Received: Sat 06, Sep 2025
Accepted: Thu 02, Oct 2025
Abstract
Objective: To evaluate the clinical presentation, demographic characteristics, and surgical outcomes of patients with split cord malformation (SCM) who underwent neurosurgical intervention.
Background: Split cord malformation (SCM) represents a spectrum of congenital spinal anomalies characterized by longitudinal division of the spinal cord into two hemicords. This study aims to analyze the clinical features, demographic profile, and postoperative outcomes in surgically treated SCM patients.
Methods: A single-center, retrospective cross-sectional study was conducted at St. Paul’s Hospital Millennium Medical College, AaBET Hospital, including patients treated between January 2021 and February 2025. SCM was diagnosed via preoperative MRI, which guided surgical planning. Postoperative outcomes focused on neurological function. Data were collected using structured questionnaires and analyzed with SPSS version 25.0.
Results: The majority of patients (37.8%) were aged 1-5 years, with females comprising 78.4% (male-to-female ratio ~1:3.65). Back deformity was the most common presenting symptom (73.0%), followed by lower limb neurological deficits (18.9%) and sphincter dysfunction (13.5%). Surgical indications included tethered cord (59.5%) and planned deformity correction (21.6%). Postoperatively, 60% of patients with sphincter disturbances and 45.9% with lower limb weakness showed improvement. No cases of permanent neurological deterioration were observed among the 37 patients.
Conclusions: SCM, though rare, frequently presents with spinal deformity in pediatric patients. Timely surgical intervention is associated with favorable neurological outcomes. Early screening for spinal cord anomalies in children with spinal deformities is essential. Surgical management of SCM and tethered cord prior to deformity correction is safe and may reduce the risk of intraoperative neurological complications.
Keywords
Split cord malformation, tethered cord, spinal deformity, congenital spinal anomalies
1. Background
Split cord malformation (SCM) is a rare congenital neural tube defect characterized by longitudinal division of the spinal cord into two hemicords. Historically, terms such as diastematomyelia and diplomyelia were used interchangeably to describe these anomalies. The term diastematomyelia was first introduced by Hertwig in 1892 and later refined by Bruce et al. in 1906. Today, SCM is the preferred term, reflecting a more precise anatomical classification. Split Cord Malformation (SCM) is divided into two primary types: Type I, where hemicords are housed in separate dural tubes and separated by an osseocartilaginous septum, and Type II, where the hemicords are enclosed in a single dural tube and divided by a fibrous septum [1, 2].
SCM presents with a broad spectrum of neurological and orthopedic symptoms. Radicular low back pain (RLBP) is the most commonly reported symptom, with a frequency ranging from 26% to 88.8% [3, 4]. Neurological deficits, including muscle weakness, atrophy, and hypoesthesia, occur in 22.2% to 68.1% of cases [3, 4]. Although bladder dysfunction and impotence are less common, some studies report neurogenic bladder in up to 68% of cases [4, 5]. Scoliosis is the most frequently associated condition, occurring in 85% of Type I and 75% of Type II SCM cases. The most common anatomical location is the lumbar region (48%), followed by the lumbosacral area (20%) [6].
SCM predominantly affects pediatric populations and has a higher prevalence in females. Mahapatra et al. reported a mean age of 7.3 years with a female-to-male ratio of 1.5:1 [4], findings echoed in other studies [2, 6-9]. Surgery remains the standard treatment for symptomatic SCM or cases with progressive neurological impairment. Outcomes are generally favorable, with up to 78.6% of patients experiencing postoperative neurological improvement [7]. Masroor et al. reported 64% neurologic stability, 16% improvement, and 4% deterioration postoperatively [6].
