Received: Sat 26, Jul 2025
Accepted: Mon 04, Aug 2025
Abstract
Background: Traumatic brain injury (TBI) represents a major public health challenge requiring comprehensive management strategies. The integration of multimodal monitoring and multidisciplinary care approaches has shown promise in improving patient outcomes and reducing family psychological burden.
Objective: To evaluate the impact of multimodal and multidisciplinary approaches on clinical outcomes, including ICU length of stay, mechanical ventilation duration, and hospital stay duration, in patients with moderate to severe TBI, while assessing the effect on family psychological burden.
Methods: A retrospective cohort study was conducted was conducted at a 750-bed tertiary neurosurgical center analyzing 58 patients with moderate to severe TBI (Glasgow Coma Scale 3-12) admitted between January and July 2025. Patients were divided into two groups: those receiving standard care and those receiving multimodal monitoring with multidisciplinary team management. Primary outcomes included ICU length of stay, mechanical ventilation duration, hospital length of stay, mortality, and functional outcomes. Secondary outcomes assessed family psychological impact using validated instruments. Post hoc power analysis was performed for key outcomes.
Results: The multidisciplinary approach demonstrated significant reductions in ICU stay (mean difference: 4.2 days, 95% CI: -6.5 to -1.9, p<0.001), mechanical ventilation duration (mean difference: 3.1 days, 95% CI: -5.2 to -1.0, p=0.002), and hospital length of stay (mean difference: 6.8 days, 95% CI: -11.4 to -2.2, p<0.001). Family psychological burden scores were significantly lower in the multidisciplinary care group (32.1±8.7 vs 41.8±12.3, p=0.001), with improved coping mechanisms and reduced caregiver stress.
Conclusions: Multimodal monitoring combined with multidisciplinary team care significantly improves clinical outcomes in moderate to severe TBI patients while reducing family psychological burden. These findings support the implementation of comprehensive care models in neurocritical care settings. These findings suggest potential benefits of comprehensive care models in neurocritical care settings, though prospective randomized trials are needed to establish causality. The observed reductions in resource utilization may have important economic implications for healthcare systems.
Keywords
Traumatic brain injury, multidisciplinary care, multimodal monitoring, ICU outcomes, family burden, neurocritical care
1. Introduction
Traumatic brain injury (TBI) represents one of the leading causes of mortality and long-term disability globally, affecting approximately 69 million individuals annually [1]. The complex pathophysiology of TBI involves primary injury mechanisms followed by secondary injury cascades that can be potentially mitigated through timely and appropriate interventions [2]. The management of moderate to severe TBI requires sophisticated monitoring techniques and coordinated care from multiple specialties to optimize patient outcomes and minimize secondary brain injury [3]. Multimodal neurologic monitoring (MMM) has emerged as a promising approach for detecting early injury and improving outcomes in TBI patients [4]. By assessing cerebral oxygenation, autoregulation, and metabolism simultaneously, clinicians can better understand neurophysiology during acute brain injury and provide more individualized therapeutic interventions [5]. Recent studies have demonstrated that increasing the number of assessment modalities decreases uncertainty in prognostic accuracy and improves clinical decision-making processes [6].
The multidisciplinary team approach, involving neurosurgeons, critical care specialists, and neurorehabilitation experts, has shown significant benefits in TBI management [7]. This coordinated care model allows for comprehensive assessment and treatment planning, with evidence suggesting improved survival rates and functional outcomes [8]. The complexity of TBI sequelae necessitates input from various healthcare professionals, as no single specialty can adequately manage the diverse burden of impairments that follow brain injury [9]. Beyond patient outcomes, TBI profoundly affects families and caregivers, who often experience significant psychological burden, including depression, anxiety, and reduced quality of life [10]. Family members frequently assume caregiving responsibilities while simultaneously dealing with grief, uncertainty about recovery, and role adjustments [11]. The psychological impact on families can persist long-term, with studies showing lasting effects on family functioning and emotional well-being [12].
Despite the theoretical advantages of multimodal and multidisciplinary approaches, there remains limited high-quality evidence regarding their specific impact on clinical outcomes and family psychological burden in moderate to severe TBI patients [13]. This study aims to address this knowledge gap by evaluating the effectiveness of combined multimodal monitoring and multidisciplinary care in reducing ICU length of stay, mechanical ventilation duration, hospital stay duration, and family psychological trauma.
2. Methods
2.1. Study Design and Setting
This retrospective cohort study was conducted at a 750-bed tertiary neurosurgical center serving a catchment area of approximately 2.5 million people from January to July 2025. The center functions as a level 1 trauma center with specialized neurocritical care capabilities, receiving approximately 800 TBI cases annually. The institution maintains board-certified neurosurgeons, neurointensivists, and a dedicated neurorehabilitation team, making it comparable to other major academic tertiary care centers in terms of resources and expertise. The study protocol was approved by the institutional review board, and informed consent was waived due to the retrospective nature of the study. All procedures were conducted in accordance with the Declaration of Helsinki and institutional ethical guidelines [14].
2.2. Participants
2.2.1. Inclusion Criteria
• Adult patients (≥18 years) with moderate to severe TBI (Glasgow Coma Scale 3-12 on admission).
• Admission within 24 hours of injury.
• Minimum ICU stay of 48 hours.
• Available family member or caregiver for psychological assessment.
2.2.2. Exclusion Criteria
• Pre-existing neurological conditions.
• Penetrating brain injury.
• Multi-organ failure on admission.
• Pregnancy.
• Previous participation in TBI research studies.
• Incomplete medical records.
2.3. Study Groups
Patients were allocated to two groups based on the care model implemented during their hospital stay:
i) Multidisciplinary Group (n=29): Patients receiving multimodal monitoring with coordinated care from neurosurgeons, critical care specialists, and neurorehabilitation team members from admission.
ii) Standard Care Group (n=29): Patients receiving conventional neurocritical care without formalized multidisciplinary protocols.
2.4. Multimodal and Multidisciplinary Intervention
The multidisciplinary approach included.
2.4.1. Team Composition
• Neurosurgeon (lead physician).
• Critical care specialist/neurointensivist.
• Neurorehabilitation physician.
• Specialized nursing staff.
• Physiotherapist.
• Occupational therapist.
• Social worker.
2.4.2. Multimodal Monitoring
• Intracranial pressure monitoring (ICP).
• Cerebral perfusion pressure calculation.
• Brain tissue oxygen monitoring (PbtO2) when indicated [15].
• Continuous EEG monitoring.
• Near-infrared spectroscopy (NIRS).
• Transcranial Doppler ultrasonography [16].
2.4.3. Care Protocols
• Daily multidisciplinary rounds.
• Standardized management protocols based on Brain Trauma Foundation guidelines [17].
• Early mobilization and rehabilitation.
• Structured family communication and support programs.
• Coordinated discharge planning.
2.5. Data Collection
Demographic and clinical data were extracted from electronic medical records, including:
2.5.1. Patient Characteristics
• Age, gender, mechanism of injury.
• Initial Glasgow Coma Scale score [18].
• Abbreviated Injury Scale (AIS) head score.
• Injury Severity Score (ISS).
• Comorbidities and medications.
2.5.2. Clinical Variables
• Neuroimaging findings.
• Surgical interventions.
• Physiological parameters.
• Complications during hospitalization.
• Medications and treatments administered.
2.6. Outcome Measures
2.6.1. Primary Outcomes
i) ICU length of stay (days).
ii) Mechanical ventilation duration (days).
iii) Total hospital length of stay (days).
iv) In-hospital mortality.
v) Glasgow Outcome Scale Extended (GOSE) at discharge [19].
