Received: Tue 16, Dec 2025
Accepted: Sat 03, Jan 2026
Abstract
Introduction: Efficacy of embolization of the middle meningeal artery (EMMA) for chronic subdural hematoma (cSDH) has been shown. Arteriovenous fistula (AVF) has been noted in patients with cSDH but the prevalence of AVFs in cSDH patients is not known. Our study aimed to ascertain the prevalence of AVFs and associated factors in patients undergoing EMMA.
Methodology: A prospective cohort study was conducted on patients with cSDH who underwent EMMA. Two independent reviewers assessed angiographic images to determine the presence of non-iatrogenic AVF, their location, arterial supply, venous drainage, and whether seen on diagnostic, microcatheter angiograms, or during embolization. The prevalence of non-iatrogenic AVF was calculated and the association with various demographic and clinical factors were assessed.
Results: From May 2021 to June 2023, 55 patients (43 Males; Median age 65 years;14 primary EMMA; 41 post-surgical EMMA) underwent 64 EMMA procedures. Ten patients had bilateral cSDH were all male. Non-iatrogenic AVF were identified in 18 patients (32.7%) and in 21 EMMAs (32.8%) with 4 demonstrating multiple AVFs. Most common arterial supply was from the posterior division of MMA (18 out of 26) and venous drainage into the osteodural vein (18 out of 26). Higher prevalence (p=0.028) of AVF was seen in those who did not undergo surgical drainage.
Conclusion: Non-iatrogenic AVFs were seen in one-third patients with cSDH undergoing EMMA with a higher prevalence in those without surgical drainage.
Keywords
Arterio-venous fistula, chronic subdural hematoma, embolization of middle meningeal artery, middle meningeal artery
1. Introduction
Chronic subdural hematoma (cSDH) is a common neurosurgical condition, particularly affecting the elderly, and is managed through observation, medical therapy, or surgical intervention [1-6]. Recent research has suggested the efficacy of embolization of middle meningeal artery (EMMA) as either an adjunct or standalone treatment for cSDH [7-11]. There have been reports of arteriovenous fistulas (AVFs) in patients with cSDH [12-14]. AVFs are abnormal shunts between meningeal arteries and dural sinuses or veins. The presence of AVFs has been suggested to be a risk factor for cSDH [2, 12]. AVFs are also suggested to be iatrogenic, potentially resulting either from surgical drainage or EMMA itself [12, 14]. However the trure prevalence of non-iatrogenic arterio-venous fistulous communications in patients with cSDH has not been systematically studied. Our study aimed to determine the prevalence and angiographic characteristics of non-iatrogenic AVFs in patients cSDH undergoing EMMA.
2. Methodology
The study was approved by the institution’s Research Ethics Board and written informed consent was obtained from all participants. We included consecutive patients who underwent EMMA as a part of prospective registry for cSDH between May 2021 to June 2023. Demographic and clinical data were collected including prior surgical interventions, embolization related complications, and anticoagulant use.
2.1. Image Analysis
The angiographic images were independently reviewed by two reviewers: a senior medical student (TU) and an interventional neuroradiologist (JS) with more than 15 years of experience. Images were assessed for the presence of AVFs, their location, arterial supply, venous drainage, and embolization technique and embolic agent used.
AVFs were documented based on whether they were visualized on the diagnostic angiography, microcatheter injections, or during liquid embolic agent injection. AVFs were considered iatrogenic if they were identified near the microcatheter tip (suggestive of micro-guidewire injury), or at the prior craniotomy site. Patients with suspected iatrogenic AVFs were excluded to assess the true incidence of non-iatrogenic AVFs underlying CSDH. Selection of embolic agents was operator-dependent, based on angiographic anatomy and other logistic considerations.
2.2. Data Analysis
Statistical analysis was performed using the Stata 13.1 (StataCorp, Texas, USA). The prevalence of AVFs was calculated. The association of AVFs with demographic and clinical factors were assessed using univariate analysis. Inter-rater agreement was evaluated using Kappa statistics. A p-value of less than 0.05 was considered statistically significant.
3. Results
A total of 55 patients (43 male; mean age- 62.7 years; median age- 65 years) underwent EMMA at our institution during the study period. Table 1 summarizes the demographic, clinical, and angiographic characteristics of the cohort. Of these, 41 underwent surgical intervention prior to EMMA, while 14 did not. One patient who underwent surgery after embolization was included in the non-surgical group for analysis. Bilateral CSDH was observed in 15 patients, all of whom were male. A total of 64 middle meningeal angiograms were performed. Three patients did not undergo embolization due to difficulties accessing the MMA, MMA origin from ophthalmic artery, and MMA ligation during surgery. Arteriovenous Fistula (Figure 1). Eighteen patients (32.7%) were found to have AVFs. Because some patients underwent bilateral EMMA, a total of 64 embolizations were analyzed. AVFs were detected in 21 angiograms (32.8%). Four patients had more than one AVF (Table 1).