Management of SCM with congenital scoliosis remains controversial. While some advocate for prophylactic detethering before deformity correction, others suggest that spinal deformity correction can be safely performed in neurologically stable patients without prior detethering [10-13]. Conservative management is often recommended for asymptomatic Type II SCM and selected Type I cases [8]. A two-stage approach is often favored when spinal deformity correction is necessary, to reduce neurological risk [3]. However, Sergeenko et al. found that single-stage surgery, while associated with greater surgical trauma and complications, may be appropriate in select cases, such as when a wide bony septum (Type I SCM) is located at the apex of the spinal deformity. In other scenarios, a two-stage approach is preferred to minimize complications [14]. Intraoperative neurophysiological monitoring (IONM) is essential in these cases, significantly enhancing the safety of detethering and deformity correction surgeries [15].
Data on the rare condition of split cord malformation (SCM) is limited globally, and especially scarce in developing countries. This study investigated the patterns and surgical outcomes of SCM at a Sub-Saharan African tertiary center, where advanced tools like intraoperative neuromonitoring are unavailable.
2. Methods and Materials
2.1. Study Area and Period
This study will be conducted at St. Paul's Hospital Millennium Medical College, AaBET Hospital, Addis Ababa, Ethiopia, covering patients operated between January 2021 and February 2025. AaBET Hospital, an affiliate center of SPHMMC, is located in the northern part of Addis Ababa. It serves as a top-tier referral center, providing advanced medical and surgical care to patients from across the country. The hospital also functions as a teaching institution for both undergraduate and postgraduate students. Additionally, it is well recognized for its expertise in spine deformity management and its collaboration with organizations dedicated to supporting spine deformity care.
2.2. Study Design
Single-center, cross-sectional, descriptive, retrospective study.
2.3. Population
2.3.1. Source Population
All patients who underwent surgery for SCM during the study period at St. Paul's Hospital Millennium Medical College, AaBET Hospital.
2.4. Inclusion Criteria
All patients who underwent surgery for SCM during the study period at St. Paul's Hospital Millennium Medical College, AaBET Hospital.
2.5. Exclusion Criteria
Incomplete medical record.
2.6. Sampling Technique and Sample Size
All eligible patients meeting the inclusion criteria were included in the study.
2.7. Data Collection
Data was collected using a structured questionnaire by trained research assistants.
2.8. Data Analysis and Interpretation
Data was analyzed using SPSS version 25. A comparative analysis with previous studies will be conducted to contextualize findings.
3. Results
3.1. Sociodemographic Characteristics
The majority of patients were between 1 to 5 years old, comprising 37.8% of the total, and most were female (78.4%) (Table 1) with a male-to-female ratio was 1 to 3.65.
Table. 1. Sociodemographic
characteristics of study participants.
|
Variable |
Category |
Frequency |
Percent |
|
Age |
0-1 years |
2 |
5.4% |
|
1-5 years |
14 |
37.8% |
|
|
6-10 years |
8 |
21.6% |
|
|
11-13 years |
5 |
13.5% |
|
|
14-18 years |
8 |
21.6% |
|
|
Gender |
Male |
8 |
21.6% |
|
Female |
29 |
78.4% |
3.2. Clinical Presentation
Back deformity was the most common clinical feature, observed in 73.0% of patients. Approximately 40.5% had leg deformities, and none reported leg pain at the time of presentation (Table 2). The forms of leg deformity include leg length discrepancy, foot and ankle deformity. A hairy patch on the back was the most common skin stigmata noted, seen in 8 (21.6%) patients. Other stigmata include hair with hyperpigmentation 2 (5.4%), dark nevus 1 (2.7%), hemangioma 1 (2.7%), and dimple 1 (2.7%). Out of the patients presenting with lower limb weakness, three had asymmetric paraparesis while four had symmetrical paraparesis.
3.3. Comorbidity Status
Scoliosis was the commonest comorbidity seen in 94% (35) of the patients. Orthopedic lower limb (LL) anomalies were observed in 37.8% and visceral anomalies in 8.1% (3) of patients (Table 3).
