresholds increase siphoning and subdural risk. Outcomes are heterogeneous; complications include infection and mechanical failure. Godoy Hurtado [21] reported mixed performance of gravity assisted valves requiring reprogramming or replacement in some cases. Kalani [23] proposed an in line dual reservoir pump to maintain active negative pressure; one patient weaned off pumping after a year, whereas another died from a fungal infection, underscoring vulnerability to device related morbidity. In the absence of randomised trials or uniform endpoints, device choice remains individualised, shaped by anatomy, baseline compliance, and centre expertise.
Where repeated subatmospheric drainage or shunting proves suboptimal, endoscopic third ventriculostomy can re-establish communication in suspected compartmental or absorptive block. Series by Foster [24], Hamilton [5], Lin [25] and Strand [10] suggest that ETV can reduce shunt dependence; Hamilton [5] reported long term shunts in about 20 percent after ETV versus about 70 percent without ETV, although selection bias likely contributes. Lin [25] used stabilisation with subatmospheric drainage before ETV to facilitate controlled drain removal. Diffuse scarring of basal cisterns or poor cisternal capacity predicts failure. Advanced MRI methods that assess local flow near the third ventricular floor and post ETV compliance could improve candidate selection.
Not all patients recover despite physiological correction. Irreversible white matter injury after prolonged hypoperfusion or repeated haemorrhage, advanced age, comorbidity, or limited tolerance for surgery may cap recovery. Early recognition and swift treatment shorten exposure to abnormal compliance and likely improve trajectory. Future markers of tissue viability, such as metabolic imaging or high-resolution intracranial pressure waveform analysis during staged drainage, could guide escalation and avoid futile intervention.
Multi-centre registries that capture demographics, aetiology, imaging, physiological metrics, interventions, and longitudinal outcomes are needed. Standardised endpoints would enable comparison of rapid versus slow weaning of negative pressure, adjustable versus fixed or valveless systems, and selection criteria for ETV. Prospective stratification by aetiology should clarify which subgroups respond best to leak repair, drainage strategies, ETV, or specific shunt configurations. Harmonised datasets would support meta-analysis of survival, functional scales such as modified Rankin Scale and Glasgow Outcome Scale, complications, reoperation, and device failure.
Across reports, strategies from leak repair to subatmospheric drainage, ETV, and tailored shunting can reverse ventriculomegaly and often improve function in NegPH/LPH. Heterogeneous physiology and patient factors explain variable efficacy. Robust, standardised, collaborative studies are required to define durable, safe pathways for each NegPH/LPH subtype.
4. Discussion
This discussion synthesises a heterogeneous evidence base to organise the literature on NegPH/LPH into five recurrent themes; features, difficulties in diagnosis, theories, variation in treatment, and early intervention. Clinically, a recognisable phenotype emerges: ventriculomegaly with normal or low ICP, deterioration with standard-pressure diversion, and improvement with subatmospheric drainage [2]. Diagnostic uncertainty persists given debated pressure thresholds (e.g., <70 mm H2O) and overlap with other hydrocephalus variants, complicating timely recognition [2, 6]. Pathophysiologically, impaired brain compliance remains central, with early elastography data showing reduced stiffness even after apparent radiological recovery [7]. Therapeutically, approaches span targeted dural repair/epidural blood patch for CSF leaks [11, 16], subatmospheric EVD with protocol variability [13, 17, 19], programmable or tailored shunt strategies [18, 21, 22, 26], and selective ETV (alone or combined with EVD) when flow obstruction predominates [5, 13, 25]. Across these domains, the weight of current evidence underscores the importance of prompt intervention to limit secondary injury and consolidate recovery [7, 13, 16, 17, 19]. Framed by these themes, the subsequent sections integrate findings, delineate gaps, and propose priorities for standardised diagnostics, compliance measurement, and outcome reporting.
4.1. Features of NegPH/LPH
This review highlights the danger and variability of NegPH/LPH and the complexity in its diagnosis, management and multiple aetiologies, including: TBI, SAH, postoperative complications, chronic shunt dependence, and idiopathic presentations. Untreated or poorly managed NegPH/LPH leads to increased morbidity and mortality. Diverse pathogenesis means most neurosurgeons or neurointensivists will encounter it. A history of TBI, SAH, or neurosurgery increases risk of NegPH/LPH, which may masquerade as persistent shunt malfunction, NPH with paradoxical features, or an intractable headache syndrome, delaying diagnosis. Greater awareness is essential for effective care and rapid diagnosis.
