Received: Mon 24, Nov 2025
Accepted: Tue 16, Dec 2025
Abstract
Objective: Glioblastoma (GB) are aggressive and terminal brain tumors. Given the limited evidence of benefit from highly invasive treatments in elderly patients, it is important to evaluate patient centered outcomes to guide practice and avoid harm. This study assesses next of kin (NOK) perspectives on surgical outcomes for GB treatment in elderly patients.
Methods: Next of kin of all patients 60 years and older who underwent surgical intervention for a histologically confirmed GB at a single center between February 2010 and November 2018 were sent a post-mortem questionnaire about the outcomes of their loved ones’ treatment. Questions addressed a range of pre-operative processes and post-operative outcomes. Responses were analyzed thematically and for statistical association with days of functional independence (FI) and days to death. A reproducibility study assessed questionnaire response reliability.
Results: Of 206 eligible participants, 124 NOK responded (60%). Patients felt well-informed and outcomes met expectations set pre-operatively. Responses demonstrated widespread variability as to whether treatments improved symptoms or enabled patients to retain a sufficient quality of life. There was an observable trend that patients were less capable post-operatively, and a clear majority did not return to their baseline FI. Despite the variability in clinical improvement, most respondents would make the same decision again but were less inclined to advise another loved one to undergo the same treatment. Overall, NOK of patients who maintained FI responded more positively than those with prolonged survival alone. The reproducibility study indicated 86% reliability in responses by our measure.
Conclusion: This study finds limited evidence that surgery enables elderly GB patients to retain a modest quality of life prior to death and observes that post-operative morbidity is valued over prolonged survival. Our recommendation is for pre-operative discussions to include post-operative morbidity to help facilitate care that is in keeping with patients’ priorities.
Keywords
Elderly, Glioblastoma, Morbidity, Mortality, Next of kin, Quality of life
1. Introduction
Glioblastoma (GB) is the most common malignant primary brain cancer in adults. The gold standard treatment consists of maximal safe surgical resection and radiotherapy with concurrent and adjuvant temozolomide [1]. This Stupp protocol provides a median survival of 14.6 months in young healthy patients [1]. However, the majority of patients with GBs are neither young, nor healthy. The median age at diagnosis is 66 with peak incidence between 75 - 84 and traditionally, patients over 70 have been excluded from clinical trials [2-4]. In a study where elderly comorbid patients 70 years and older were included, those patients had a median post-operative survival as low as 4.5 months [5]. Clinical status is also a strong prognostic factor for post-operative survival, but elderly cohorts with low Karnofsky Performance Status (KPS) scores (< 70) have also most often been excluded from trials [6]. Despite diminishing returns, the Stupp protocol continues to be utilized in elderly and comorbid populations.
Gross total resection (GTR) and chemoradiotherapy are highly invasive treatment options that have the potential to increase physical and psychological stress for the patient, in addition to their tumor burden. Dr. Atul Gawande wrote in Being Mortal that “Our most cruel failure in how we treat the sick and the aged is the failure to recognize that they have priorities beyond merely…living longer” [7]. Given the potential for serious harm, it is imperative to assess patients’ and their family members’ views of treatment options to ensure care is tailored to their priorities, and importantly, minimizes harm.
Health Related Quality of Life (HRQOL) questionnaires assess the morbidity of treatments. Only a small number of studies have been published looking at the impact of surgical treatment on HRQOL in GBs. In 2011, Jakola [8] found that improved HRQOL was not a probable post-operative outcome and, furthermore, that surgery-related deficits worsen HRQOL. In addition, neither the extent of resection nor use of modern surgical techniques altered outcomes. While these findings are pertinent, patients were only assessed pre-operatively and once at six weeks post-operatively, making it difficult to ascertain the overall impact of treatment. In 2016 a smaller prospective longitudinal Norwegian study measured pre- and post-operative HRQOL in 30 participants with GBs over a year [9]. It found a correlation between progression-free survival and HRQOL. Most recently, Laigle-Donadey [10] conducted a randomized controlled trial comparing biopsy to GTR in 109 patients over 70 years of age. Patients were followed until death with monthly reviews of KPS and mental state, and bimonthly assessments of QOL and radiologic progression. While there was no significant difference in overall survival between groups, those in the GTR group had significantly improved QOL and progression free survival.
Despite the gradually increasing focus on HRQOL, the evidence base remains small. Furthermore, patients' views of their treatments have been largely anecdotal. There is no literature systematically investigating the patients’ perspective from diagnosis to death. This study investigates the views of next of kin (NOK) of deceased, elderly patients who underwent surgical intervention for a GB in order to learn about the patients’, carers’ and families’ pre- and post-operative experiences through their entire journey.
2. Materials & Methods
2.1. Study Population and Participants
A single-center observational study was performed using a closed questionnaire analysis of NOK responses. NOK of deceased patients who were aged 60 years and older when operated on at the Department of Neurosurgery at Wellington Regional Hospital (WRH; Wellington, New Zealand) between February 2010 and November 2018 were recruited. For inclusion, patients needed to have had a histologically confirmed diagnosis of a supratentorial glioblastoma from a biopsy, sub-total or gross total resection, without a concurrent cause of death. Prospective NOK participants were identified using WRH’s patient management system or via patients’ general practitioners and were approached by letter with an information sheet about the study, a consent form, the study questionnaire, and a pre-paid return envelope. No monetary or alternative compensation was offered. No psychological support was made available outside of the counselling participants may have received by the respective Palliative Care Teams around the time of their loved one’s decline and death.
Six months following the first questionnaire, approximately half of respondents were mailed a second re-randomized questionnaire to assess reproducibility, i.e., that the questions were unambiguous and elicited the same responses and that, upon further reflection, NOKs’ perspectives (and their recall of their loved one’s experiences and opinions of their treatments) remained unchanged. Again, no incentives were supplied. This study was approved by the Capital & Coast District Health Board (CCDHB) Clinical Audit and Research Committee, the CCDHB Research Advisory Group Māori, and the New Zealand Ministry of Health - Health and Disability Ethics Committee.