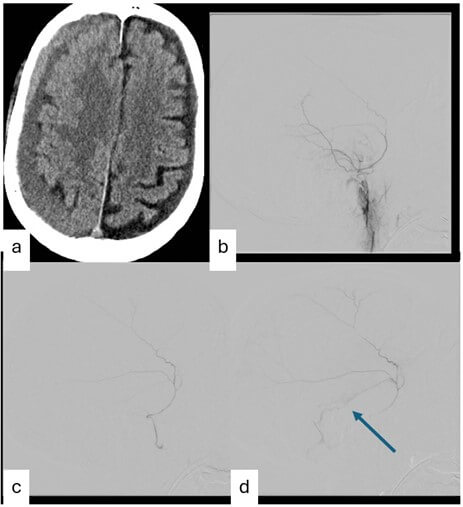
Table. 1. Demographic and Angiographic Characteristics of the Study Cohort.
|
Characteristics |
Number (%) |
|
Total number of
patients |
55 |
|
Female |
12 (21.8) |
|
Male |
43 (78.2) |
|
Age (years) |
|
|
£60 |
34 (61.8) |
|
>60 |
21 (38.2) |
|
cSDH Location |
|
|
Bilateral |
15 (27.3) |
|
Right Hemisphere |
19 (34.5) |
|
Left Hemisphere |
21 (38.2) |
|
Surgical drainage Prior to-EMMA |
|
|
Yes |
41 (74.5) |
|
No |
14 (25.5) |
|
EMMAs |
64 |
|
Right Hemispheres |
31 (48.4) |
|
Left Hemispheres |
33 (51.6) |
|
Patients with ≥1AVF |
18 (32.7) |
|
Hemispheric
Angiograms with AVF |
21 (32.8) |
|
Hemispheric
Angiograms with more than one AVF |
4 (6.3) |
|
2 AVF |
3 |
|
3 AVF |
1 |
|
Visualization of AVF |
|
|
Global angiography |
0 |
|
Microcatheter
angiography |
21 (100) |
|
During liquid embolic
injection |
4 (19.0) |
|
Arterial Supply of AVFs* |
|
|
Accessory MMA |
1 (3.8) |
|
Anterior Division of
MMA |
7 (26.9) |
|
Posterior Division of
MMA |
18 (69.2) |
|
Venous Drainage of AVFs* |
|
|
Osteo-dural Vein |
18 (69.2) |
|
Sigmoid Sinus |
2 (7.7) |
|
Superior Sagittal
Sinus |
3 (11.5) |
|
Transverse Sinus |
3 (11.5) |
Values are reported as
n (%) unless otherwise stated.
*Percentages calculated
per total number of AVFs (n = 26).
AVF: Arterio-Venous Fistula; cSDH: Chronic Subdural Hematoma; EMMA: Embolization of Middle Meningeal Artery; MMA: Middle Meningeal Artery.
The most common arterial supply was from the posterior division of the MMA (18/26, 69.2%) and the most common venous drainage was into the osteodural veins (18/26, 69.2%). No residual AVFs were observed on post-embolization control angiograms. No patient experienced recurrence of cSDH during the follow-up period. Table 2 presents various demographic and clinical factors associated with AVFs. Prior surgical drainage was associated with lower prevalence of AVFs (32% vs 53%, p=0.028). Inter-rater agreement was moderate (k= 0.42). In five patients, the AVFs were deemed iatrogenic and excluded from the analysis.