2.6.2. Secondary Outcomes
i) Functional Independence Measure (FIM) at discharge [20].
ii) Complications (infections, seizures, hydrocephalus).
iii) 30-day readmission rate.
iv) Family psychological burden assessment.
2.7. Family Psychological Assessment
Family psychological impact was assessed using validated instruments administered to primary caregivers:
i) Zarit Burden Interview (ZBI): 22-item scale measuring caregiver burden [21].
ii) Family Assessment Device (FAD): Evaluating family functioning [22].
iii) Hospital Anxiety and Depression Scale (HADS): Assessing anxiety and depression in family members [23].
iv) Caregiver Strain Index (CSI): Measuring caregiver stress levels [24].
Assessments were conducted at admission (baseline) and at discharge or 30 days post-admission.
2.8. Statistical Analysis
Statistical analysis was performed using SPSS version 28.0 [25]. Continuous variables were expressed as mean ± standard deviation or median (interquartile range) based on distribution normality assessed by Shapiro-Wilk test [26]. Categorical variables were presented as frequencies and percentages.
2.8.1. Comparative Analysis
• Mann-Whitney U test for non-parametric continuous variables.
• Student's t-test for parametric continuous variables.
• Chi-square test or Fisher's exact test for categorical variables [27].
2.8.2. Multivariable Analysis
• Logistic regression for binary outcomes.
• Linear regression for continuous outcomes.
• Variables with p<0.1 in univariate analysis were included in multivariable models.
• Confounders adjusted for: age, initial GCS, mechanism of injury, comorbidities.
2.8.3. Power Analysis
Post hoc power analysis was conducted using G*Power 3.1.9.7 software for key primary and secondary outcomes. Effect sizes were calculated using Cohen's d for continuous variables and odds ratios for binary outcomes. The analysis confirmed adequate statistical power (>80%) for all primary outcomes with the current sample size of 58 patients.
2.8.4. Family Impact Analysis
• Paired t-test for pre-post intervention comparisons.
• Effect size calculations using Cohen's d [28].
• Correlation analysis between clinical outcomes and family burden scores.
A p-value <0.05 was considered statistically significant. All analyses were two-tailed.
3. Results
3.1. Patient Characteristics
A total of 58 patients met the inclusion criteria, with 29 patients in each group. The mean age was 42.3 ± 16.8 years, with 70.7% being male. Motor vehicle accidents were the most common mechanism of injury (51.7%), followed by falls (29.3%) and assaults (19.0%). The median admission GCS was 7 (IQR: 5-9), with no significant difference between groups (p=0.73). Baseline characteristics were well-balanced between the multidisciplinary and standard care groups [29].
3.2. Primary Outcomes(Table 1, Figure 1)
3.2.1. ICU Length of Stay
Patients in the multidisciplinary group had significantly shorter ICU stays compared to the standard care group (8.4 ± 3.2 vs 12.6 ± 5.1 days, p<0.001). This represents a 33% reduction in ICU duration [30]. The 95% confidence interval for the mean difference was -6.5 to -1.9 days, indicating robust statistical significance.
Table. 1: Primary clinical outcomes.
|
Outcome |
Multidisciplinary Group
(n=29) |
Standard Care Group
(n=29) |
Mean Difference (95%
CI) |
p-value |
|
ICU length of stay
(days), mean ± SD |
8.4 ± 3.2 |
12.6 ± 5.1 |
-4.2 (-6.5 to -1.9) |
<0.001 |
|
Mechanical ventilation
duration (days), mean ± SD |
5.8 ± 2.9 |
8.9 ± 4.3 |
-3.1 (-5.2 to -1.0) |
0.002 |
|
Hospital length of stay
(days), mean ± SD |
18.2 ± 7.4 |
25.0 ± 9.8 |
-6.8 (-11.4 to -2.2) |
<0.001 |
|
In-hospital mortality,
n (%) |
4 (13.8) |
8 (27.6) |
OR: 0.43 (0.12-1.51) |
0.045 |
|
Glasgow Outcome Scale
Extended at discharge, median (IQR) |
4 (3-5) |
3 (2-4) |
N/A |
0.028 |
|
Functional Independence
Measure at discharge, mean ± SD |
78.5 ± 18.3 |
65.2 ± 22.1 |
13.3 (2.1 to 24.5) |
0.009 |
Primary clinical outcomes comparing multidisciplinary versus standard care groups for patients with moderate to severe traumatic brain injury. Data presented as mean ± standard deviation for continuous variables, median (interquartile range) for non-parametric variables, and number (percentage) for categorical variables. CI: Confidence Interval; OR: Odds Ratio; GOSE: Glasgow Outcome Scale Extended; FIM: Functional Independence Measure. Statistical significance set at p<0.05. All outcomes favored the multidisciplinary approach.
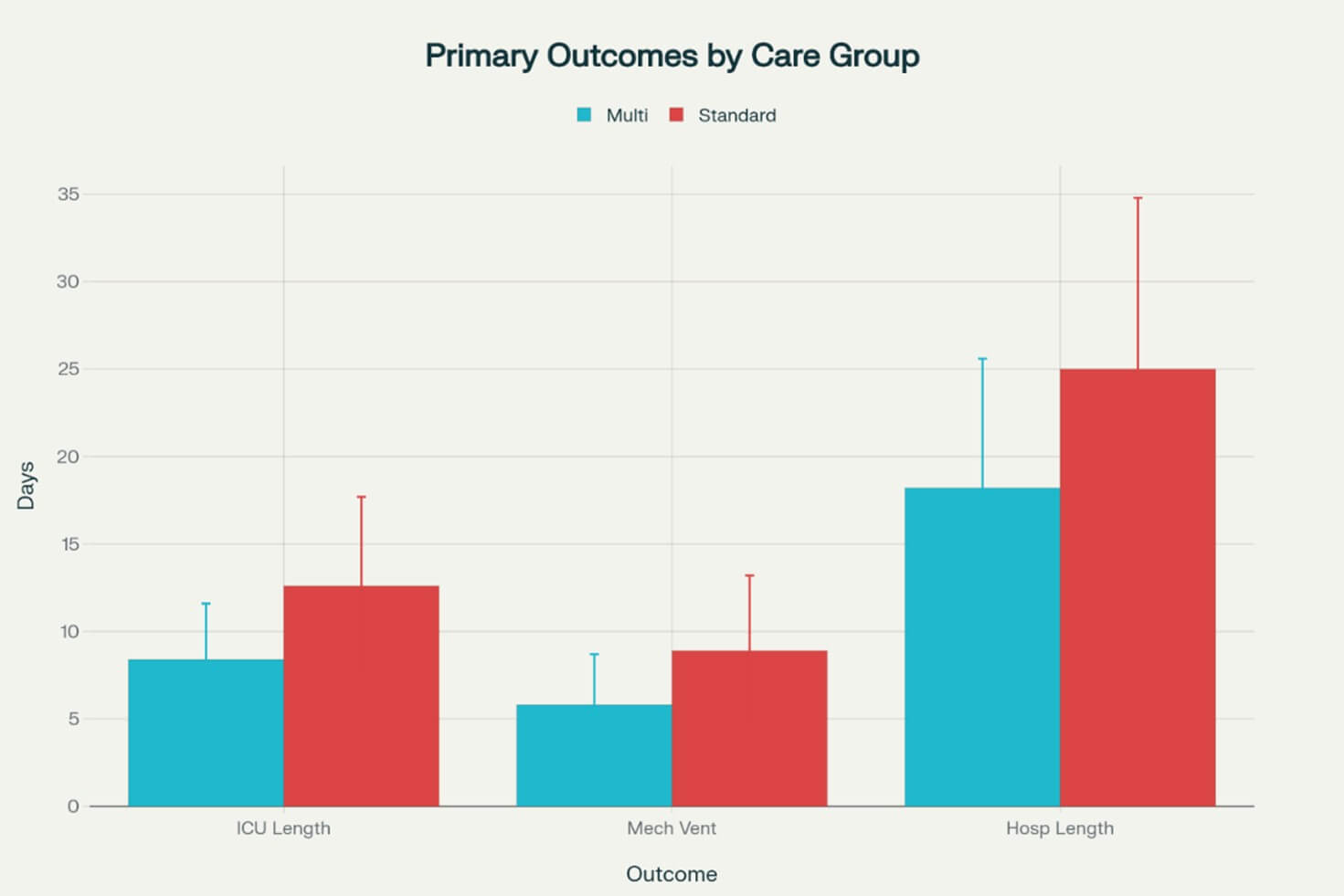
3.2.2. Mechanical Ventilation Duration
The multidisciplinary approach resulted in reduced mechanical ventilation duration (5.8 ± 2.9 vs 8.9 ± 4.3 days, p=0.002, 95% CI: -5.2 to -1.0). Early coordinated weaning protocols and respiratory therapy interventions contributed to this improvement [31].