Table. 2. Clinical presentation.
|
Clinical presentation |
||
|
Back Deformity |
30 |
81.1% |
|
Deformity of legs |
15 |
40.5% |
|
Skin Stigmata |
13 |
35.1% |
|
Lower limb weakness |
7 |
18.9% |
|
Abnormal Bowel/Bladder Function |
5 |
13.5% |
|
Back Pain |
2 |
5.4% |
Table. 3. Comorbidities.
|
Scoliosis |
35 |
94.60% |
|
Orthopedic LL Anomalies |
14 |
37.80% |
|
Visceral Anomalies |
3 |
8.10% |
|
Chiari Malformation |
2 |
5.40% |
|
Meningocele |
1 |
2.70% |
|
Teratoma (SC) |
1 |
2.70% |
3.4. Imaging Finding
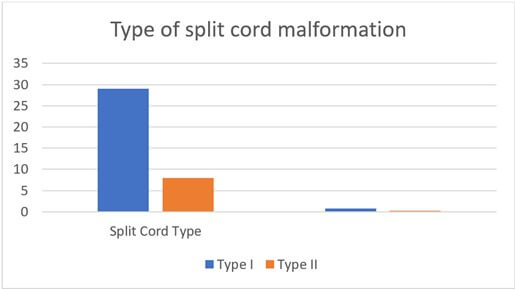
On the MRI type one split cord was the commonest type, accounting for 78.4% of the cases (Figure 1).
Type 1 split cord is the commonest type seen in 78.4 percent of the patients and type 2 accounts for 21.6 percent of the patients. One of the findings that was noted on the MRI was Low-lying conus, which was noted in 97.3% cases (Table 4). Most of the bony spurs in type one SCM were located in the lumbar spine (55%) (Figure 2).
3.5. Surgical Indications and Outcomes
Tethered cord and planned deformity correction were the most common indications for surgery (59.5% and 21.6%, respectively) (Table 5). Most patients (51.4%) had moderate estimated blood loss (EBL) during surgery (Table 6). The commonest post-operative complication was CSF leak, with surgery for the leak that happened in 6 patients (16.2%) (Table 7).
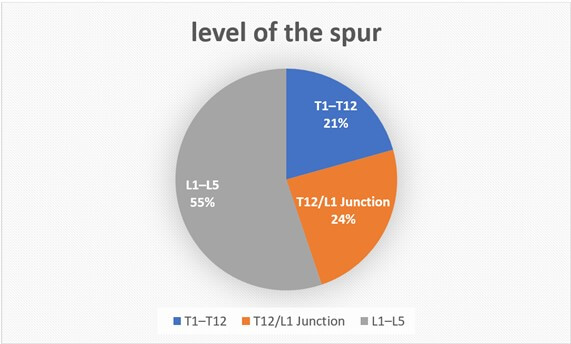
Table. 4. MRI findings.
|
Vertebral Anomalies |
19 |
51.40% |
|
Low-Lying Conus |
36 |
97.30% |
|
Lipoma |
7 |
18.90% |
|
Syrinx/Hydromyelia |
7 |
18.90% |
Table. 5. Surgical
indications.
|
Surgical Indication |
||
|
Planned deformity surgery |
8 |
21.6% |
|
Bowel/Bladder Dysfunction |
2 |
5.4% |
|
Tethered Cord |
22 |
59.5% |
|
Prophylactic |
5 |
13.5% |
Table 6: estimated blood loss.
|
Estimated Blood Loss |
||
|
Low |
14 |
37.8% |
|
Moderate |
19 |
51.4% |
|
Severe |
4 |
10.8% |
Table. 7. Post-operative complications.
|
Complication Type |
||
|
SSI |
1 |
2.70% |
|
CSF Leak |
1 |
2.70% |
|
Reoperation |
1 |
2.70% |
|
CSF Leak + Reoperation |
6 |
16.22% |
|
Transient urinary retention symptoms |
1 |
2.70% |
3.6. Postoperative Functional Outcomes
Out of the five patients who had urinary symptoms 3 had improvement post operatively and 2 remained the same. Lower limb weakness showed improvement in 3 (42.9%), and 4 (57.1%) remained the same (Table 8). There was no persistent motor or bowel bladder deterioration among the patient with intact function.
3.7. Lower Limb Weakness vs. Neurological Outcome
The most frequent surgery performed was spur excision with filum resection for Type 1 SCM (54.1%) (Table 9).