Pang and Altschuler [2] provided core features for diagnosing NegPH/LPH: ventriculomegaly coexisting with normal or low ICP, clinical deterioration with medium or standard-pressure CSF diversion, and improvement upon subatmospheric drainage. This separates NegPH/LPH from hydrocephalus which classically has elevated ICP. However, these broad criteria leave room for clinical variants. NegPH/LPH overlaps with other hydrocephalus variants, including NPH and slit-ventricle syndrome. Misdiagnosis leads to mismanagement, including increased shunt valve pressure when negative-pressure drainage is indicated. Diagnosis often relies on imaging findings, CSF pressure managements, and the individual clinical picture.
The potential for permanent sequelae promotes early detection and treatment. The systematic review highlights how protracted brain distortion or compromised perfusion can lead to chronic white matter injury. NegPH/LPH requires individual tailored management suggesting it as a diagnostic entity rather than variant.
4.2. Difficulties in Diagnosis
NegPH/LPH’s prevalence is unclear due to absence of large-scale epidemiological studies. Existing literature consists of 12 case reports, 9 case series, and a handful retrospective studies or small cohort analyses, hindering the ability to draw conclusions. Ethical and logistical barriers involved with complex neurological disease further obstruct large scale study. Wu [6] posited <70 mm H2O as “low-pressure.” This criterion may help create standardisation, the practicality of measuring continuous ICP remains questionable. Wu [2] attempts to incorporate relevant risk-factors, but a significant portion present without a preceding event. This ambiguity suggests refined classification systems can struggle to capture complex or comorbid presentations.
Evidence shows brain compliance and CSF disruptions as core to pathogenesis, but no theory explains the unique low and negative pressure states. This discussion synthesises findings, implications, knowledge gaps, and proposes further research, and addresses current treatment options.
4.3. Theories
Altered brain compliance is regarded as the foundation of NegPH/LPH pathophysiology. MRE allows quantification of compliance, but with limited feasibility in acute or critical populations and resource-limited settings. Olivero [7] documented reduced stiffness in a patient with LPH even post clinical and radiological improvement. Inferring potential future decompressions are a risk for patients with refractory stiffness. MRE’s low accessibility leaves NegPH/LPH patients’ compliance unassessed, with existing studies being case reports or series. Perhaps future, more-accessible MRE methods could facilitate routine stiffness measurement, assisting prognosis and management.
The review highlights four principal models of NegPH/LPH, each highlighting different contributors to persistent low or negative ICP:
Viscoelasticity Theory: Chronic/repeated insults cause increased compliance, expanding with minimal pressure.
Transmantle Pressure Gradient Theory: Malfunctioning CSF pathways establish differential pressures between ventricles and CSAS, driving ventriculomegaly.
Poroelastic Model of the Brain: The brain is a multi-phase medium with dynamic fluid shifts. Local factors alter fluid absorption/release, increasing compliance.
Pulsatile Vector Theory: CSF distribution relies on arterial and venous pulsatility. Altered pulsation permits ventricular enlargement under subnormal ICP.
These models establish NegPH/LPH as not uniform but of diverse pathophysiology. A patient recovering from severe TBI, for instance, may have a distinct pattern of microvascular damage, whereas LPH post-LP exhibits changes in CSF pressure dynamics. A single pathophysiological framework may prove incomplete for guiding universal treatment. The glymphatic system, though under-researched, might link local parenchymal changes and global CSF flow. This periarterial clearance network is sensitive to haemorrhage or inflammatory products, potentially impacting brain stiffness and ICP regulation. Direct clinical evidence, along with the system, is under-researched. Future research including imaging or molecular markers which assess system function, potentially a link to NegPH/LPH; if so, the glymphatic system may be a therapeutic target.
4.4. Variation in Treatment
Epidural blood patches or direct dural repairs offer non-invasive approaches that can be curative. Smalley [16] observed patients, specifically with LP-induced leaks, having total recovery post patch. Success rates vary, reflecting patient comorbidities, chronicity of leaks, or location and size of the dural defects. Filippidis [11] noted partial improvement, suggesting increased compliance and ischaemic injury may cause refractory symptoms. These studies reaffirm early detection and management. Future research may investigate treatment delay and impacts on outcome, potentially tracking ventriculomegaly and brain compliance pre/post-intervention with imaging.