2.2. Functional Independence
For this study’s purposes, functional independence (FI) is defined as independence with activities of daily living (ADLs). ADLs are six fundamental skills required to care for oneself [11]. These skills include the ability to feed, dress, bathe, and toilet oneself, mobilize independently and retain urinary and bowel continence. Post-operatively, all patients were assessed by occupational therapists or physiotherapists to determine whether they had retained or regained FI. Upon achieving independence with ADLs, they were discharged home and that was considered Day 1 of FI. WRH, local hospital, clinic, rest home, general practice and hospice records were reviewed to determine the date of the first documented evidence that the patient had lost their FI. The previous day was then considered their last day of FI. Any transient losses of FI, such as seizure activity requiring hospitalization, were subtracted from the patient’s total length of FI. Patients who did not retain or regain FI post-operatively were recorded as having zero days of FI.
2.3. NOK Questionnaire & Pilot Study
A novel questionnaire was developed by clinicians from the Neurosurgery, Radiation Oncology, Palliative Care, Pathology and Neuroradiology Departments at WRH. Questions were in the form of paired positively and negatively worded statements, each requiring a Likert scale response. Nine questions were agreed upon covering a range of patient and NOK experiences such as whether they (patient and NOK) received sufficient information during the informed consent process, whether the patient’s symptoms improved post-operatively or post-all treatments i.e., surgery, chemoradiotherapy, and rehabilitation, and whether the NOK believed their loved one would retrospectively elect to have surgery again. A pair of statements was used for each question to reduce bias from leading statements and to allow for internal validation of responses.
To identify any poorly worded or ambiguous statements, a pilot study on the questionnaire was first conducted. Without regard to their age, gender or disease type, the NOK of ten randomly selected inpatients who were undergoing treatment at the WRH Neurosurgery Department in April 2020 were approached. Verbal consent was obtained from visiting NOK; they were asked to read the questionnaire independently, then to rephrase the statements to RR in their own words. This pilot study was performed to ascertain whether the NOKs’ understanding of each statement agreed with the researchers’ intended enquiry. Responses resulted in the rewording of two of the nine statement pairs. After the 18 statements were formalized, six different questionnaires were created by randomizing the order of the statements, under the constraint that paired statements were to be separated. Each NOK then received one of the questionnaires chosen at random. Table 1 for the complete list of statements.
Table. 1. List of paired statements.
|
P1 - Your loved one and their
family got enough information about the risks and benefits of surgery before
the operation |
|
N1
- Your loved one and their family did not get enough information about the
risks and benefits of surgery before the operation |
|
P2 - You thought your loved one’s
symptoms (e.g. headache or confusion) got better after surgery. |
|
N2
- You thought your loved one’s symptoms (e.g. headache or confusion) got
worse after surgery. |
|
P3 - Your loved one was able to do
more (e.g. dressing, showering, preparing a cup of tea etcetera) after
surgery than before. |
|
N3
- Your loved one needed more help (e.g. dressing, showering, preparing a cup
of tea etcetera) after surgery than before. |
|
P4 - After all treatments (e.g.
surgery, rehabilitation and/or chemoradiotherapy), your loved one was able to
do most of the same activities as before having cancer. |
|
N4
- After all treatments (e.g. surgery, rehabilitation and/or
chemoradiotherapy), your loved one was not able to do most of the same
activities as before having cancer. |
|
P5 - The outcome of surgery met
your and your loved one’s hopes. |
|
N5
- The outcome of surgery did not meet your and your loved one’s hopes. |
|
P6 - If you could go back in time,
you would make the same decision again. You would encourage your loved one to
have surgery. |
|
N6
- If you could go back in time, you would not make the same decision again.
Instead, you would discourage your loved one to have surgery. |
|
P7 - You would recommend surgery to
another family member or friend with the same brain cancer. |
|
N7
- You would not recommend surgery to another family member or friend with the
same brain cancer. |
|
P8 - Treatment (e.g. surgery,
rehabilitation and/or chemoradiotherapy) helped your loved one stay well
enough to enjoy their remaining life with family and friends. |
|
N8
- After treatment (e.g. surgery, rehabilitation and/or chemoradiotherapy),
your loved one struggled to enjoy their remaining life with family and
friends. |
|
P9 - The outcome of surgery fitted
with what you were told to expect by the surgeons. |
|
N9
- The outcome of surgery did not fit with what you were told to expect by the
surgeons |
|
Note: P = positively phrased
statement, N = negatively phrased statement |
In addition to the paired statements, one question and one statement were shared with the NOK, and space was provided for written responses. The question was “What do you know now that you wish you had known at the start of your loved one’s treatment(s)?” and the statement was “Any additional comments to add to this study would be appreciated.”
2.4. Volumetric Assessment of Tumors
Tumor volumes were assessed with 3D Slicer 4.11 software and the segment editor and quantification/segment statistics features. Magnetic Resonance Imaging (MRI) scans uploaded into a Picture Archiving and Communications Software (PACS) package were used. Preference was given to post-contrast axial T1 images, but any post-contrast T1 Stealth, fluid-attenuated inversion recovery or T2-weighted axial images were used if necessary.
2.5. Analysis
The Likert scale for each statement contained five possible answers: completely disagree, disagree somewhat, neutral, agree somewhat, and completely agree, which were scored from 0 to 4 respectively. To analyze responses, the negatively worded statements were reverse-scored from 0 to 4. Subsequently, any scores from a statement pair that differed by 2 or more were deemed unreliable and disregarded, i.e., treated as missing. For each of the nine questions, the scores from the pair of statements were then summed to give a combined score ranging from 0 to 8, meaning that the positively and negatively worded statements were given equal weight. The combined score is referred to as the NOK ‘response’ for that question. In accordance with the Likert scale, responses of 0, 2, 4, 6, and 8 are interpreted as ‘completely disagree,’ ‘disagree somewhat,’ ‘neutral,’ ‘agree somewhat,’ and ‘completely agree,’ respectively. Responses of 1, 3, 5, and 7 represent occasions when scores from a question pair differed by 1 and have not been given specific labels.