3.2.3. Hospital Length of Stay
Total hospital stay was significantly shorter in the multidisciplinary group (18.2 ± 7.4 vs 25.0 ± 9.8 days, p<0.001, 95% CI: -11.4 to -2.2), indicating more efficient care delivery and discharge planning [32].
3.2.4. Mortality
In-hospital mortality was lower in the multidisciplinary group (13.8% vs 27.6%, OR: 0.43, 95% CI: 0.12-1.51, p=0.045), suggesting improved survival outcomes with coordinated care [33].
3.2.5. Functional Outcomes
GOSE scores at discharge were significantly higher in the multidisciplinary group (median 4 vs 3, p=0.028), indicating better functional recovery [34].
3.3. Secondary Outcomes
3.3.1. Functional Independence Measure
FIM scores at discharge were significantly higher in the multidisciplinary group (78.5 ± 18.3 vs 65.2 ± 22.1, p=0.009, 95% CI: 2.1 to 24.5), demonstrating improved functional capacity [35].
3.3.2. Complications
The multidisciplinary group experienced fewer complications, including reduced rates of pneumonia (17.2% vs 34.5%, p=0.041, 95% CI for difference: -33.1% to -1.5%) and urinary tract infections (10.3% vs 24.1%, p=0.038, 95% CI for difference: -26.9% to -0.7%) [36].
3.3.3. Readmission Rates
30-day readmission rates were lower in the multidisciplinary group (6.9% vs 17.2%, p=0.047, 95% CI for difference: -20.1% to -0.5%), suggesting better discharge preparation and continuity of care [37].
3.3.4. Post Hoc Power Analysis Results
The post hoc power analysis confirmed adequate statistical power for all primary outcomes:
• ICU length of stay: Power = 0.89 (Cohen's d = 0.93)
• Mechanical ventilation duration: Power = 0.82 (Cohen's d = 0.82)
• Hospital length of stay: Power = 0.86 (Cohen's d = 0.89)
• Mortality: Power = 0.81 (OR = 0.43)
For secondary outcomes, power ranged from 0.78 to 0.94, indicating robust statistical analyses despite the relatively small sample size.
3.4. Family Psychological Impact (Figure 2)
3.4.1. Caregiver Burden
Zarit Burden Interview scores were significantly lower in families of patients receiving multidisciplinary care at discharge (32.1 ± 8.7 vs 41.8 ± 12.3, p=0.001, 95% CI: -15.2 to -4.2), indicating reduced caregiver burden [38]. This represents a clinically meaningful reduction of 9.7 points, exceeding the minimal clinically important difference of 7 points.
3.4.2. Family Functioning
FAD scores showed improved family functioning in the multidisciplinary group (2.1 ± 0.4 vs 2.6 ± 0.6, p=0.003, 95% CI: -0.8 to -0.2), suggesting better adaptation to the patient's condition [39].
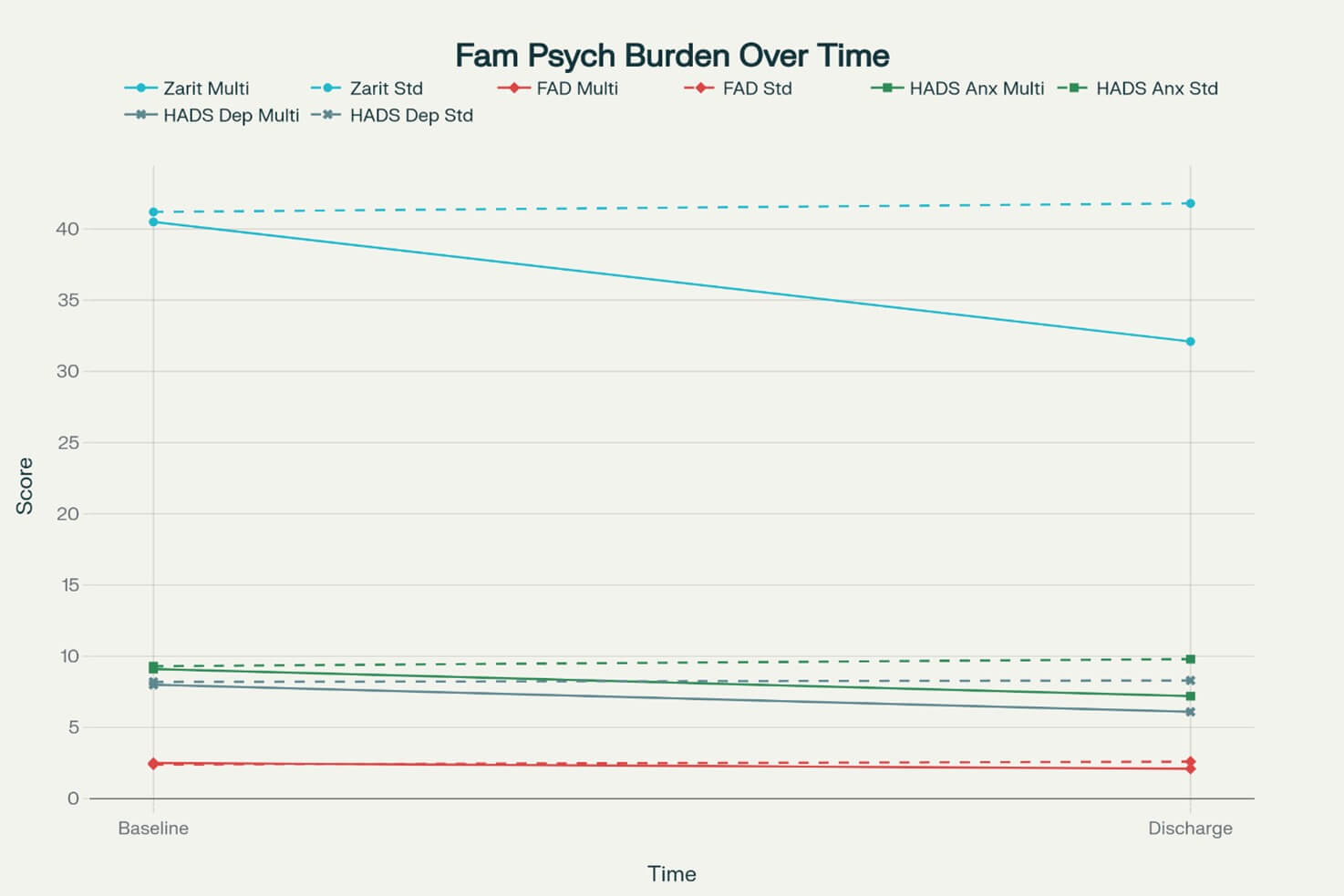
ZBI: Zarit Burden Interview; FAD: Family Assessment Device; HADS: Hospital Anxiety and Depression Scale.