Successful repairs herald significant quality-of-life gains and avoiding invasive measures. Given the low morbidity of an epidural blood patch compared to long-term EVD, standardised protocols for managing suspected leaks (including early imaging) could improve patient outcomes. Subatmospheric EVD is an acute intervention for rapid deterioration. Cheng [17], Hunn [19], Pandey [13], and others have shown a drainage threshold below the zero reference can “pull” ventricles back. When individualised, EVD has shown improvements to functional status and consciousness in patients.
Subatmospheric drainage protocols vary. Some recommend daily outputs of 120-360 mL, others prefer weaning of drainage threshold over days or weeks. Pulling too forcefully on compromised brain can over drain, precipitating subdural haematomas or exacerbate cortical collapse. Prospective studies could identify “optimal” negative pressure for each aetiological subtype, and the benefits of intermittent negative drainage. Telemetry and imaging use may refine protocols and limit excessive interventions as well.
Following ICP stabilisation via EVD, many authors place a permanent shunt system, commonly VP, and rarely VPL. Houlden [18] and Hunn [19] used medtronic strata II valves, adjusting them to 0.5 or 1, while Godoy Hurtado [21] preferred the Codman Certas or Miethke systems (ProSA, M-Blue, proGAV). These configurations showed success in maintaining ventricular volume and compliance post-EVD, reducing risk of recurrence. Some cohorts reported recurrent ventriculomegaly, requiring valve adjustments or additional procedures, each procedure increasing risk. Owler [22] tried valveless systems with mixed outcomes, while Galbarriatu [26] explored custom combinations, like Bactiseal peritoneal catheters and silver-coated ventricular tubing. Despite promising outcomes, the lack of randomised trials and large-scale data limits application. Future research may investigate dynamic/adjustable valves, or biodegradable sent-like devices which sustain low-pressure drainage.
ETV as an alternative when CSF flow is blocked at the aqueduct, fourth ventricle outlets, or basal cisterns, through a stoma in the third ventricle floor permitting CSF flow without permanent shunting. Hamilton [5] reported lower rates post ETV treatment, while Lin [25] reported EVD combined with ETV yields rapid improvements. ETV remains limited by anatomy, scarring, and hampered reabsorption. Risk remains for stoma closure requiring stenting. Future research could evaluate morphological and physiological markers (basal cistern patency on MRI, subarachnoid scarring) which predict successful ETV treatment. Some authors used validated neurological scales (MRS, Karnofsky Performance Status, or GOS), others used qualitative descriptors (“patient did well,” “partial improvement,” “good recovery”). This variability, along with methodological heterogeneity, limits cross-comparison and meta-analysis. A standardised framework of outcome assessment might include:
• Functional independence measures (mRS, GOS, or Barthel Index).
• Cognitive evaluations (where feasible).
• Radiological indices (Evans’ index, frontal horn ratio, or volumetric analyses).
• Quality of life metrics (EQ-5D).
Quality-of-life outcomes are critical: while ICP normalisation is important, the goal should be restoring patient cognitive and physical baseline. A robust, validated tool could assist measuring recovery beyond radiologically. Robust biomarkers are needed to distinguish NegPH/LPH from other forms of secondary hydrocephalus with atypical ICP. Future research may identify imaging and molecular markers which could guide diagnosis. These might assist the individual application of Subatmospheric drainage or identify care escalation criteria. Certain signs may also be revealed which indicate severe compromise, suggesting palliation or rehabilitative care. Effective NegPH/LPH treatment requires individual tailoring. Future research may refine decision-trees based on compliance states, anatomical blockages, or iatrogenic leaks. Larger prospective cohort studies could randomise or stratify patients to different weaning, valve, or ETV protocols, enabling quantitative assessments of results. These could also identify certain subgroups which would benefit from earlier advanced interventions (ETV, negative-pressure shunting) rather than conventional approaches.
Managing NegPH/LPH requires multidisciplinary input from neurosurgeons, neuroradiologists, and neuropsychologists. Collaboration reduces fragmentation and facilitates multicentre registries to standardise imaging, outcomes, and definitions on remission and relapse. This facilitates robust meta-analyses and comparison of interventions (e.g EVD vs ETV-&-EVD). Such networks foster the identification of therapeutic targets; if inflammatory or aquaporin-related pathways prove pivotal, then testing targeted pharmacological adjuncts could follow. Multi-centre expertise would only support the creation of evidence-based guidelines.