For each of the nine questions, the strengths of the association between NOK response and days to death (DTD) and the association between NOK response and days of functional independence (DFI) were assessed using Kendall’s rank correlation coefficient (Kendall’s tau). Kendall’s tau is the most appropriate correlation coefficient for inference on a meaningful population parameter with ordinal data [12]. In the reproducibility study, the NOK response from the second questionnaire was subtracted from the response in the first questionnaire. No data points were excluded even if the respondent clearly misread a statement. A Wilcoxon signed-rank test was used to assess differences.
2.6. Post-hoc Analyses
The strength of association between surgery type (biopsy versus resection) and NOK responses for all questions unrelated to consent (Q2-8) were examined graphically and two-sided Wilcoxon-Mann-Whitney tests were carried out. The p values were adjusted for multiple comparisons by the Free Step-Down Resampling Method [13]. In a separate post-hoc analysis NOK responses to the three questions (Q2-4) pertaining to post-operative clinical function were averaged for testing for the existence of an association with pre-operative KPS.
3. Results
3.1. Patient Characteristics
Of 206 eligible participants, 124 NOK responded (60%). The characteristics of the patients of the NOK are summarized in (Table 2). Patients had a mean age of 68.8 years (SD ± 6.4) and an age range of 60 - 86 years. There were 70 men (56%) and 54 women (44%). The median pre-operative KPS was 60 (IQR 40 - 80). Twenty patients (16%) had multifocal tumors, and 13 (10%) had bilateral tumors. Tumor volumes were available for 121 patients and median tumor volume was 34.4 cm3 (IQR 13.3 - 61.4 cm3). Total gross resections, subtotal resections and biopsies were performed in 37 (30%), 69 (56%), and 18 (15%) patients, respectively. Post-operatively, 76 patients (61%) received chemoradiotherapy and a further 23 (19%) had radiotherapy alone. Patients maintained FI for a median of 62 days, which improved to 99 days if they received a sub or gross total resection, but fell to zero if they had a biopsy. Median DTD for the entire cohort, resection subcohort and biopsy subcohort were 278, 319 and 118 days, respectively.
3.2. NOK Questionnaire
Next of kin responses by question are shown in (Figure 1), where each question (Q) is represented by an abridged positively worded phrase. The majority of NOK agreed somewhat or completely agreed that the patient and their family received enough information during the informed consent process (Q1) and that the outcome of surgery fit with what the surgeon told them to expect (Q9). While the surgical results were as expected, there was no NOK consensus on whether the outcomes were as they and the patient had hoped (Q5). Widespread variability in NOK responses was also observed when asked if the patient’s symptoms improved post-op (Q2) or if treatments allowed the patient to stay well enough to enjoy their remaining time with family and friends (Q8). There was an observable trend that NOK completely disagreed or disagreed somewhat that patients were more capable post-op (Q3) and a clear majority indicated that patients did not regain most of their baseline functioning post-all treatments, i.e., surgery, chemoradiotherapy and rehabilitation (Q4). Despite the lack of consensus with regard to clinical improvement post-op or post-all treatments, the majority of respondents agreed that they would encourage their loved one to undergo surgery again (Q6). However, when asked whether NOK would advise another family member or friend who developed a GB to have the same surgery, the results were slightly less favorable (Q7). While most still agreed, a higher number of NOK were neutral in their response.
Table. 2. Cohort characteristics.
|
Cohort size |
124 |
|
Age range (years) |
60 - 86 |
|
Mean age ± SD |
68.8 ± 6.4 |
|
Median age |
68 |
|
60 - 64.9 years |
31% |
|
65 - 74.9 years |
49% |
|
75+ years |
19% |
|
Gender |
|
|
Male |
56% |
|
Female |
44% |
|
Median KPS (IQR) |
60 (40 - 80) |
|
KPS < 70 |
54% |
|
KPS ≥ 70 |
46% |
|
Biopsy |
15% |
|
Subtotal resection |
56% |
|
Gross total
resection |
30% |
|
Median DTD |
|
|
Biopsy |
118 |
|
Resection |
319 |
|
Total cohort |
278 |
|
Median DFI |
|
|
Biopsy |
0 |
|
Resection |
99 |
|
Total cohort |
62 |
|
Volume of tumor
(cm3) |
|
|
Mean ± SD |
39.7 ± 30.1 |
|
Median (IQR) |
34.4 (13.3 - 61.4) |
|
Multifocal tumors |
16% |
|
Bilateral tumors |
10% |
|
Radiotherapy alone |
19% |
|
Chemoradiotherapy |
61% |
KPS: Karnofsky Performance Status Score, DTD: Days to Death, DFI: Days of Functional Independence.