Table. 8. Neurologic outcome.
|
LL Weakness Type |
Improved |
Same |
|
Asymmetric (n=4) |
1 (25%) |
3 (75%) |
|
Symmetric Incomplete
Weakness (n=3) |
2 (66.7%) |
1 (33.3%) |
|
No weakness (n=30) |
|
30 (100%) |
Table. 9. Type of surgery.
|
Surgery type |
Number |
Percent |
|
Band
release and filum resection for Type 2 SCM |
5 |
13.5% |
|
Exploration
with no band found and filum resection for Type 2 SCM |
3 |
8.1% |
|
Filum
resection only for Type 2
SCM |
1 |
2.7% |
|
Spur
excision and filum resection for Type 1 SCM |
20 |
54.1% |
|
Spur excision without
filum resection Type 1 SCM |
8 |
21.6% |
4. Discussion
Split Cord Malformation (SCM) is a rare congenital spinal anomaly, with an estimated prevalence of 1 in 5,499 live births (0.02%) [16]. SCM encompasses a range of spinal cord anomalies characterized by the division of the spinal cord into two hemicords.
In our study, we observed a notable predominance of SCM in females, aligning with findings from other studies. [1, 4, 8, 9]. The most common clinical manifestation was back deformity, present in 81% of patients, with radiological imaging revealing deformities in 94%. This aligns with other research indicating that scoliosis occurs in approximately 80% of cases [4]. Interestingly, while Beuriat et al. reported that 88% of patients experienced low back pain, only 5.4% of our patients reported this symptom [3]. Skin stigmata were observed in 35% of the patients, with hairy patches being the most common type of stigmata, a finding consistent with existing literature [4].
MRI scans revealed that a low-lying conus was the most frequently associated finding, identified in 97% of patients. The most common level was at the L3, in 46% of cases. Despite the prevalence of low-lying conus, only 32% of patients exhibited neurological deficits. Type I SCM was the most frequently encountered type, representing 97% of cases, with the majority of spurs located in the lumbar spine (55%).
Surgeries performed were based on the categorization of patients into Type I and Type II SCM per MRI/CT findings. Our institution does not have neuromonitoring, so it was not employed in these cases. Filum resection was accomplished by identifying the filum based on anatomical characteristics using a high-magnification operative microscope. The surgical approaches included:
- Band release and filum resection for Type 2 SCM
- Exploration with no band found and filum resection for Type 2 SCM
- Filum resection only for Type 2 SCM
- Spur excision and filum resection for Type 1 SCM
- Spur excision without filum resection for Type 1 SCM
In Type II SCM surgeries, the primary goal has been to release fibrous bands at the level of the split and untether the filum. For cases with a lengthy segment split of the cord, exploration of the lower end of the split was conducted. Among the eight patients who underwent exploration of the Type 2 SCM segment, 5 patients (62.5%) exhibited fibrous adhesions at the level of the split that required release.
One patient, who initially only had a filum untethering procedure, later after 8 months developed neurogenic bladder dysfunction and subsequently underwent exploration of the split segment. During this exploration, fibrous adhesions were identified and released, resulting in improvement of bladder symptoms. This suggests the importance of exploring the cord split segment of the Type II SCM and releasing fibrous adhesions prior to filum untethering.
For patients diagnosed with type I spinal cord malformation (SCM), our main surgical approach involved excising the bone spur, resecting the dural sleeve to split the dural sac, and then recanalizing the sac by ensuring a watertight closure of the dorsal dura. In our study, this surgical method was successfully implemented in 71.4% of type I SCM cases. In 8 patients (29%), we performed bone spur excision without filum resection. This decision was due to intraoperative challenges in identifying the filum in three cases, while the other five patients presented with significant bony morphological anomalies that complicated spur excision, leading to considerable blood loss. Consequently, we opted for untethering to mitigate complications related to extended surgical duration and blood loss. Notably, the patients who did not undergo filum untethering did not experience neurological deterioration or worsening conditions during the follow-up, although long-term monitoring is necessary to assess any future impacts.