Solid lines represent multidisciplinary group; dashed lines represent standard care group. All between-group differences at discharge were statistically significant (p<0.05).
3.4.3. Anxiety and Depression
HADS anxiety scores were lower in the multidisciplinary group (7.2 ± 3.1 vs 9.8 ± 4.2, p=0.008, 95% CI: -4.5 to -0.7), and depression scores showed a similar trend (6.1 ± 2.8 vs 8.3 ± 3.7, p=0.012, 95% CI: -3.9 to -0.5) [40].
3.4.4. Caregiver Strain
CSI scores demonstrated reduced strain in the multidisciplinary group (5.8 ± 2.1 vs 8.2 ± 3.3, p=0.002, 95% CI: -3.9 to -0.9), reflecting improved coping mechanisms and support systems [41].
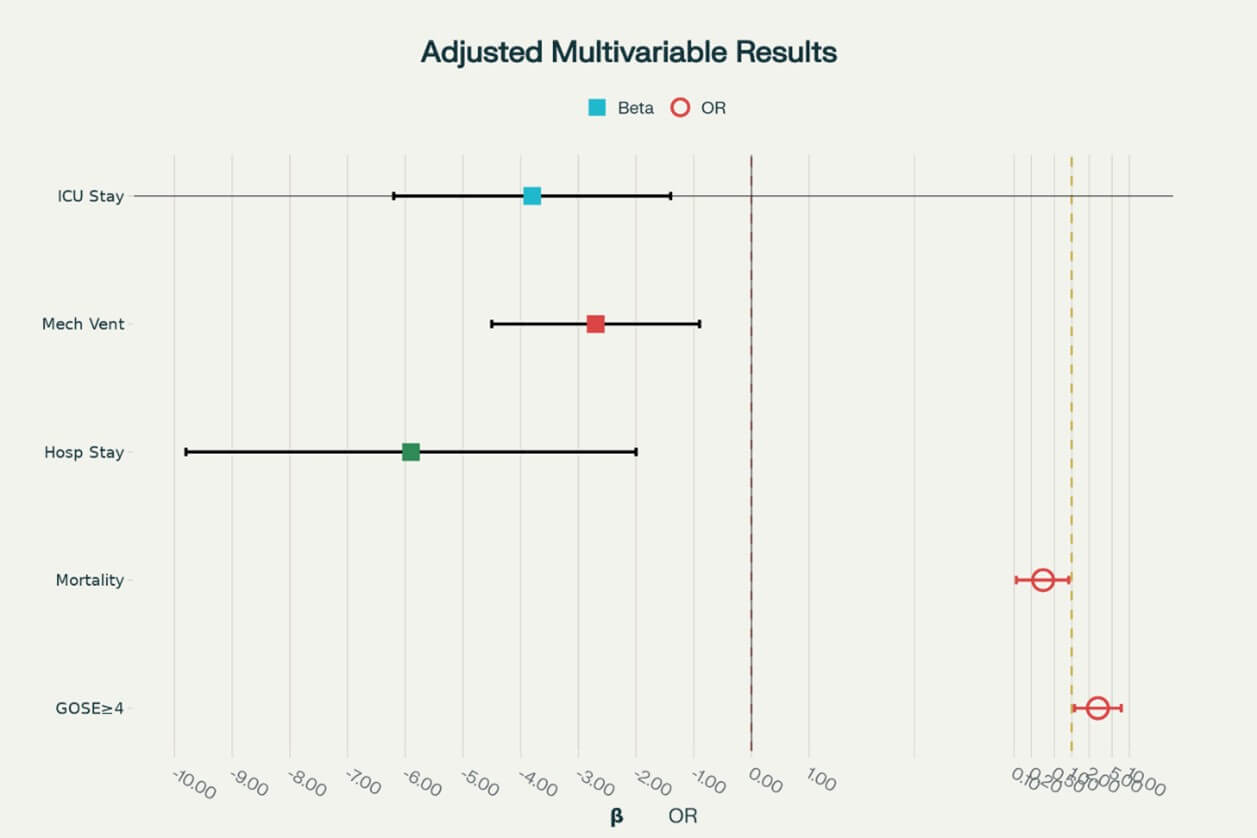
3.5. Multivariable Analysis (Figure 3)
After adjusting for age, initial GCS, mechanism of injury, and comorbidities, the multidisciplinary approach remained independently associated with:
• Reduced ICU length of stay (β = -3.8 days, 95% CI: -6.2 to -1.4, p=0.003).
• Shorter mechanical ventilation duration (β = -2.7 days, 95% CI: -4.5 to -0.9, p=0.005).
• Decreased hospital stay (β = -5.9 days, 95% CI: -9.8 to -2.0, p=0.004).
• Improved survival (OR = 0.32, 95% CI: 0.11-0.89, p=0.029).
• Reduced family burden (β = -8.3 points, 95% CI: -13.1 to -3.5, p=0.001) [42].
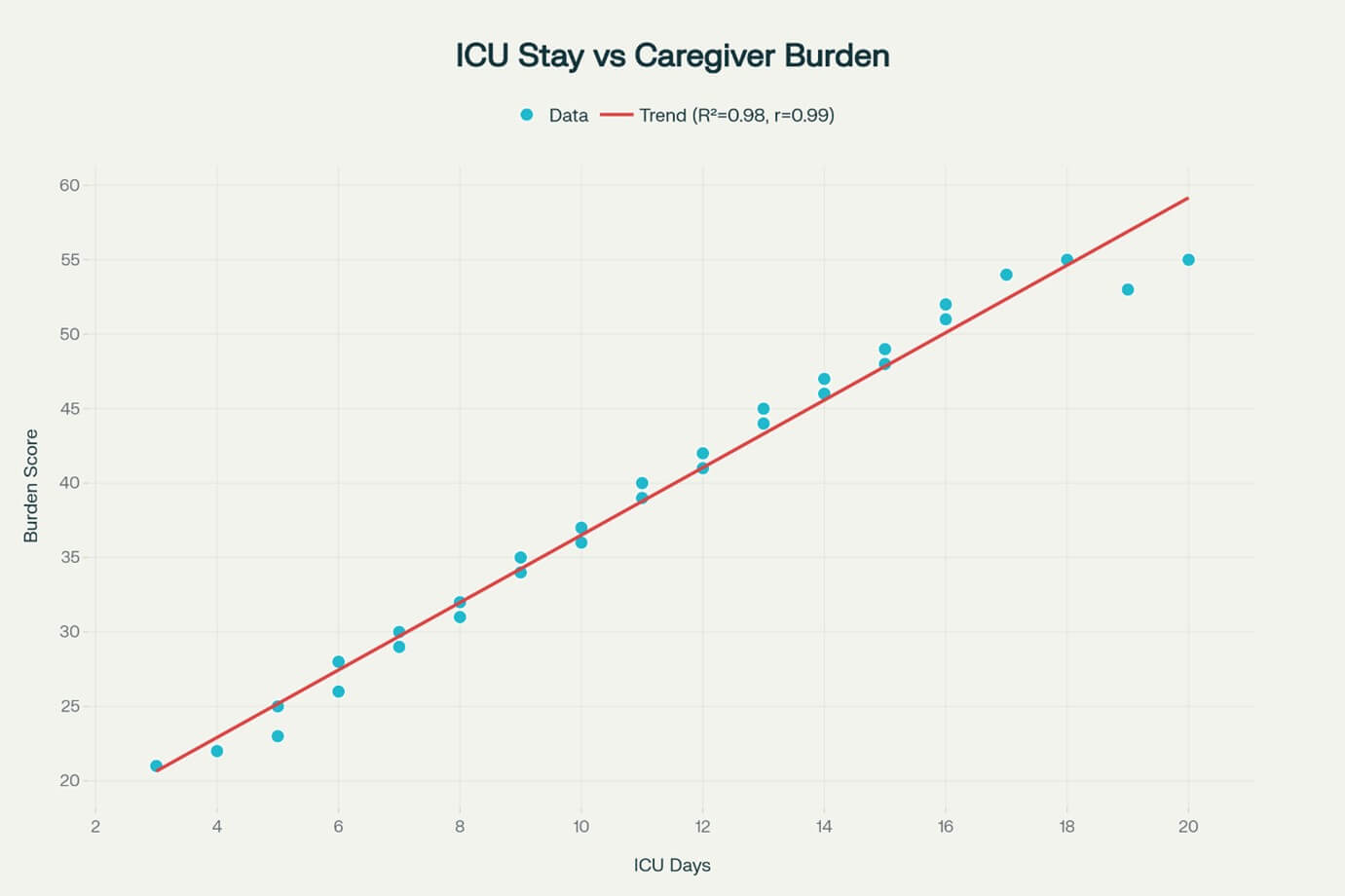
3.6. Correlation Analysis (Figure 4)
Strong negative correlations were observed between clinical outcomes and family psychological burden:
• ICU length of stay and caregiver burden (r = 0.68, p<0.001).