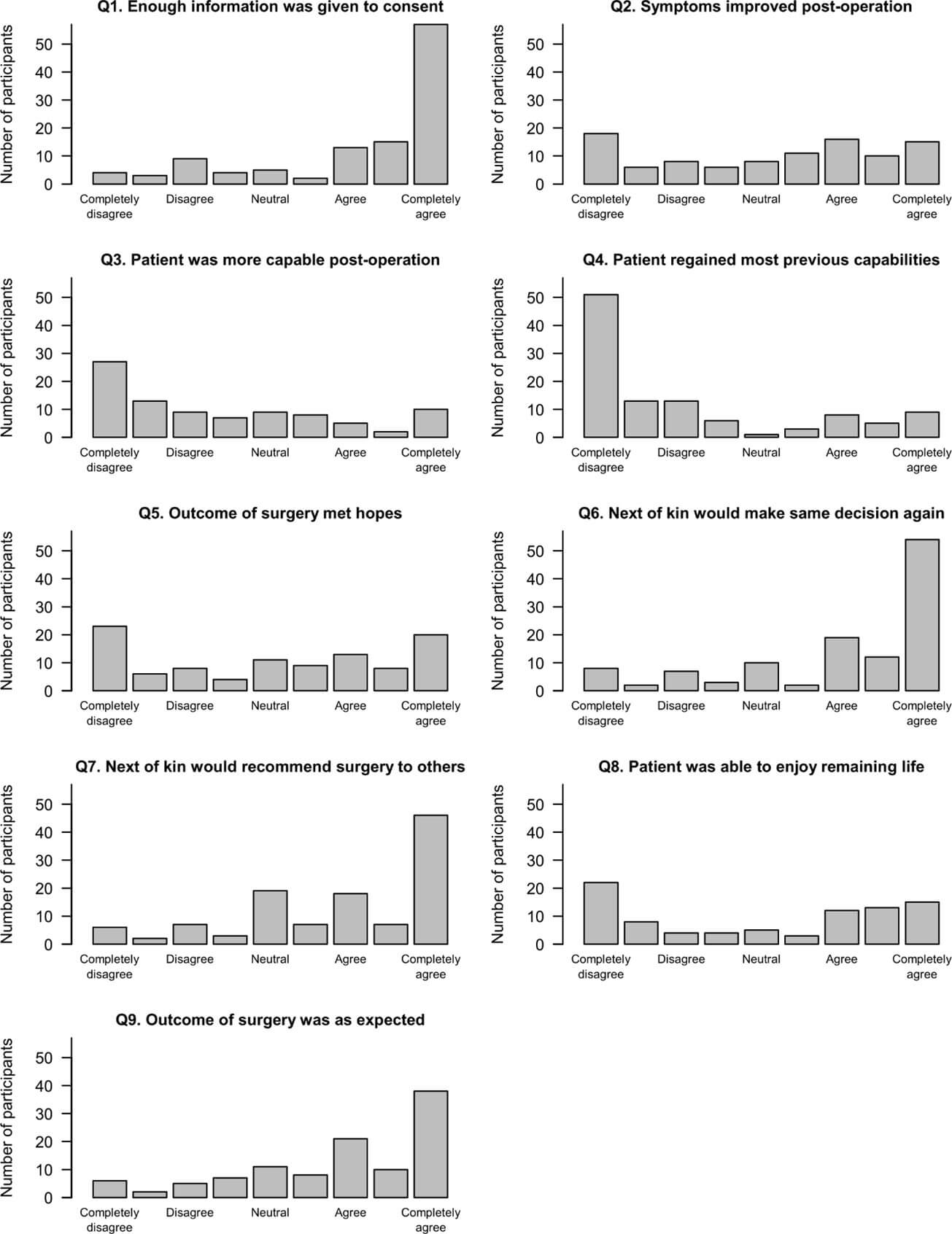
As might be expected a priori, the sample provides statistically significant evidence that NOK responses are positively associated with both DTD and DFI. This conclusion holds for all questions apart from Q1, which addresses whether patients and NOK received sufficient information during the informed consent process (Table 3).
Table. 3. Kendall’s tau results demonstrating the
strength of association between next of kin responses to each question (Q) and
days to death (DTD) and days of functional independence (DFI).
|
Abridged
positively worded questions |
Kendall’s tau -
DFI |
p value |
Kendall’s tau -
DTD |
p value |
|
Q1. Enough
information was given to consent |
0.074 |
0.324 |
0.118 |
0.095 |
|
Q2. Symptoms
improved post-operation |
0.304 |
6 x 10-5 |
0.304 |
3 x 10-5 |
|
Q3. Patient was
more capable post-operation |
0.335 |
3 x 10-5 |
0.256 |
0.001 |
|
Q4. Patient
regained most previous capabilities |
0.333 |
1 x 10-5 |
0.260 |
3 x 10-4 |
|
Q5. Outcome of
surgery met hopes |
0.173 |
0.023 |
0.192 |
0.007 |
|
Q6. Next of kin
would make same decision again |
0.268 |
3 x 10-4 |
0.205 |
0.003 |
|
Q7. Next of kin
would recommend surgery to others |
0.212 |
0.004 |
0.212 |
0.002 |
|
Q8. Patient was
able to enjoy remaining life |
0.281 |
0.001 |
0.254 |
0.001 |
|
Q9. Outcome of
surgery was as expected |
0.159 |
0.035 |
0.172 |
0.014 |
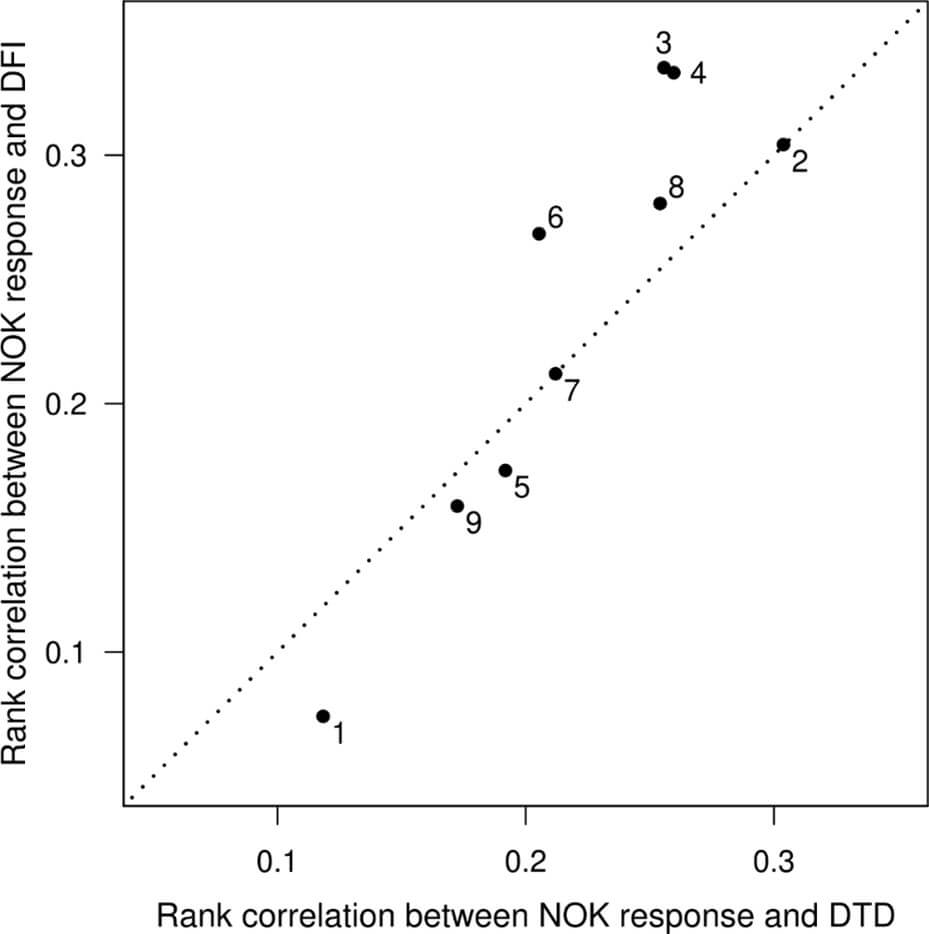
The results suggest that there is a stronger association between NOK response and DFI than between NOK response and DTD. This is illustrated in (Figure 2), where most of the points lie above the line of equality. A stronger association with DFI is most conspicuous in questions related to symptoms and alternative clinical outcome measures (Q3, Q4, Q6 and Q8). However, (even disregarding Q1, which was about information during consent) the overall contrast does not reach statistical significance in this sample (p = 0.11 in one-sample t-test using differences).