Table. 2. Association Between Demographic and Angiographic Characteristics and
Presence of Arteriovenous Fistula.
|
Characteristics |
Total (n) |
Without AVF (n) |
With AVF (n) |
p-value |
|
Angiograms performed |
64 |
43 |
21 |
0.659 |
|
Right Hemispheres |
31 |
20 |
11 |
|
|
Left Hemispheres |
33 |
23 |
10 |
|
|
Gender |
|
|
|
0.200 |
|
Female |
12 |
10 |
2 |
|
|
Male |
53 |
34 |
19 |
|
|
Age |
|
|
|
0.114 |
|
£60-years |
40 |
24 |
16 |
|
|
>60-years |
24 |
19 |
5 |
|
|
EMMA performed |
61 |
|
|
|
|
Embolic Agent |
|
|
|
0.729 |
|
Onyx |
22 |
15 |
7 |
|
|
Particle |
8 |
6 |
2 |
|
|
Squid |
31 |
19 |
12 |
|
|
Surgical drainage before EMMA |
|
|
|
0.028 |
|
Yes |
45 |
34 |
11 |
|
|
No |
19 |
9 |
10 |
|
4. Discussion
Our study demonstrates that the non-iatrogenic AVFs are present in approximately one-third of patients with cSDH undergoing EMMA, with multiple AVFs observed in 6.25% of patients. Prior surgical drainage was associated with a lower prevalence of AVFs. Most AVFs were supplied by posterior division of MMA and drained into osteo-dural veins. Prior literature has reported AVFs in association with cSDH primarily through isolated case reports and small series, limiting accurate estimation of prevalence [12, 14, 15]. Proposed mechanisms included congenital dural shunts becoming patent after hemodynamic changes or angiogenesis following craniotomy. However, these mechanisms alone do not fully explain our findings, as AVFs were present in only one-third of patients and were less prevalent in those who had undergone surgery.
Importantly, AVFs in our cohort were often visible only on microcatheter angiography, supporting the hypothesis that these may represent physiological dural shunts (< 12 μm) not visible on global DSA [12, 14, 16]. This observation suggests that such shunts may be underrecognized during routine angiography. Although procedural iatrogenesis remains a concern, cases suspicious for iatrogenic AVFs were systematically excluded, strengthening the validity of our prevalence estimate. Consistent with prior case reports, no patient experienced cSDH recurrence following embolization, even in the presence of AVFs [12, 18]. Given the potential role of both MMA flow and dural AVFs in sustaining cSDH, systematic evaluation for AVFs during EMMA is warranted. When AVFs are identified, liquid embolic agents may be preferable, as they allow effective fistula occlusion without venous migration.
This single-center study is limited by sample size and potential selection bias, as three patients did not undergo embolization. Nevertheless, this study represents the first prospective assessment of non-iatrogenic AVF prevalence in cSDH patients undergoing EMMA.
5. Conclusion
Non-iatrogenic AVFs were observed in approximately one-third of patients with cSDH undergoing EMMA. Prior surgical drainage was associated with a lower incidence of AVFs. Larger, multicenter studies are needed to validate these findings and further elucidate the role of AVFs in cSDH pathophysiology and recurrence.
Funding
None.
Author Contributions
JS- conceptualized the study, monitored the study conduct, analyzed the data and finalized the manuscript.
Conflicts of Interest
JS is PI for EMMA Can study funded by Medtronic Canada.
Disclaimer
The study was presented in WFITN meeting, New York in October 2024.
REFERENCES
[1] Seung Pil Ban, Gyojun
Hwang, Hyoung Soo Byoun, et al. “Middle meningeal artery embolization for
chronic subdural hematoma.” Radiology, vol. 286, no. 3, pp. 992-999,
2018. View at: Publisher
Site | PubMed
[2] Jinlu Yu 1, Yunbao Guo 1,
Baofeng Xu, et al. “Clinical importance of the middle meningeal artery: A
review of the literature.” Int J Med Sci, vol. 13, no. 10, pp. 790-799,
2016. View at: Publisher
Site | PubMed
[3] Min Xu, Pin Chen, Xun Zhu,
et al. “Effects of Atorvastatin on Conservative and Surgical Treatments of
Chronic Subdural Hematoma in Patients.” World Neurosurg, vol. 91, pp.
23-28, 2016. View at: Publisher
Site | PubMed
[4] J Shapey, L J Glancz, P M
Brennan “Chronic Subdural Haematoma in the Elderly: Is It Time for a New
Paradigm in Management?” Curr Geriatr Rep, vol. 5, pp. 71-77, 2016. View
at: Publisher Site | PubMed
[5] Yasuaki Abe, Keisuke
Maruyama, Shigeomi Yokoya, et al. “Outcomes of chronic subdural hematoma with
preexisting comorbidities causing disturbed consciousness.” J Neurosurg,
vol. 126, no. 4, pp. 1042-1046, 2017. View at: Publisher Site | PubMed
[6] Joshua Dian, Janice
Linton, Jai Js Shankar “Risk of recurrence of subdural hematoma after EMMA vs
surgical drainage - Systematic review and meta-analysis.” Interv Neuroradiol,
vol. 27, no. 4, pp. 577-583, 2021. View at: Publisher Site | PubMed
[7] Joshua S Catapano, Andrew
F Ducruet, Visish M Srinivasan, et al. “Radiographic clearance of chronic
subdural hematomas after middle meningeal artery embolization.” J
Neurointerventional Surg, vol. 14, no. 12, pp. 1279-1283, 2022. View at: Publisher Site | PubMed
[8] Evan Joyce, Michael T
Bounajem, Jonathan Scoville, et al. “Middle meningeal artery embolization
treatment of nonacute subdural hematomas in the elderly: a multiinstitutional
experience of 151 cases.” Neurosurg Focus, vol. 49, no. 4, pp. E5, 2020.