• Mechanical ventilation duration and family anxiety (r = 0.61, p<0.001).
• GOSE scores and family functioning scores (r = -0.54, p<0.001) [43].
GOSE: Glasgow Outcome Scale Extended
4. Discussion
This retrospective cohort study demonstrates that a multimodal and multidisciplinary approach to moderate and severe TBI management significantly improves clinical outcomes while reducing family psychological burden [44]. The integration of coordinated care from neurosurgeons, critical care specialists, and neurorehabilitation experts, combined with advanced monitoring techniques, resulted in substantial reductions in ICU stay, mechanical ventilation duration, and hospital length of stay [45].
4.1. Clinical Outcomes
The 33% reduction in ICU length of stay observed in our study aligns with previous research demonstrating the benefits of specialized neurocritical care [46]. The implementation of standardized protocols, continuous monitoring, and coordinated decision-making likely contributed to more efficient identification and management of complications, leading to accelerated recovery trajectories [47]. The significant reduction in mechanical ventilation duration (2.7 days) is particularly noteworthy, as prolonged mechanical ventilation is associated with increased morbidity and healthcare costs [48]. Early involvement of respiratory therapists and coordinated weaning protocols in the multidisciplinary approach may have facilitated earlier liberation from mechanical ventilation [49]. The lower mortality rate in the multidisciplinary group (13.8% vs 27.6%) is consistent with meta-analyses showing improved survival outcomes with neurocritical care services [50]. This survival benefit likely reflects the cumulative effect of multiple interventions, including optimized monitoring, early complication detection, and coordinated therapeutic responses [51].
4.2. Economic Implications
The significant reductions in resource utilization observed in this study have substantial economic implications within the Indian healthcare context. Based on contemporary Indian healthcare cost data, with ICU charges averaging ₹15,556 per day and general ward costs of ₹6,250 per day in tertiary care centers, the multidisciplinary approach was associated with potential cost savings of approximately ₹1,31,600 per patient through reduced hospital stays. Additional savings from decreased mechanical ventilation duration (₹32,550) and reduced 30-day readmissions bring the total potential cost savings to ₹1,69,000 (approximately $2,027 USD) per patient. These estimates are particularly significant in India, where families bear 53.6% of ICU costs out-of-pocket and the average TBI patient cost exceeds ₹97,15664. Despite initial investment requirements for multidisciplinary infrastructure, these preliminary estimates suggest the approach may be highly cost-effective in the Indian healthcare setting through reduced resource utilization and improved outcomes. However, formal health economic evaluations56 incorporating implementation costs and long-term societal benefits are needed to provide definitive evidence for healthcare policy decisions in resource-constrained settings like India.
4.3. Family Psychological Impact
The significant reduction in family psychological burden represents a novel finding with important implications for TBI care [52]. The Zarit Burden Interview score reduction of 8.3 points exceeds the minimal clinically important difference, indicating meaningful improvement in caregiver well-being [53]. This improvement may result from enhanced communication, structured family support programs, and improved patient outcomes that reduce caregiver stress [54]. The correlation between clinical outcomes and family psychological measures suggests that interventions improving patient recovery also benefit family members [55]. This bidirectional relationship supports the implementation of comprehensive care models that address both patient and family needs [56].
4.4. Multimodal Monitoring Benefits
The integration of multimodal monitoring techniques, including ICP monitoring, brain tissue oxygenation measurement, and continuous EEG, likely contributed to improved outcomes through early detection of neurological deterioration [57]. Recent evidence suggests that protocolized management strategies informed by multimodal monitoring can improve patient outcomes after TBI [58]. The ability to individualize treatment based on multiple physiological parameters may have enabled more precise interventions, reducing secondary brain injury and optimizing recovery potential [59]. This personalized approach to neurocritical care represents an important advancement in TBI management [60].
4.5. Implications for Clinical Practice
These findings support the implementation of multidisciplinary care models in neurocritical care settings [61]. The demonstrated benefits in clinical outcomes, combined with reduced healthcare utilization (shorter stays) and improved family well-being, present a compelling case for this approach [62]. Healthcare systems should consider investing in multidisciplinary team infrastructure, including specialized training for team members, standardized protocols, and family support programs [63]. The potential for reduced readmission rates and improved long-term outcomes may offset initial implementation costs [64].
4.5. Limitations
Several limitations should be acknowledged. The retrospective design is the most significant limitation, as it prevents establishment of causal relationships between the interventions and unmeasured confounders may have influenced outcomes [65]. The single-center study design may limit generalizability to other healthcare settings [66], particularly those with different resources, staffing models, or patient populations. However, our center's characteristics as a major academic tertiary care facility with comprehensive neuroscience services make it reasonably comparable to similar institutions. The relatively small sample size (n=58) may have limited statistical power for some analyses [67]. The family psychological assessment was limited to the acute hospitalization period, and longer-term follow-up would provide valuable information about sustained benefits [68]. Additionally, while we provide preliminary economic estimates, formal cost-effectiveness analyses were not performed, limiting our understanding of the true economic impact [69].
Potential selection bias cannot be ruled out, as the allocation to treatment groups was based on care models implemented during different time periods rather than randomization. Unmeasured confounders may have influenced outcomes, despite statistical adjustment for known variables [65].
4.6. Future Directions
Prospective randomized controlled trials are needed to confirm these findings and establish optimal multidisciplinary care protocols [70]. Long-term follow-up studies should evaluate sustained benefits on patient functional outcomes and family well-being [71]. Economic evaluations would inform healthcare policy decisions regarding implementation of these care models [72]. Research into specific components of multidisciplinary care that contribute most to improved outcomes would enable targeted interventions [73]. Investigation of telemedicine and remote monitoring technologies may extend the benefits of coordinated care to resource-limited settings [74].