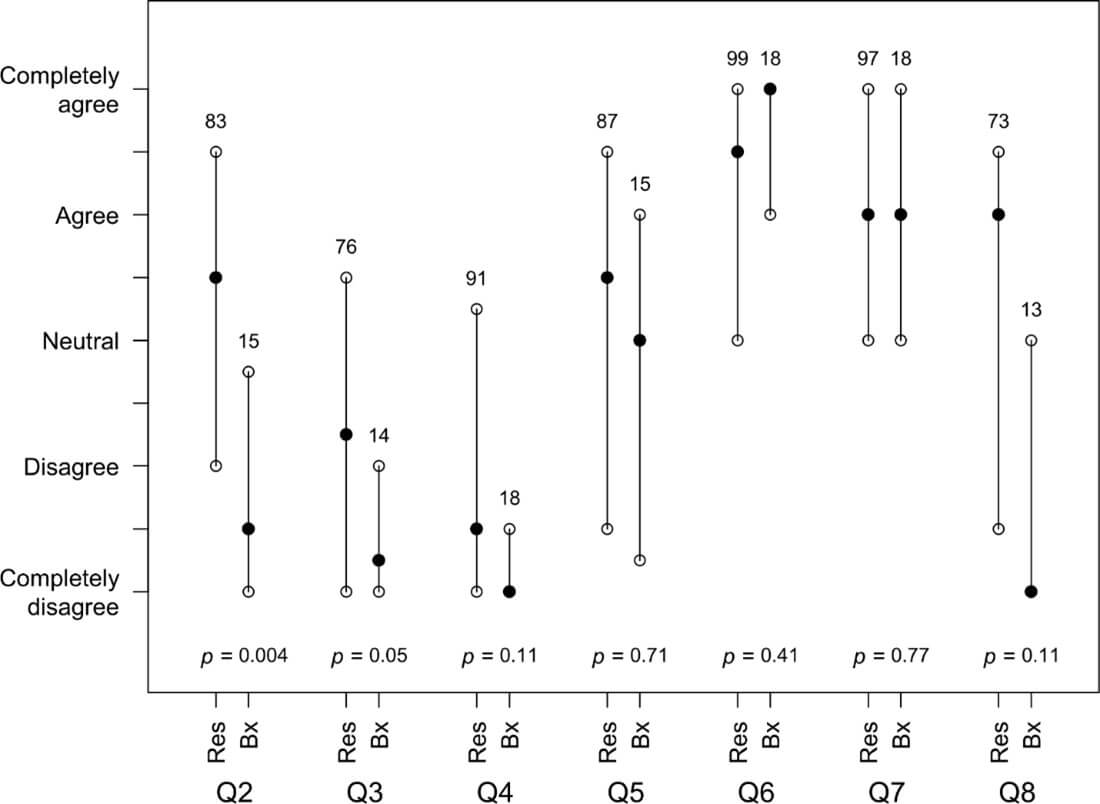
3.3. Post-hoc Analyses
The relationship of surgery type to NOK responses is illustrated in (Figure 3), which gives median responses with upper and lower quartiles, and adjusted p-values for tests of no difference between resection and biopsy. There is strong evidence of a statistical difference for Q2 and an observable difference for Q3, Q4 and Q8, which all relate to the improvement of symptoms. The expectation is that larger sample sizes would have resulted in the evidence to be less equivocal.
There was insufficient evidence for an association between KPS and average NOK responses for Q2-4.
3.4. NOK Written Feedback
Responses by NOK to the additional question and statement were prolific. Participants were thankful for the opportunity to provide feedback to the neurosurgical team and several found the exercise emotionally restorative. In summary, the vast majority of NOK and patients were grateful for their neurosurgical care. One NOK wrote “The treatment and care my mother received from all the medical professionals in the hospital system showed professionalism, skill, commitment, dedication and empathy.” Time estimates, even if incorrect, were appreciated, because being given a number allowed patients and family to get their finances and wills in order and to say goodbye.
While many were thankful for the additional time, a greater proportion of respondents commented that they hadn’t grasped that the prolonged life was of a poor quality with high morbidity, and how extensive the care requirement would be. Multiple participants reflected that QOL had not been part of the consent discussion and implied that the patient’s decision may have been different had morbidity information been given. A NOK commented, “We also wished we had known how physically debilitating surgery would have made him and how highly dependent on help he would be.” Another stated “…(her) belief was that although the treatments had lengthened Dad’s life, it also prolonged or enhanced his suffering too.” One NOK specified that although “The original surgery (biopsy) was successful and gave us and dad the information we needed. It is my belief that the subsequent treatment prolonged his suffering. Dad was in a vegetative state for a long time with no way of real communication but we could see he wanted to but just couldn’t.”
While it is well known that post-operative chemoradiotherapy prolongs life, the NOK responded unanimously that the intensive chemoradiotherapy treatment regimen was physically and emotionally damaging and prolonged the patients’ suffering.
Forty of the patients underwent a second debulking surgery and many of their NOK commented specifically on that surgery. In each case the NOK reported that the second surgery was harder for the patient to handle, took longer to recover from, and was less beneficial. One NOK wrote “The second one was tough, she was very slow to recover & was not the same afterwards. In retrospect, we wish we had not agreed to the second de-bulking, but the surgeon was optimistic it would help. I don’t know if he factored in such a hard recovery.” All NOK that made statements about it expressed regret with the decision to consent to surgery a second time.