View at: Publisher
Site | PubMed
[9] Peter Kan, Georgios A
Maragkos, Aditya Srivatsan, et al. “Middle Meningeal Artery Embolization for
Chronic Subdural Hematoma: A Multi-Center Experience of 154 Consecutive
Embolizations.” Neurosurgery, vol. 88, no. 2, pp. 268-277, 2021. View
at: Publisher Site | PubMed
[10] Gary B Rajah, Muhammad
Waqas, Rimal H Dossani, et al. “Transradial middle meningeal artery
embolization for chronic subdural hematoma using Onyx: case series.” J
Neurointerventional Surg, vol. 12, no. 12, pp. 1214-1218, 2020. View at: Publisher Site | PubMed
[11] Justin Schwarz, Joseph A
Carnevale, Jacob L Goldberg, et al. “Perioperative prophylactic middle
meningeal artery embolization for chronic subdural hematoma: a series of 44
cases.” J Neurosurg, vol. 135, no. 6, pp. 1627-1635, 2021. View at: Publisher Site | PubMed
[12] Armin Tavakkoli, Julio D
Montejo, Daniel R Ca, et al. “Intra-operative emergence of occult dural
arteriovenous fistula after middle meningeal artery embolization for chronic
subdural hematoma: Case report and literature review.” Radiol Case Rep,
vol. 17, no. 5, pp. 1470-1474, 2022. View at: Publisher Site | PubMed
[13] Rami O Almefty, M Yashar S
Kalani, Andrew F Ducruet, et al. “Middle meningeal arteriovenous fistulas: A
rare and potentially high-risk dural arteriovenous fistula.” Surg Neurol Int,
vol. 7, no. Suppl 9, pp. S219-S222, 2016. View at: Publisher Site | PubMed
[14] Lorenzo Piergallini, Cyril
Dargazanli, Imad Derraz, et al. “Immediate Development of Dural Arteriovenous
Fistula After Middle Meningeal Artery Embolization: First Angiographic
Demonstration.” World Neurosurg, vol. 128, pp. 606-610.e1, 2019. View
at: Publisher
Site | PubMed
[15] A Watanabe, Y Takahara, Y
Ibuchi, et al. “Two cases of dural arteriovenous malformation occurring after
intracranial surgery.” Neuroradiology, vol. 26, no. 5, pp. 375-380,
1984. View at: Publisher
Site | PubMed
[16] C W Kerber, T H Newton
“The macro and microvasculature of the dura mater.” Neuroradiology, vol.
6, no. 4, pp. 175-179, 1973. View at: Publisher Site | PubMed
[17] Lorenz Ertl, Hartmut
Brückmann 1, Mathias Kunz, et al. “Endovascular therapy of low- and
intermediate-grade intracranial lateral dural arteriovenous fistulas: a
detailed analysis of primary success rates, complication rates, and long-term
follow-up of different technical approaches.” J Neurosurg, vol. 126, no.
2, pp. 360-367, 2017. View at: Publisher Site | PubMed
[18] Tushit Mewada, Tomotaka
Ohshima, Taiki Yamamoto, et al. “Usefulness of Embolization for Iatrogenic
Dural Arteriovenous Fistula Associated with Recurrent Chronic Subdural
Hematoma: A Case Report and Literature Review.” World Neurosurg, vol.
92, pp. 584.e7-584.e10, 2016. View at: Publisher Site | PubMed
[19] Hirohisa Yajima, Satoshi Koizumi, Tomoki Kanda, et al. “Possible Association between Recurrent Chronic Subdural Hematoma and Dural Arteriovenous Fistula: A Case Report with Three-dimensional Fusion Images.” NMC Case Rep J, vol. 10, pp. 41-45, 2023. View at: Publisher Site | PubMed