4.5. Early Intervention
Early intervention is paramount, as prolonged deformation may translate into permanent deficits. Key questions remain: Is recovery from supportive care possible? When does low ICP become irreversibly harmful? Prospective studies with patients at defined intervals post-insults could provide insight. Prophylactic measures, including blood patches, may also delay the transition to NegPH/LPH in early signs of impaired compliance.
4.6. Non-Surgical Management Options
Beyond mechanical diversion, several conservative or pharmacological adjuncts may be beneficial in selected NegPH/LPH phenotypes. Leak-directed strategies—including epidural blood patch and targeted dural repair—can reverse ventriculomegaly when iatrogenic spinal or cranial CSF loss is causal [11, 15, 42]. Temporary external compression (e.g., neck wrapping) has been used to augment cranial turgor with short-lived benefit in postoperative cases [11]. Limited case evidence also suggests that alterations in osmotic/volume status may modulate symptoms (e.g., improvement coinciding with hyponatraemia), but intentional induction cannot be recommended; such observations underscore physiological complexity rather than a reproducible therapy [13]. In selected patients who transition to a positive-pressure hydrocephalus phenotype after successful leak repair and restoration of CSF pathways, a trial of acetazolamide (carbonic anhydrase inhibition) may be considered as an adjunct to reduce CSF formation and mitigate over-accumulation, recognising that evidence in NegPH/LPH remains limited and such use should be individualised and closely monitored. Overall, non-surgical measures should be instituted early when a leak is suspected, while recognising that no pharmacological agent has demonstrated durable efficacy; at present these approaches are adjuncts to definitive leak repair and/or carefully titrated CSF drainage.
5. Conclusion
NegPH/LPH remains a diagnostic and therapeutic challenge. Historically overshadowed by more classic forms of hydrocephalus, NegPH/LPH carries comparable morbidity, risk of misdiagnosis, and management difficulty. The review highlights potential triggers and current pathophysiological theories. It shows the broad range of interventions and creative attempts by clinicians to normalise brain compliance in various clinical contexts.
A need for methodological consistency and larger datasets is apparent in the review. Measuring outcomes with validated tools and multi-centre studies would clarify effective management, functionally and radiologically. Given the interactions between local tissue changes, fluid dynamics, and secondary injuries, management must be tailored and timely. The review highlights a foundation which randomised, prospective, and systematic trials can build upon to guide evidence-based care.
Future direction includes diagnostics, treatment pathways, and identifying subtypes and respective best management. Larger datasets would identify demographics and at-risk patient profiles, as well as guiding prognosis. Deeper pathophysiological understanding may also provide novel therapy beyond surgical solutions. With further understanding, the neurosurgical field could devise effective, evidence-based guidelines for NegPH/LPH, reducing its morbidity and improving patient outcomes for a global, complex issue.
Limitations
This systematic review is limited by small sample sizes and low-quality evidence that NegPH/LPH literature consists of. Publication bias sways study themes and heterogeneity of outcomes, unvalidated definitions of ‘improvement,’ and incomplete follow-up all make conclusions difficult. Reliance on case reports/series limit generalisability. These often highlight abnormal presentations, lacking the systematic rigour to control for confounders and properly assess intervention effects. This reinforces the need for prospective, studies with standardised protocols. The review could not analyse demographic data or institutional differences in diagnostic criteria. Future studies must adopt consistent definitions, including for ICP states and ventriculomegaly, to lay the foundation for cross-study comparisons and multi-centre cooperation.
Conflicts of Interest
None.
Funding
None.
Acknowledgements
I would like to acknowledge the continuing support and guidance of Mr. Stephen Honeybul, Neurosurgeon, Statewide Neurosurgical Service, Western Australia. His expert opinion, critical review, and constructive feedback were invaluable to the development of this work and to my professional growth.
Ethical Approval Statement
No ethical approval was required as this study was based on previously published data.
Data Availability Statement
All data underlying this article are available in the published literature cited within this manuscript.