3.5. Reproducibility Study
Of the 124 respondents to the initial NOK questionnaire, 50 were randomly selected to be sent a second, differently ordered, questionnaire. One of these 50 was subsequently excluded because they were a child of parents who both had GBs treated at WRH one year apart, and therefore had already filled out two questionnaires, one for each parent. This participant was replaced. Of the resulting 50, 32 (64%) responded. For each of the 18 statements, the scores from the two questionnaires were deemed reliable if they were in perfect agreement or if they differed by only one point on the Likert scale. Of the 576 scores (18 statements x 32 respondents), 493 (86%) exhibited reliability. There was no statistically significant indication of a change in NOK opinion with time (p = 0.10).
4. Discussion
Assessing patients’ perspectives on GB treatments provides a unique challenge. GBs are infiltrative, aggressive and terminal, resulting in cognitive impairment that compromises patients’ ability to reflect comprehensively upon the entirety of their post-operative and end-of-life (EOL) experience. NOKs witness the evolution of patients’ GB-related symptoms, presentation to hospital, operative consent discussion, post-operative treatments, subsequent morbidity, and eventual death. Their ability to accurately recall the patients’ and caregivers’ experiences and opinions is what allows NOKs to be used as surrogates for patients and are the only appropriate respondents to assess the entire EOL period [14-16].
The NOK responses from the questionnaire indicated that the majority of the cohort felt well informed about the risks and benefits of surgery, and the outcomes of treatment met the expectations set during the consent process. However, responses were largely neutral or negative as to whether outcomes met patients’ and NOKs’ hopes. There is no previously published evidence specific to patients with GBs, but this result is in keeping with patients’ attitudes, or unrealistic expectations, to alternative cancer surgeries. For instance, patients with metastatic lung and colon cancer overestimate their survival probabilities despite accurate prognostic estimates from clinicians [17]. In those studies, patients who believed they would survive longer than 6 months were more likely to opt for aggressive treatments but consequently, gained no survival benefit.
While the cohort felt well informed about surgical risks, they were ill-prepared for the morbidity associated with treatment and the disease process. The poor responses to symptom improvement and ability to enjoy life post-operatively are key findings from this study. Responses were particularly negative when NOK were asked if patients regained pre-diagnosis capabilities post-operatively (Q4). In the post-hoc analysis there was significant evidence (Q2) that patients receiving sub or gross total resections experienced a greater reduction in post-operative symptoms than those receiving a biopsy alone. However, that difference did not extend to patient capabilities (Q4), the amount of physical assistance required (Q3) and their ability to maintain a sufficient QOL (Q8). This result may reflect a reduction in potential benefit gained by debulking surgery and maintains a risk of adverse outcomes following neurosurgical intervention, which is in keeping with the findings of the ANOCEF trial [10]. Despite these consistent results, and while NOK opinions most accurately reflect patient EOL experience, previous research has indicated that NOK can underestimate patient ability, thereby overestimating patient morbidity and suffering [15]. The consequence is that NOK responses may err towards minimizing the potential benefits of surgery [15].
Questionnaire responses were further analyzed for their correlation with patient morbidity (DFI) and mortality (DTD). There was a closer association of positive responses with DFI than DTD, excluding questions related to information (Figure 2). This indicates that patients and their NOK may ascribe a higher value to post-operative functional status than length of survival when decision-making. This result fits with the recent shift to include patient reported outcomes in clinical trials [18-20].
If elderly patients potentially value independence above survival, then any information we have about post-operative morbidity should be included in the informed consent process. Unfortunately, novel evidence from Part I in this series demonstrates that only a minority of elderly patients with GBs retain functional independent post-operatively [21]. This simultaneous study completed by our research group focused on post-operative morbidity and found that in the oldest subset of patients, 75 years and above, 57% had zero DFI following their operation [21]. For those over 75 years who only received a biopsy, 92% had zero DFI post-operatively. These results are in keeping with the current study’s lack of reported improvement of symptoms post-operatively, particularly in biopsied patients. When a survival benefit is already significantly reduced in the elderly, the severe morbidity and general dissatisfaction with post-operative QOL raises the question of what benefit is achieved by invasive treatment in an ultimately terminal disease. This highlights a need for enhanced communication specific to a palliative context.
Discussing surgical options for this palliative disease is difficult because patients are still grappling with the diagnosis of a potentially life-limiting brain tumor and surgery can represent a last hope. Having an in-depth discussion about differences in QOL and survival times between treatment options may be beyond the neurologically impaired patients’ ability to assimilate at that time. However, the results of this study and written correspondence from NOK clearly indicate that patients and NOK want exactly that. They want to understand what life may look like as the patient deteriorates, how surgery may impact upon morbidity and mortality, and what support may be necessary for the patient. Effective communication reduces the burden on patients and clinicians alike, but formal training and resources to assist surgeons with such challenging conversations are limited [22, 23]. Earlier involvement of palliative care specialists has been shown to improve QOL for patients with other terminal cancers requiring surgical input and provide support to facilitate critical conversations [24, 25].
Upon reflection of the entire process from hospital presentation to death, and despite high morbidity and early demise, NOK responded with a clear positive majority that in retrospect, their patients would elect to undergo surgical treatment again. However, responses were less positive when NOK were asked whether they would recommend the same treatment to another family member or friend with a radiographic diagnosis of a GB. This difference may be an example of cognitive consistency theory, where one’s attitude changes retrospectively to justify one’s actions. This can create inflated positive responses regarding past experiences, so answers relating to a new situation, which were less positive, may be more reflective of the NOKs’ true feelings. This is especially important in the context of the negative responses regarding post-operative improvement and suggests that perhaps this cohort may be less likely to repeat treatment when faced with the same situation again.