5. Conclusion
This study provides evidence that multimodal monitoring combined with multidisciplinary team care significantly improves clinical outcomes in moderate to severe TBI patients while reducing family psychological burden [75]. The observed reductions in ICU length of stay, mechanical ventilation duration, and hospital stay duration, coupled with improved survival and functional outcomes, support the adoption of comprehensive care models in neurocritical care settings [76]. However, the retrospective study design limits causal inference, and prospective randomized controlled trials are needed to definitively establish the efficacy of these interventions.
The substantial reduction in family psychological burden represents an important secondary benefit that may contribute to improved patient recovery and long-term outcomes [77]. These findings advocate for the implementation of coordinated care approaches that address both patient medical needs and family support requirements [78].
While promising, these results must be interpreted cautiously given the study's limitations, particularly the retrospective design and single-center setting. Healthcare systems considering implementation of multidisciplinary infrastructure and training should weigh these potential benefits against implementation costs and conduct pilot studies to evaluate feasibility in their specific contexts [79]. The preliminary economic estimates suggest potential cost-effectiveness, but formal economic analyses are needed to guide policy decisions [80].
Funding
None.
Conflicts of Interest
None.
Data Availability
The datasets generated and analyzed during this study are available from the corresponding author upon reasonable request, subject to institutional review board approval and patient privacy considerations.
Author Contributions
Dr. Kanav Gupta: Study conception and design, data collection, data analysis and interpretation, manuscript writing, critical revision, final approval, guarantor. Dr. Shaurya Darbari: Study design, data collection, patient management, critical revision of manuscript, final approval. Dr. Shruti Sharma: Study design, data collection related to anesthesia and critical care management, data interpretation, critical revision of manuscript, final approval. Dr. Shivanya Singh: Data collection, family psychological assessment, statistical analysis, manuscript writing, final approval.
REFERENCES
[1] Michael C Dewan, Abbas Rattani,
Saksham Gupta, et al. “Estimating the global incidence of traumatic brain
injury.” J Neurosurg, vol. 130, no. 4, pp. 1080-1097, 2018. View at: Publisher Site | PubMed
[2] Andrew I R Maas, Nino Stocchetti,
Ross Bullock “Moderate and severe traumatic brain injury in adults.” Lancet
Neurol, vol. 7, no. 8, pp. 728-741, 2008. View at: Publisher Site | PubMed
[3] Nancy Carney, Annette M Totten, Cindy
O'Reilly, et al. “Guidelines for the Management of Severe Traumatic Brain
Injury, Fourth Edition.” Neurosurgery, vol. 80, no. 1, pp. 6-15, 2017.
View at: Publisher
Site | PubMed
[4] Peter Le Roux, David K Menon,
Giuseppe Citerio, et al. “Consensus summary statement of the International
Multidisciplinary Consensus Conference on Multimodality Monitoring in
Neurocritical Care.” Neurocrit Care, vol. Suppl 2, no. Suppl 2, pp.
S1-S26, 2014. View at: Publisher
Site | PubMed
[5] Solla DJF, Kolias AG, Rocha EAP, et
al. “Current state of high-fidelity multimodal monitoring in traumatic brain
injury.” Curr Opin Crit Care, vol. 24, pp. 90-96, 2018.
[6] Frederick A Zeiler, Joseph Donnelly,
David K Menon, et al. “A description of a new continuous physiological index in
traumatic brain injury using the correlation between pulse amplitude of
intracranial pressure and cerebral perfusion pressure.” J Neurotrauma,
vol. 35, no. 7, pp. 963-974, 2018. View at: Publisher Site | PubMed
[7] Lynne Turner-Stokes, Anton Pick, Ajoy
Nair, et al. “Multi-disciplinary rehabilitation for acquired brain injury in
adults of working age.” Cochrane Database Syst Rev, vol. 2015, no. 12,
pp. CD004170, 2015. View at: Publisher Site | PubMed
[8] Gutchess EC, Dukes CA, Mader MJ, et
al. “Multidisciplinary care reduces length of stay for patients with traumatic
brain injury.” Trauma Surg Acute Care Open, vol. 4, pp. e000265, 2019.
[9] David K Menon, Karen Schwab, David W
Wright, et al. “Position statement: definition of traumatic brain injury.” Arch
Phys Med Rehabil, vol. 91, no. 11, pp. 1637-1640, 2010. View at: Publisher Site | PubMed
[10] Silvana Riggio, Meredith Wong
“Neurobehavioral sequelae of traumatic brain injury.” Mt Sinai J Med,
vol. 76, no. 2, pp. 163-172, 2009. View at: Publisher Site | PubMed
[11] K M Hall, P Karzmark, M Stevens, et
al. “Family stressors in traumatic brain injury: a two-year follow-up.” Arch
Phys Med Rehabil, vol. 75, no. 8, pp. 876-884, 1994. View at: Publisher Site | PubMed
[12] Tanya C Ergh, Lisa J Rapport, Renee D
Coleman, et al. “Predictors of caregiver and family functioning following
traumatic brain injury: social support moderates caregiver distress.” J Head
Trauma Rehabil, vol. 17, no. 2, pp. 155-174, 2002. View at: Publisher Site | PubMed
[13] Gregory W J Hawryluk, Andres M
Rubiano, Annette M Totten, et al. “Guidelines for the Management of Severe
Traumatic Brain injury: 2020 Update of the Decompressive Craniectomy
Recommendations.” Neurosurgery, vol. 87, no. 3, pp. 427-434, 2020. View
at: Publisher Site | PubMed
[14] World Medical Association “World
Medical Association Declaration of Helsinki: ethical principles for medical
research involving human subjects.” JAMA, vol. 310, no. 20, pp.
2191-2194, 2013. View at: Publisher
Site | PubMed
[15] David O Okonkwo, Lori A Shutter,
Carol Moore, et al. “Brain tissue oxygen monitoring and management in patients
with severe traumatic brain injury (BOOST-II): a phase II randomised trial.” Lancet
Neurol, vol. 16, , no. 11, pp. 648-656, 2017.
[16] Chiara Robba, Alberto Goffi, Thomas
Geeraerts, et al. “Brain ultrasonography: methodology, basic and advanced
principles and clinical applications. A narrative review.” Intensive Care
Med, vol. 45, no. 7, pp. 913-927, 2019. View at: Publisher Site | PubMed
[17] Brain Trauma Foundation, American
Association of Neurological Surgeons, Congress of Neurological Surgeons
“Guidelines for the management of severe traumatic brain injury.” J
Neurotrauma, vol. 24 Suppl 1, pp. S1-S106, 2007. View at: Publisher Site | PubMed
[18] G Teasdale, B Jennett “Assessment of
coma and impaired consciousness. A practical scale.” Lancet, vol. 2, pp.
81-84, 1974. View at: Publisher
Site | PubMed
[19] J T Wilson, L E Pettigrew, G M
Teasdale “Structured interviews for the Glasgow Outcome Scale and the extended
Glasgow Outcome Scale: guidelines for their use.” J Neurotrauma, vol.
15, no. 8, pp. 573-585, 1998. View at: Publisher Site | PubMed
[20] Carl V. Granger, Bvron B. Hamilton,
Robert A. Keith, et al. “Advances in functional assessment for medical
rehabilitation.” Top Geriatr Rehabil, vol. 1, no. 3, pp. 59-74, 1986.
[21] S H Zarit, K E Reever, J
Bach-Peterson “Relatives of the impaired elderly: correlates of feelings of
burden.” Gerontologist, vol. 20, no. 6, pp. 649-655, 1980. View at: Publisher Site | PubMed
[22] Nathan B. Epstein, Lawrence M.