In our cohort, the primary surgical indication was tethered cord observed on imaging, which accounted for approximately 60% of the patients, followed by planned deformity surgery at 21.6%. The latter group included patients who had screening MRIs before undergoing corrective surgery for deformities, at which time the SCM is identified and sent to be dealt first before the deformity surgery.
The most common complication we observed was a cerebrospinal fluid (CSF) leak, which occurred in 19% of patients. Six of these individuals required reoperation to repair the leak. Our analysis revealed no statistically significant relationship between surgical duration and postoperative complications, with a confidence interval (CI) at 95% of 0.92 (0.56, 1.52) and an unstandardized regression coefficient (B) of -0.08. Notably, patients treated with Type 1 SCM spur excision combined with filum resection had a 0.86 times lower risk of developing postoperative complications compared to those who underwent Type 1 SCM spur excision without filum resection, with an adjusted odds ratio (AOR) of 0.14 (0.02, 0.98). This finding may stem from the more complex anatomy encountered during surgery in the latter cases, which could have affected the durability of dural closure.
Among the five patients who reported urinary symptoms, three experienced improvement postoperatively, while two remained unchanged. Of the patients with lower limb weakness, 46% (3 out of 7) showed improvement in motor strength, with the remainder maintaining their preoperative status. Importantly, there was no permanent postoperative deterioration in bowel, bladder, or motor functions observed. We did not routinely conduct postoperative imaging unless surgery for spine deformity was required, following literature recommendations regarding the follow-up of tethered cord malformation [17].
5. Conclusion
Split Cord Malformation is a rare condition that often presents with spinal deformity in children. This study demonstrates that timely surgical intervention can lead to neurological improvement. In pediatric patients presenting with spinal deformities, early screening for spinal cord anomalies is essential. The findings also indicate that SCM and associated tethered cord can be safely managed surgically prior to deformity correction, potentially minimizing the risk of neurological complications during deformity correction surgery.
Limitation
Our institution does not have intraoperative neuromonitoring; therefore, it was not utilized in any of our surgical cases. Filum terminale resection was performed by identifying the structure based on its anatomical features under a high-magnification operative microscope. This study does not include long-term patient outcomes, which will require future follow-up investigations.
Funding
None.
Ethical Approval
St Paul Hospital Millennium Medical College, Institutional research Ethics Review Committee (IRERC). Ref No- Pm23,525.
Author Contributions
Dr. Abraham Tadele Gebremedihen: Study conception or design, data collection, data analysis or interpretation, and manuscript writing. Dr. Petros Teklewold: Study conception or design, data collection, data analysis or interpretation, and manuscript writing. Dr. Biniam Gebreegziabher Gebru: Study conception and manuscript writing. Dr. Milena Gebreegziabher: Study conception, data analysis and manuscript writing. Dr Dagmawi Sileshi Tekelmariam: Study conception, data analysis and manuscript writing. Dr Fassil B Mesfin: Study conception and manuscript writing.
REFERENCES
[1] D Pang, M S
Dias, M Ahab-Barmada “Split Cord Malformation.” Neurosurgery, vol. 31,
no. 3, pp. 451-480, 1992. View at: Publisher Site | PubMed
[2] D Pang “Split
Cord Malformation.” Neurosurgery, vol. 31, no. 3, pp. 481-500, 1992.
View at: Publisher Site | PubMed
[3] Pierre-Aurélien
Beuriat, Federico Di Rocco, Alexandru Szathmari, et al. “Correction to:
Management of split cord malformation in children: the Lyon experience.” Childs
Nerv Syst, vol. 34, no. 7, pp. 1433-1433, 2018. View at: Publisher Site | PubMed
[4] Ashok K
Mahapatra, Deepak Kumar Gupta “Split cord malformations: a clinical study of
254 patients and a proposal for a new clinical—imaging classification.” J
Neurosurg, vol. 103, no. 6 Suppl, pp. 531-536, 2005. View at: Publisher Site | PubMed
[5] Kamil Melih
Akay, Yusuf Izci, Alper Baysefer, et al. “Split cord malformation in adults.” Neurosurg
Rev, vol. 27, no. 2, pp. 99-105, 2004. View at: Publisher Site | PubMed
[6] Mehar
Masroor, Qurat-Ul-Ain Virani, Saqib Kamran Bakhshi, et al. “Quality of Life of
Children Treated for Split Cord Malformation.” World Neurosurg, vol.