Utilizing NOK as a proxy is a strength of this study as it enabled us to gain unique insights into patients’ experiences and opinions throughout their post-operative and EOL periods until death. Additional strengths include the large elderly cohort, the comprehensive nature of the results, and the focus on creating a robust and reproducible questionnaire with the ability to elicit novel information vital to assessing patient outcomes. The cohort is considered large within the limitations of a rare disease, and the mortality is comparable to similar studies from much larger international centers, increasing the generalizability of the results [4, 5, 26-28]. The 60% response rate for participation in the initial study, and 64% in the reproducibility study, meet JAMAs required response rate to minimize non-response bias [29]. Questions were framed both positively and negatively to reduce acquiescence bias and allowed for identification of incongruent responses. Lastly, the questionnaire was robust, able to elicit consistent responses between the first and second questionnaires, with a reproducibility rate of 86%.
Evaluating the post-operative and EOL experience required delaying the posting of the questionnaires until all patients were deceased. This, along with the rare nature of GBs, necessitated a retrospective design based on surrogate opinions, which introduces potential for multiple biases that can result in more or less favorable responses. Great efforts were made to fill gaps in the data from external sources to reduce inconsistencies and measurement bias, but these could not be entirely excluded. The initial cohort was identified by time since death to reduce recall bias and fading-affect bias, in which negative experiences are forgotten more rapidly than positive ones [30]. A final cut-off of greater than 10 years post-death was implemented to account for these biases. To ensure no NOK received a questionnaire too soon after the death of their loved one, during the acute grief period, a cut-off of 6 months from patient death was used. This was in keeping with similar studies using questionnaires to explore the EOL experience and validated for a New Zealand population [31].
There might be a desire among NOK to justify treatments undergone (cognitive consistency), a response to clinician concern rather than outcome (Hawthorne effect), a provision of a socially acceptable answer (social desirability) and a desire to avoid future prejudice from healthcare providers (ingraining response bias). These effects were reduced by providing anonymity. However, complete anonymity was not achievable due to a need to match responses with patient data. This again may have resulted in a positive skew to responses from NOK. Inclusion of patients receiving a biopsy only may have skewed responses negatively given the aim and expectation of their surgery was not to improve symptoms or prolong life, but to confirm the diagnosis. Despite the potential biases inherent in a retrospective study design, assessing the entirety of the post-operative to EOL patient experience requires a retrospective analysis by NOK who are cognitively unimpaired.
5. Conclusion
This study demonstrates that despite feeling well informed prior to commencing treatment, elderly patients and their NOK underestimated the morbidity associated with surgical and chemoradiotherapy treatment of GB. The NOK responses clearly show that the benefits gained from GB treatment in elderly patients are limited and suggest that their loved ones, the patients, placed a higher value on maintaining independence than survival alone. Decision making for invasive palliative treatments is difficult for clinicians and patients alike [22, 25]. Deeming invasive treatments futile when consulting with patients and family members is clinically, emotionally and ethically challenging, particularly given the lack of a universal standard for an acceptable level of QOL [32, 33]. To prevent unnecessary harm, early in-depth conversations, which align treatments with patients’ wishes, should be undertaken while patients are well enough to participate. Training should be provided to surgeons to facilitate such complex preoperative conversations, and early input from palliative care specialists can assist in this process [22, 23].
Data Availability
The deidentified data set may be made available upon written request to the corresponding author. Access is conditional upon approval by the CCDHB Clinical Audit and Research Committee and the CCDHB Research Advisory Group Māori.
Conflicts of Interest
None.
Funding
None.
REFERENCES
[1] Roger Stupp, Warren P
Mason, Martin J van den Bent, et al. “Radiotherapy plus concomitant and
adjuvant temozolomide for glioblastoma.” N
Engl J Med, vol. 352, no. 10, pp. 987-996, 2005. View at: Publisher
Site | PubMed
[2] Quinn T Ostrom, Mackenzie
Price, Corey Neff, et al. “CBTRUS Statistical Report: Primary Brain and Other
Central Nervous System Tumors Diagnosed in the United States in 2016—2020.” Neuro Oncol, vol. 25, no. 12 Suppl 2,
pp. iv1-iv99, 2023. View at: Publisher Site | PubMed
[3] Qian Han, Hengpo Liang,
Peng Cheng, et al. “Gross Total vs. Subtotal Resection on Survival Outcomes in
Elderly Patients With High-Grade Glioma: A Systematic Review and
Meta-Analysis.” Front Oncol, vol. 10,
pp. 151, 2020. View at: Publisher Site | PubMed
[4] Dieter Henrik Heiland,
Gerrit Haaker, Ralf Watzlawick, et al. “One decade of glioblastoma multiforme
surgery in 342 elderly patients: what have we learned?” J Neurooncol, vol. 140, no. 2, pp. 385-391, 2018. View at: Publisher Site | PubMed
[5] Jacob G Scott, John H Suh,
Paul Elson, et al. “Aggressive treatment is appropriate for glioblastoma
multiforme patients 70 years old or older: a retrospective review of 206
cases.” Neuro Oncol, vol. 13, no. 4,
pp. 428-436, 2011. View at: Publisher Site | PubMed
[6] Agi Oszvald, Erdem
Güresir, Matthias Setzer, et al. “Glioblastoma therapy in the elderly and the
importance of the extent of resection regardless of age.” J Neurosurg, vol. 116, no. 2, pp. 357-364, 2012. View at: Publisher
Site | PubMed
[7] Gawande A. Being Mortal; Illness, Medicine and What
Matter in the End. Metropolitan Books, Henry Holt and Company LLC; 2014.
[8] Asgeir S Jakola, Geirmund
Unsgård, Ole Solheim “Quality of life in patients with intracranial gliomas:
the impact of modern image-guided surgery.” J
Neurosurg, vol. 114, no. 6, pp. 1622-1630, 2011. View at: Publisher
Site | PubMed
[9] Lisa Millgård Sagberg, Ole
Solheim, Asgeir S Jakola, et al. “Quality of survival the 1st year with
glioblastoma: a longitudinal study of patient-reported quality of life.” J Neurosurg, vol. 124, no. 4, pp.
989-997, 2016. View at: Publisher Site | PubMed
[10] Florence Laigle-Donadey,
Philippe Metellus, Jacques Guyotat, et al. “Surgery for glioblastomas in the
elderly: an Association des Neuro-oncologues d'Expression Française (ANOCEF)
trial.” J Neurosurg, vol. 138, no. 5,
pp. 1199-1205, 2022. View at: Publisher Site | PubMed
[11] Peter F. Edemekong, Deb L.