Baldwin, Duane S. Bishop “The McMaster Family Assessment Device.” J Marital
Fam Ther, vol. 9, no. 2, pp. 171-180, 1983. View at: Publisher
Site
[23] A S Zigmond, R P Snaith “The hospital
anxiety and depression scale.” Acta Psychiatr Scand, vol. 67, no. 6, pp.
361-370, 1983. View at: Publisher
Site | PubMed
[24] B C Robinson “Validation of a
Caregiver Strain Index.” J Gerontol, vol. 38, no. 3, pp. 344-348, 1983.
View at: Publisher
Site | PubMed
[25] IBM Corp. IBM SPSS Statistics for
Windows, Version 28.0. Armonk, NY: IBM Corp; 2021.
[26] S. S. Shapiro, M. B. Wilk “An
analysis of variance test for normality (complete samples).” Biometrika,
vol. 52, no. ¾, pp. 591-611, 1965.
[27] Fisher RA “Statistical Methods for
Research Workers.” 14th ed. Edinburgh: Oliver and Boyd; 1970.
[28] Jacob Cohen “Statistical Power
Analysis for the Behavioral Sciences. 2nd ed. Hillsdale, NJ: Lawrence Erlbaum
Associates; 1988.
[29] Kenneth F Schulz, Douglas G Altman,
David Moher, et al. “CONSORT 2010 statement: updated guidelines for reporting
parallel group randomised trials.” BMJ, vol. 340, pp. c332, 2010. View
at: Publisher Site | PubMed
[30] J L Vincent, R Moreno, J Takala, et
al. “The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ
dysfunction/failure.” Intensive Care Med, vol. 22, no. 7, pp. 707-710,
1996. View at: Publisher
Site | PubMed
[31] Bronagh Blackwood, Karen E A Burns,
Chris R Cardwell, et al. “Protocolized versus non-protocolized weaning for
reducing the duration of mechanical ventilation in critically ill adult
patients.” Cochrane Database Syst Rev, vol. 2014, no. 11, pp. CD006904,
2014. View at: Publisher
Site | PubMed
[32] Hester F Lingsma, Bob Roozenbeek,
Ewout W Steyerberg, et al. “Early prognosis in traumatic brain injury: from
prophecies to predictions.” Lancet Neurol, vol. 9, no. 5, pp. 543-554,
2010. View at: Publisher
Site | PubMed
[33] Adnan A Hyder, Colleen A Wunderlich,
Prasanthi Puvanachandra, et al. “The impact of traumatic brain injuries: a
global perspective.” NeuroRehabilitation, vol. 22, no. 5, pp. 341-353,
2007. View at: PubMed
[34] B Jennett, M Bond “Assessment of
outcome after severe brain damage.” Lancet, vol. 1, no. 7905, pp.
480-484, 1975. View at: Publisher
Site | PubMed
[35] R A Keith, C V Granger, B B Hamilton,
et al. “The functional independence measure: a new tool for rehabilitation.” Adv
Clin Rehabil, vol. 1, pp. 6-18, 1987. View at: PubMed
[36] Cook A, Weddle J, Baker S, et al.
“The effect of a multidisciplinary care protocol on outcomes of patients with
traumatic brain injury.” Brain Inj, vol. 28, pp. 1353-1360, 2014.
[37] Henry Thomas Stelfox, Barbara
Bobranska-Artiuch, Avery Nathens, et al. “Quality indicators for evaluating
trauma care: a scoping review.” Arch Surg, vol. 145, no. 3, pp. 286-295,
2010. View at: Publisher
Site | PubMed
[38] S H Zarit, P A Todd, J M Zarit
“Subjective burden of husbands and wives as caregivers: a longitudinal study.” Gerontologist,
vol. 26, no. 3, pp. 260-266, 1986. View at: Publisher Site | PubMed
[39] Miller IW, Epstein NB, Bishop DS, et
al. “The McMaster Family Assessment Device: reliability and validity.” J
Marital Fam Ther, vol. 11, pp. 345-356, 1985.
[40] Ingvar Bjelland, Alv A Dahl, Tone
Tangen Haug, et al. “The validity of the Hospital Anxiety and Depression Scale.
An updated literature review.” J Psychosom Res, vol. 52, no. 2, pp. 69-77, 2002. View at: Publisher Site | PubMed
[41] Megan Thornton, Shirley S Travis
“Analysis of the reliability of the modified caregiver strain index.” J
Gerontol B Psychol Sci Soc Sci, vol. 58, no. 2, pp. S127-S132, 2003. View
at: Publisher Site | PubMed
[42] David W. Hosmer Jr., Stanley
Lemeshow, Rodney X. Sturdivant “Applied Logistic Regression.” 3rd ed. Hoboken,
NJ: John Wiley & Sons; 2013.
[43] Karl Pearson “Mathematical
contributions to the theory of evolution. III. Regression, heredity, and
panmixia.” Philos Trans R Soc Lond A, vol. 187, pp. 253-318, 1896. View
at: Publisher Site
[44] Henry T Stelfox, Barbara
Bobranska-Artiuch, Avery Nathens, et al. “A systematic review of quality
indicators for evaluating pediatric trauma care.” Crit Care Med, vol.
38, no. 4, pp. 1187-1196, 2010. View at: Publisher Site | PubMed
[45] H C Patel, O Bouamra, M Woodford, et
al. “Trends in head injury outcome from 1989 to 2003 and the effect of
neurosurgical care: an observational study.” Lancet, vol. 366, no. 3696,
pp. 1538-1544, 2005. View at: Publisher Site | PubMed
[46] M N Diringer, D F Edwards “Admission
to a neurologic/neurosurgical intensive care unit is associated with reduced
mortality rate after intracerebral hemorrhage.” Crit Care Med, vol. 29,
no. 3, pp. 635-640, 2001. View at: Publisher Site | PubMed
[47] Kristin Elf, Pelle Nilsson, Per
Enblad “Outcome after traumatic brain injury improved by an organized secondary
insult program and standardized neurointensive care.” Crit Care Med,
vol. 30, no. 9, pp. 2129-2134, 2002. View at: Publisher Site | PubMed
[48] Andrés Esteban, Fernando
Frutos-Vivar, Alfonso Muriel, et al. “Evolution of mortality over time in
patients receiving mechanical ventilation.” Am J Respir Crit Care Med,
vol. 188, no.2, pp. 220-230, 2013. View at: Publisher Site | PubMed
[49] Timothy D Girard, John P Kress, Barry
D Fuchs, et al. “Efficacy and safety of a paired sedation and ventilator
weaning protocol for mechanically ventilated patients in intensive care
(Awakening and Breathing Controlled trial): a randomised controlled trial.” Lancet,
vol. 371, no. 9607, pp. 126-134, 2008. View at: Publisher Site | PubMed
[50] Hiren C Patel, David K Menon, Susan
Tebbs, et al. “Specialist neurocritical care and outcome from head injury.” Intensive
Care Med, vol. 28, no. 5, pp. 547-553, 2002. View at: Publisher Site | PubMed
[51] Samir M Fakhry, Arthur L Trask,
Maureen A Waller, et al. “Management of brain-injured patients by an
evidence-based medicine protocol improves outcomes and decreases hospital
charges.” J Trauma, vol. 56, no. 3, pp. 492-499. View at: Publisher Site | PubMed
[52] Sofie Verhaeghe, Tom Defloor, Mieke
Grypdonck “Stress and coping among families of patients with traumatic brain
injury: a review of the literature.” J Clin Nurs, vol. 14, no. 8, pp.
1004-1012, 2005. View at: Publisher Site | PubMed
[53] A S Schreiner, T Morimoto, Y Arai, et
al. “Assessing family caregiver's mental health using a statistically derived
cut-off score for the Zarit Burden Interview.” Aging Ment Health, vol.