188, pp. e163-e167, 2024. View at: Publisher Site | PubMed
[7] Makhmud
Akhmediev, Gulnarakhan Alikhodjaeva, Odilkhon Usmankhanov, et al. “Management
of split cord malformation and tethered cord syndrome: Experience of a main
referral center in Uzbekistan.” Clin Neurol Neurosurg, vol. 245, pp.
108510, 2024. View at: Publisher Site | PubMed
[8] Sheng-Li
Huang, Xi-Jing He, Kun-Zheng Wang, et al. “Diastematomyelia: A 35-Year
Experience.” Spine, vol. 38, no. 6, pp. E344-E349, 2013. View at: Publisher Site | PubMed
[9] Fan Feng,
Haining Tan, Xingye Li, et al. “Radiographic characteristics in congenital
scoliosis associated with split cord malformation: a retrospective study of 266
surgical cases.” BMC Musculoskelet Disord, vol. 18, no. 1, pp. 420,
2017. View at: Publisher Site | PubMed
[10] Kaustubh
Ahuja, Syed Ifthekar, Samarth Mittal, et al. “Is detethering necessary before
deformity correction in congenital scoliosis associated with tethered cord
syndrome: a meta-analysis of current evidence.” Eur Spine J, vol. 30,
no. 3, pp. 599-611, 2021. View at: Publisher Site | PubMed
[11] Jianxiong
Shen, Jianguo Zhang, Fan Feng, et al. “Corrective Surgery for Congenital
Scoliosis Associated with Split Cord Malformation: It May Be Safe to Leave
Diastematomyelia Untreated in Patients with Intact or Stable Neurological
Status.” J Bone Jt Surg, vol. 98, no. 11, pp. 926-936, 2016. View at: Publisher
Site | PubMed
[12] Huiren Tao,
Kai Yang, Tao Li, et al. “Is It Possible to Correct Congenital Spinal Deformity
Associated With a Tethered Cord Without Prophylactic Intradural Detethering?” Clin
Orthop, vol. 477, no. 7, pp. 1689-1697, 2019. View at: Publisher Site | PubMed
[13] Qinghua Zhao,
Benlong Shi, Xu Sun, et al. “Do untreated intraspinal anomalies in congenital
scoliosis impact the safety and efficacy of spinal correction surgery? A
retrospective case-control study.” J Neurosurg Spine, vol. 31, no. 1,
pp. 40-45, 2019. View at: Publisher Site | PubMed
[14] Olga M
Sergeenko Pavlova, Dmitry M Savin, Sergey O Ryabykh “Treatment of spinal
deformity with diastematomyelia type I: one-stage, two-stage surgery and new
technique (vertebral column resection through wide bony septum).” Childs
Nerv Syst, vol. 38, no. 1, pp. 163-172, 2022. View at: Publisher Site | PubMed
[15] Gábor Fekete,
László Bognár, László Novák “Surgical treatment of tethered cord
syndrome—comparing the results of surgeries with and without
electrophysiological monitoring.” Childs Nerv Syst, vol. 35, no. 6, pp.
979-984, 2019. View at: Publisher Site | PubMed
[16] Gulsah
Bademci, Meral Saygun, Funda Batay, et al. “Prevalence of Primary Tethered Cord
Syndrome Associated with Occult Spinal Dysraphism in Primary School Children in
Turkey.” Pediatr Neurosurg, vol. 42, no. 1, pp. 4-13, 2006. View at: Publisher
Site | PubMed
[17] P David Halevi, Suhas Udayakumaran, Liat Ben-Sira, et al. “The value of postoperative MR in tethered cord: a review of 140 cases.” Childs Nerv Syst, vol. 27, no. 12, pp. 2159-2162, 2011. View at: Publisher Site | PubMed