Bomgaars, Sukesh Sukumaran, et al. “Activities of Daily Living. StatPearls
Publishing, 2020. View at: PubMed
[12] G. E. NOETHER “Why Kendall
Tau?” Teaching Statistics, vol. 3, no. 2, pp. 41-43, 1981. View at: Publisher Site
[13] Peter H. Westfall, S.
Stanley Young. Algorithm 2.8, pg 66. In Resampling-Based Multiple Testing,
Examples and methods for p-value adjustment. John Wiley & Sons, Inc. 605
Third Avenue, New York, NY 1993.
[14] J Magaziner, E M
Simonsick, T M Kashner, et al. “Patient-proxy response comparability on
measures of patient health and functional status.” J Clin Epidemiol, vol. 41, no. 11, pp. 1065-1074, 1988. View at: Publisher Site | PubMed
[15] Jessica K. Roydhouse, Roee
Gutman, Nancy L. Keating, et al. “Proxy and patient reports of health-related
quality of life in a national cancer survey.” Health Qual Life Outcomes, vol. 16, pp. 6, 2018. View at: Publisher Site
[16] F J Fowler Jr, K M
Coppola, J M Teno “Methodological Challenges for Measuring Quality of Care at
the End of Life.” J Pain Symptom Manage,
vol. 17, no. 2, pp. 114-119, 1999. View at: Publisher Site | PubMed
[17] J C Weeks, E F Cook, S J
O'Day, et al. “Relationship between cancer patients' predictions of prognosis
and their treatment preferences.” JAMA,
vol. 279, no. 21, pp. 1709-1714, 1998. View at: Publisher
Site | PubMed
[18] Terri S Armstrong, Linda
Dirven, David Arons, et al. “Glioma patient-reported outcome assessment in
clinical care and research: a Response Assessment in Neuro-Oncology
collaborative report.” Lancet Oncol,
vol. 21, no. 2, pp. e97-e103, 2020. View at: Publisher Site | PubMed
[19] Raymond Liu, Margaretta
Page, Karla Solheim, et al. “Quality of life in adults with brain tumors:
current knowledge and future directions.” Neuro
Oncol, vol. 11, no. 3, pp. 330-339, 2009. View at: Publisher Site | PubMed
[20] Catherine E Wassef, Sarah
A Cain, Katharine J Drummond “Changing practice to improve quality of life in
glioma.” J Neurosurg, vol. 141, no.
5, pp. 1270-1280, 2024. View at: Publisher
Site | PubMed
[21] Abhinav Bhasin, Robin
Willink, Mira Steinmetz, et al. “Part 1 - Do Less Harm: A Quantitative Analysis
of Morbidity and Mortality Following Treatment of Glioblastoma in the Elderly
and the Development of a Clinical Prognostic Tool.” World Neurosurg,
vol. 12, no. 3, pp. 1-9, 2025.
[22] Pasithorn A Suwanabol, Ari
C Reichstein, Z Tuba Suzer-Gurtekin, et al. “Surgeons' Perceived Barriers to
Palliative and End-of-Life Care: A Mixed Methods Study of a Surgical Society.” J Palliat Med, vol. 21, no. 6, pp.
780-788, 2018. View at: Publisher Site | PubMed
[23] Daniel D Klaristenfeld,
David T Harrington, Thomas J Miner “Teaching palliative care and end-of-life
issues: a core curriculum for surgical residents.” Ann Surg Oncol, vol. 14, no. 6, pp. 1801-1806, 2007. View at: Publisher Site | PubMed
[24] Jennifer S Temel, Joseph A
Greer, Alona Muzikansky, et al. “Early palliative care for patients with
metastatic non-small-cell lung cancer.” N
Engl J Med, vol. 363, no. 8, pp. 733-742, 2010. View at: Publisher
Site | PubMed
[25] Camilla Zimmermann, Nadia
Swami, Monika Krzyzanowska, et al. “Early palliative care for patients with
advanced cancer: a cluster-randomised controlled trial.” Lancet, vol. 383, no. 9930, pp. 1721-1730, 2014. View at: Publisher Site | PubMed
[26] Kaisorn L Chaichana, Khan
K Chaichana, Alessandro Olivi, et al. “Surgical outcomes for older patients
with glioblastoma multiforme: preoperative factors associated with decreased
survival.” Clinical article. J Neurosurg,
vol. 114, no. 3, pp. 587-594, 2011. View at: Publisher
Site | PubMed
[27] Fabio M Iwamoto, Anne S
Reiner, Lakshmi Nayak, et al. “Prognosis and patterns of care in elderly
patients with glioma.” Cancer, vol.
115, no. 23, pp. 5534-5540, 2009. View at: Publisher
Site | PubMed
[28] Kalil G Abdullah, Ashwin
Ramayya, Jayesh P Thawani, et al. “Factors associated with increased survival
after surgical resection of glioblastoma in octogenarians.” PloS One, vol. 10, no. 5, pp. e0127202,
2015. View at: Publisher Site | PubMed
[29] JAMA Network. Instructions
for Authors. Accessed 12 March 2025.
[30] Jeffrey A Gibbons, Sherman
A Lee, Ashley M A Fehr, et al. “Grief and Avoidant Death Attitudes Combine to
Predict the Fading Affect Bias.” Int J
Environ Res Public Health, vol. 15, no. 8, pp. 1736, 2018. View at: Publisher
Site | PubMed
[31] Kate Reid, Ray Kirk,
Pauline Barnett, et al. “VOICES: South Island pilot survey of bereaved people.”
N Z Med J, vol. 133, no. 1518, pp.
64-72, 2020. View at: PubMed
[32] Mark Ewanchuk, Peter G
Brindley “Perioperative do-not-resuscitate orders--doing 'nothing' when
'something' can be done.” Crit Care,
vol. 10, no. 4, pp. 219, 2006. View at: Publisher Site | PubMed
[33] Scott B Grant, Parth K Modi, Eric A Singer “Futility and the care of surgical patients: ethical dilemmas.” World J Surg, vol. 38, no. 7, pp. 1631-1637, 2014. View at: Publisher Site | PubMed