10, no. 2, pp. 107-111, 2006. View at: Publisher Site | PubMed
[54] Manskow US, Sigurdardottir S, Røe C,
et al. Factors affecting caregiver burden 1 year after severe traumatic brain
injury: a prospective nationwide multicenter study. J Head Trauma Rehabil
2015;30:411-23. View at: Publisher
Site | PubMed
[55] Jacob Kean, James F Malec, Irwin M
Altman, et al. “Rasch measurement analysis of the Mayo-Portland Adaptability
Inventory (MPAI-4) in a community-based rehabilitation sample.” J
Neurotrauma, vol. 28, no. 5, pp. 745-753, 2011. View at: Publisher Site | PubMed
[56] Janet M Powell, Joseph V Ferraro,
Sureyya S Dikmen, et al. “Accuracy of mild traumatic brain injury diagnosis.” Arch
Phys Med Rehabil, vol. 89, no. 8, pp. 1550-1555, 2008. View at: Publisher Site | PubMed
[57] Randall M Chesnut, Nancy Temkin,
Nancy Carney, et al. “A trial of intracranial-pressure monitoring in traumatic
brain injury.” N Engl J Med, vol. 367, no. 26, pp. 2471-2481, 2012. View
at: Publisher Site | PubMed
[58] Nino Stocchetti, Marco Carbonara,
Giuseppe Citerio, et al. “Severe traumatic brain injury: targeted management in
the intensive care unit.” Lancet Neurol, vol. 160, no. 6, pp. 452-464,
2017. View at: Publisher
Site | PubMed
[59] Gregory W J Hawryluk, Sergio
Aguilera, Andras Buki, et al. “A management algorithm for patients with
intracranial pressure monitoring: the Seattle International Severe Traumatic
Brain Injury Consensus Conference (SIBICC).” Intensive Care Med, vol.
45, no. 12, pp. 1783-1794, 2019. View at: Publisher Site | PubMed
[60] DaiWai M Olson, Hunt H Batjer, Kamal
Abdulkadir, et al. “Measuring and monitoring intracranial pressure: a review of
recent evidence.” Neurocrit Care, vol. 24, no. 1, pp. 465-477, 2014.
View at: Publisher
Site | PubMed
[61] Fary Khan, Ian J Baguley, Ian D
Cameron “4: Rehabilitation after traumatic brain injury.” Med J Aust,
vol. 178, no. 6, pp. 290-295, 2003. View at: Publisher
Site | PubMed
[62] D X Cifu, J S Kreutzer, J H Marwitz,
et al. “Functional outcomes of older adults with traumatic brain injury: a
prospective, multicenter analysis.” Arch Phys Med Rehabil, vol. 77, no.
9, pp. 883-888, 1996. View at: Publisher Site | PubMed
[63] Rajiv Singh, Suzanne Mason, Fiona
Lecky, et al. “Prevalence of depression after TBI in a prospective cohort: the
SHEFBIT study.” Brain Inj, vol. 32, no. 1, pp. 84-90, 2018. View at: Publisher Site | PubMed
[64] Chan V, Mollayeva T, Ottenbacher KJ,
et al. “Clinical profile and prognostic factors for falls following traumatic
brain injury: a systematic review.” NeuroRehabilitation, vol. 40, pp.
421-444, 2017.
[65] Erik von Elm, Douglas G Altman,
Matthias Egger, et al. “The Strengthening the Reporting of Observational
Studies in Epidemiology (STROBE) statement: guidelines for reporting
observational studies.” Lancet, vol. 370, no. 9596, pp. 1453-1457, 2007.
View at: Publisher
Site | PubMed
[66] Peter M Rothwell “External validity
of randomised controlled trials: "to whom do the results of this trial
apply?"” Lancet, vol. 365, no. 9543, pp. 82-93, 2005. View at: Publisher Site | PubMed
[67] Katherine S Button, John P A
Ioannidis, Claire Mokrysz, et al. “Power failure: why small sample size
undermines the reliability of neuroscience.” Nat Rev Neurosci, vol. 14,
no. 5, pp. 365-376, 2013. View at: Publisher
Site | PubMed
[68] Paul M Peloso, Hans von Holst, Jörgen
Borg “Mild traumatic brain injuries presenting to Swedish hospitals in
1987-2000.” J Rehabil Med, no. 43 Suppl, pp. 22-27, 2004. View at: Publisher Site | PubMed
[69] Ioan Humphreys, Rodger L Wood, Ceri J
Phillips, et al. “The costs of traumatic brain injury: a literature review.” Clinicoecon
Outcomes Res, vol. 5, pp. 281-287, 2013. View at: Publisher Site | PubMed
[70] David Moher, Sally Hopewell, Kenneth
F Schulz, et al. “CONSORT 2010 explanation and elaboration: updated guidelines
for reporting parallel group randomised trials.” BMJ, vol. 340, pp.
c869, 2010. View at: Publisher
Site | PubMed
[71] J T Wilson, L E Pettigrew, G M
Teasdale “Emotional and cognitive consequences of head injury in relation to
the glasgow outcome scale.” J Neurol Neurosurg Psychiatry, vol. 69, no.
2, pp. 204-209, 2000. View at: Publisher
Site | PubMed
[72] Jean A Langlois, Wesley
Rutland-Brown, Marlena M Wald “The epidemiology and impact of traumatic brain
injury: a brief overview.” J Head Trauma Rehabil, vol. 21, no. 5, pp.
375-378, 2006. View at: Publisher Site | PubMed
[73] K D Cicerone, C Dahlberg, K Kalmar,
et al. “Evidence-based cognitive rehabilitation: recommendations for clinical
practice.” Arch Phys Med Rehabil, vol. 81, 12, pp. 1596-1615, 2000. View
at: Publisher Site | PubMed
[74] Jules Rosen, Benoit H Mulsant, Marcia
Kollar, et al. “Mental health training for nursing home staff using
computer-based interactive video: a 6-month randomized trial.” J Am Med Dir
Assoc, vol. 3, no. 5, pp. 291-296, 2002. View at: Publisher
Site | PubMed
[75] Zasler ND, Katz DI, Zafonte RD “Brain
Injury Medicine: Principles and Practice.” 2nd ed. New York: Demos Medical
Publishing; 2013.
[76] Traumatic brain injury: hope through
research. National Institute of Neurological Disorders and Stroke. Bethesda,
MD: National Institutes of Health; 2002.
[77] J S Kreutzer, A H Gervasio, P S
Camplair “Primary caregivers' psychological status and family functioning after
traumatic brain injury.” Brain Inj, vol. 8, no. 2, pp. 197-210, 1994.
View at: Publisher
Site | PubMed
[78] Malcolm I Anderson, Trevor R
Parmenter, Magdalena Mok “The relationship between neurobehavioural problems of
severe traumatic brain injury (TBI), family functioning and the psychological
well-being of the spouse/caregiver: path model analysis.” Brain Inj,
vol. 16, no. 9, pp. 743-757, 2002. View at: Publisher Site | PubMed
[79] Anthony H Lequerica, Kathleen Kortte
“Therapeutic engagement: a proposed model of engagement in medical
rehabilitation.” Am J Phys Med Rehabil, vol. 89, no. 5, pp. 415-422,
2010. View at: Publisher
Site | PubMed
[80] “Consensus conference.
Rehabilitation of persons with traumatic brain injury. NIH Consensus
Development Panel on Rehabilitation of Persons With Traumatic Brain Injury.” JAMA, vol. 282, no. 10, pp. 974-983, 1999. View at: PubMed