REFERENCES
[1] S
Hakim, R D Adams “The special clinical problem of symptomatic hydrocephalus
with normal cerebrospinal fluid pressure. Observations on cerebrospinal fluid
hydrodynamics.” J Neurol Sci, vol. 2, no. 4, pp. 307-327, 1965. View at:
Publisher Site | PubMed
[2] D
Pang, E Altschuler “Low-Pressure Hydrocephalic State and Viscoelastic
Alterations in the Brain.” Neurosurgery, vol. 35, no. 4, pp. 643-656,
1994. View at: Publisher Site | PubMed
[3] Juan
Casado Pellejero, Jesús Moles Herbera, Silvia Vázquez Sufuentes, et al. “Acute
negative-pressure hydrocephalus: Management algorithm and value of early
endoscopic ventriculostomy.” Neurocirugia, vol. 33, no. 1, pp. 1-8,
2022. View at: Publisher Site
[4] Jonathan
Perry Funnell, Linda D'Antona, Claudia Louise Craven, et al.
“Ultra-low-pressure hydrocephalic state in NPH: benefits of therapeutic
siphoning with adjustable antigravity valves.” Acta Neurochir (Wien),
vol. 162, no. 12, pp. 2967-2974, 2020. View at: Publisher Site | PubMed
[5] Mark
G Hamilton, Angel V Price “Syndrome of inappropriately low-pressure acute
hydrocephalus (SILPAH).” Acta Neurochir Suppl, vol. 113, pp. 155-159,
2012. View at: Publisher Site | PubMed
[6] Xuehai
Wu, Di Zang, Xing Wu, et al. “Diagnosis and Management for Secondary Low- or
Negative-Pressure Hydrocephalus and a New Hydrocephalus Classification Based on
Ventricular Pressure.” World Neurosurg, vol. 124, pp. e510-e516, 2019.
View at: Publisher Site | PubMed
[7] William
C Olivero, Tracey Wszalek, Huan Wang, et al. “Magnetic Resonance Elastography
Demonstrating Low Brain Stiffness in a Patient with Low-Pressure Hydrocephalus:
Case Report.” Pediatr Neurosurg, vol. 51, no. 5, pp. 257-262, 2016. View
at: Publisher
Site | PubMed
[8] Paul
T Akins, Kern H Guppy, Yekaterina V Axelrod, et al. “The genesis of low
pressure hydrocephalus.” Neurocritical Care, vol. 15, no. 3, pp.
461-468, 2011. View at: Publisher Site | PubMed
[9] William
C Olivero, Arundhati Biswas, Tracey M Wszalek, et al. “Brain stiffness
following recovery in a patient with an episode of low-pressure hydrocephalus:
case report.” Child’s Nervous System, vol. 37, no. 8, pp. 2695-2698,
2021. View at: Publisher Site | PubMed
[10] Adam
Strand, Stephen Balise, Lawrence Jun Leung, et al. “Low-Pressure Hydrocephalus:
A Case Report and Review of the Literature.” World Neurosurg, vol. 109,
pp. e131-e135, 2018. View at: Publisher Site | PubMed
[11] Aristotelis
S Filippidis, M Yashar S Kalani, Peter Nakaji, et al. “Negative-pressure and
low-pressure hydrocephalus: the role of cerebrospinal fluid leaks resulting
from surgical approaches to the cranial base.” J Neurosurg, vol. 115,
no. 5, pp. 1031-1037, 2011. View at: Publisher Site | PubMed
[12] S
Hakim “Biomechanics of hydrocephalus.” Acta Neurol Latinoam, vol. Suppl
1, pp. 169-194, 1971. View at: PubMed
[13] Sajan
Pandey, Yi Jin, Liang Gao, et al. “Negative-Pressure Hydrocephalus: A Case
Report on Successful Treatment Under Intracranial Pressure Monitoring with
Bilateral Ventriculoperitoneal Shunts.” World Neurosurg, vol. 99, pp.
812.e7-812.e12, 2017. View at: Publisher Site | PubMed
[14] Abdul
Badran, Matthew J Shepard, Alexander Ksendzovsky, et al.
“Hemangioblastomatosis-associated negative-pressure hydrocephalus managed with
improvised shunt.” J Clin Neurosci, vol. 58, pp. 226-228, 2018. View at:
Publisher Site | PubMed
[15] Marcelo
D Vilela, Hugo As Pedrosa, Braulio F Mesquita, et al. “Low-pressure
hydrocephalus and shunt malfunction following a lumbar puncture in an adult
reversed by an epidural blood patch.” World Neurosug, vol. 143, pp.
56-61, 2020. View at: Publisher Site | PubMed
[16] Zachary
S Smalley, Garrett T Venable, Stephanie Einhaus, et al. “Low-pressure
Hydrocephalus in Children: A Case Series and Review of the Literature.” Neurosurgery,
vol. 80, no. 3, pp. 439-447, 2017. View at: Publisher
Site | PubMed
[17] Zhe
Cheng, Wenhua Wang, Yong Han, et al. “Low pressure hydrocephalus: clinical
manifestations, radiological characteristics, and treatment.” Br J Neurosurg,
vol. 31, no. 4, pp. 410-414, 2017. View at: Publisher Site | PubMed
[18] Danielle
Houlden, Dmitriy Khodorskiy, Sandra Miller-Portman, et al. “Importance of
Frontal Horn Ratio and Optimal CSF Drainage in the Treatment of Very
Low-Pressure Hydrocephalus.” J Neurol Surg B, vol. 79, no. 4, pp.
379-385, 2018. View at: Publisher Site | PubMed
[19] Benjamin
H M Hunn, Asim Mujic, Idrees Sher, et al. “Successful treatment of negative
pressure hydrocephalus using timely titrated external ventricular drainage: a
case series.” Clin Neurol Neurosurg, vol. 116, pp. 67-71, 2014. View at:
Publisher Site | PubMed
[20] Tomoya
Suzuki, Shogo Kaku, Kostadin Karagiozov, et al. “Negative-pressure
hydrocephalus in the course of a complex postoperative intracranial pressure
disturbance: illustrative case.” J Neurosurg Case Lessons, vol. 1, no.
1, pp. CASE206, 2021. View at: Publisher Site | PubMed
[21] Alicia
Godoy Hurtado, Patrick Barstchi, Juan Francisco Brea Salvago, et al. “Low- and
Negative-Pressure Hydrocephalus: New Report of Six Cases and Literature
Review.” J Clin Med, vol. 12, no. 12, pp. 4112, 2023. View at: Publisher
Site | PubMed
[22] B
K Owler, E E Jacobson, I H Johnston “Low pressure hydrocephalus: issues of
diagnosis and treatment in five cases.” Br J Neurosurg, vol. 15, no. 4,
pp. 353-359, 2001. View at: Publisher Site | PubMed
[23] M
Yashar S Kalani, Jay D Turner, Peter Nakaji “Treatment of refractory
low-pressure hydrocephalus with an active pumping negative-pressure shunt
system.” J Clin Neurosci, vol. 20, no. 3, pp. 462-466, 2013. View at: Publisher Site | PubMed
[24] Kimberly
A Foster, Christopher P Deibert, Phillip A Choi, et al. “Endoscopic third
ventriculostomy as adjunctive therapy in the treatment of low-pressure
hydrocephalus in adults.” Surg Neurol Int, vol. 7, pp. 26, 2016. View
at: Publisher Site | PubMed
[25] Bon-Jour
Lin, Hsin-I Ma “A Rare Case of Low-pressure Hydrocephalus After Skull Base
Surgery.” Neurosurgery Quarterly, vol. 26, no. 3, pp. 256-258, 2016.
View at: Publisher Site
[26] L
Galbarriatu, M Rivero-Garvía, M Olivares, et al. “Low-pressure hydrocephalus:
indication for custom-made catheters? Technical report.” Acta
Neurochirurgica, vol. 155, no. 10, pp. 1981-1985, 2013. View at: Publisher Site | PubMed
[27] M
Vassilyadi, J P Farmer, J L Montes “Negative-pressure hydrocephalus.” J
Neurosurg, vol. 83, no. 3, pp. 486-490, 1995. View at: Publisher Site | PubMed
[28] M
S Dias, V Li, J Pollina “Low-pressure shunt “malfunction” following lumbar
puncture in children with shunted obstructive hydrocephalus.” Pediatr
Neurosurg, vol. 30, no. 3, pp. 146-150, 1999. View at: Publisher
Site | PubMed
[29] Michelle
J Clarke, Cormac O Maher, Georgia Nothdurft, et al. “Very low pressure
hydrocephalus. Report of two cases.” J Neurosurg, vol. 105, no. 3, pp.
475-478, 2006. View at: Publisher Site | PubMed
[30] Alex
P Michael, Ahmad Elkouzi, Rodger J Elble “Pearls & Oy-sters: Low-pressure
hydrocephalus and inadequate shunting.” Neurology, vol. 88, no. 17, pp.
e174-e177, 2017. View at: Publisher Site | PubMed
[31] Ricardo
Diaz-Romero Paz, Pilar Avendaño Altimira, Gustavo Coloma Valverde, et al. “A
Rare Case of Negative-Pressure Hydrocephalus: A Plausible Explanation and the
Role of Transmantle Theory.” World Neurosurg, vol. 125, pp. 6-9, 2019.
View at: Publisher Site | PubMed
[32] Tomohiro
Ozeki, Asami Kubota, Yasuo Murai, et al. “Suspected Low-Pressure Hydrocephalus
Caused by Spinal Drainage after Subarachnoid Hemorrhage.” J Nippon Med Sch,
vol. 89, no. 2, pp. 238-243, 2022. View at: Publisher Site | PubMed
[33] Patrick
Czorlich, Nils Schweingruber, Jennifer Göttsche, et al. “Acute low-pressure
hydrocephalus in aneurysmal subarachnoid hemorrhage.” Neurosurg Focus,
vol. 54, no. 4, pp. E5, 2023. View at: Publisher Site | PubMed
[34] Alan
R Cohen “Low-pressure hydrocephalus.” J Neurosurg, vol. 115, no. 5, pp.
1029-1030, 2011. View at: Publisher Site | PubMed
[35]
Shanshan Duan, Jin Hu “Pathogenesis
and management of low-pressure hydrocephalus: A narrative review.” J Neurol
Sci, vol. 460, pp. 122988, 2024. View at: Publisher Site | PubMed
[36]
J Jurkiewicz “Negative-pressure
hydrocephalus.” J Neurosurg, vol. 85, no. 2, pp. 364-365, 1996. View at:
Publisher Site | PubMed
[37]
Michael B Keough, Albert M Isaacs,
Geberth Urbaneja, et al. “Acute low-pressure hydrocephalus: a case series and
systematic review of 195 patients.” J Neurosurg, vol. 135, no. 1, pp.
300-308, 2020. View at: Publisher Site | PubMed
[38]
Guangzhao Li, Bin Lin, Fei Yang
“Low-/Negative-Pressure Hydrocephalus: To Understand the Formation Mechanism
from the Perspective of Clinicians.” J Neurol Surg A Cent Eur Neurosurg,
vol. 85, no. 5, pp. 492-500, 2024. View at: Publisher Site | PubMed
[39]
McLone DG “Hydrocephalus: Historical
Perspectives and Modern Approaches.” Chicago, IL: Springer; 1980.
[40]
Harold L Rekate, Trimurti D Nadkarni,
Donna Wallace “The importance of the cortical subarachnoid space in
understanding hydrocephalus.” J Neurosurg Pediatr, vol. 2, no. 1, pp.
1-11, 2008. View at: Publisher Site | PubMed
[41]
Harold L Rekate “Low or Negative
Pressure Hydrocephalus Demystified.” World Neurosurg, vol. 128, pp.
287-288, 2019.
[42]
Harold L Rekate “Commentary on the
article “Brain stiffness following recovery in a patient with an episode of
low-pressure hydrocephalus: case report.”” Child’s Nerv Syst, vol. 37,
no. 8, pp. 2699-2700, 2021. View at: Publisher Site | PubMed
[43]
Frank W Stahnisch “Instrument
transfer as knowledge transfer in neurophysiology: François Magendie’s
(1783-1855) early attempts to measure cerebrospinal fluid pressure.” J Hist
Neurosci, vol. 17, no. 1, pp. 72-99, 2008. View at: Publisher Site | PubMed
[44]
SUN T, LIU Q, PU K, et al. “Diagnosis
and treatment of negative pressure hydrocephalus: analysis of 5 cases and
literature review.” Clinical Medicine of China, pp. 281-287, 2023.
[45]
Vesalius A “De Humani Corporis
Fabrica.” Basel, Switzerland: Johannes Oporinus; 1543.
[46]
Walker AE “A History of the Surgery
of the Brain.” Edinburgh, Scotland: Livingstone; 1971.
[47] Xuejian Wang, Zhifeng Wang, Yang Chen “Diagnosis and treatment of low intracranial pressure hydrocephalus.” Int J Neurol Phys Ther, vol. 3, no. 4, pp. 35, 2017.
