Received: Sun 30, Nov 2025
Accepted: Wed 07, Jan 2026
Abstract
Ventriculoperitoneal (VP) shunts are commonly used to treat hydrocephalus, but they often fail, sometimes without a clear cause. This study aims to determine if air introduced in the shunt during installation may be associated with system dysfunction post-surgery.
A single-center case-control retrospective study was conducted on patients who underwent VP shunt placement. Demographic data were collected, along with measurements of air quantity inside ventricles and inside the valve using available postoperative CT scans. Inclusion criteria comprised an initial VP shunt installation for the treatment of hydrocephalus and the availability of a postoperative CT scan with a resolution of .5 mm. Patients operated on by surgeons who had fewer than five patients initially selected in the study were excluded. The main objective was to determine if air introduced into the ventriculoperitoneal shunts at the time of implantation was associated with an increased risk of subsequent dysfunction.
A total of 128 patients met the inclusion criteria. Revision surgery was significantly associated with patient age at shunt placement (p = .003), hydrocephalus type (p = .018) and presence of air in the valve after installation (p = .046). Air inside ventricles (p = .892) and valve type (p = .078) were not statistically correlated with the occurrence of revision surgery. Binomial Logistic Regression analysis showed that postoperative air in the valve increases the likelihood of revision surgery by 4.2 times.
Air trapped in the valve after VP shunt placement may contribute to system dysfunction and should be removed by neurosurgeons during installation surgery.
Keywords
Air bubbles, hydrocephalus, obstruction, revision surgery, valve, ventriculoperitoneal shunt
1. Introduction
Hydrocephalus is characterized by an excess of cerebrospinal fluid (CSF) within brain ventricles [1]. It is commonly treated by the insertion of a ventriculoperitoneal (VP) shunt. VP shunts have a failure rate of 10% within the first year following initial implantation and 32% after 2 years, often necessitating revision surgery [2, 3]. Failure factors like infection or catheter disconnection are easily identifiable, but in other cases, no specific cause can be identified. In these situations, the valve is typically considered as the likely source of failure. The valve alone is estimated to be responsible for 14.1% to 18.6% of shunt revision surgeries, although this is often a diagnosis of exclusion and valve dysfunction is rarely specifically demonstrated [2, 4].
It is known in clinical practice that small air bubbles often remain in the valve reservoir at the time of placement, and this residual air can be difficult to purge (Figure 1). While the clinical relevance of air bubbles is unclear, one case report suggests that they could be a root cause of VP dysfunction [5]. This type of failure would be radiologically occult, as air is not visible in radiopaque catheters and would therefore currently go undiagnosed.
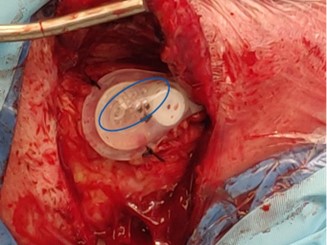
The objective of this study was to determine if air introduced into VP shunts at the time of implantation is associated with an increased risk of subsequent dysfunction justifying a revision procedure.
2. Methods
We performed a retrospective case-control study. The study population consisted of patients who underwent a VP shunt placement between January 1, 2011 and December 31, 2022 at the Centre Hospitalier Universitaire de Sherbrooke (CHUS) in Quebec, Canada. The case group consisted of patients who underwent at least one VP shunt revision surgery at any time. This served as our proxy for indicating a clinically meaningful shunt dysfunction. The control group consisted of patients who did not undergo any revision procedure during follow-up. There was no match between cases and controls.
The inclusion criteria for this study were as follows:
• Availability of a postoperative computer tomography (CT) scan with a resolution of .5 mm;
• Initial VP shunt placement performed at the CHUS;
• Indication for shunt was hydrocephalus.
The exclusion criteria for this study were as follows:
• Surgical procedure performed by a neurosurgeon with fewer than five patients included in the study cohort. This criterion was added to reduce potential bias related to individual surgeon variability and ensure sufficient procedural consistency for meaningful comparison.
This study was approved by our local institutional ethics committee. Given the retrospective nature of the study, the requirement for informed consent was not needed.
2.1. Sample Size
To detect a 25% difference in the rate of dysfunction attributable to the presence of air, and assuming a 32% rate of revision [2, 3], we estimated that a sample of 100 patients would be required, including 70 without revision and 30 with revision. All medical records of patients who underwent a VP shunt procedure at our center were reviewed for inclusion, starting with the year 2022 and proceeding backward to the beginning of 2021. Because our actual dysfunction rate was 10.7% rather than 32%, we then screened the years 2019-2020 for patients with at least one revision surgery only. To balance the distribution of surgeons, all patients operated on by surgeons other than Dr. A were included from 2015 to 2018. To balance the years, the same patient selection pattern was repeated from 2011 to 2014 (Supplemental Digital Content 1).
2.2. Variables
For each patient, demographic data such as age at the time of shunt insertion and gender were collected from medical records. Additional information was collected regarding the indication for shunt installation (i.e., hydrocephalus type), the date of installation, the surgeon who performed the procedure, and the type of valve and catheter used. For patients who underwent at least one revision surgery (cases), the date of the revision, as well as the cause and site of failure were identified in the medical record. For all surgical procedures, available preoperative and postoperative CT scans were reviewed to assess the amount of air remaining within the ventricles and the ventriculoperitoneal shunt system.
2.3. Data Source / Measurement
To measure the amount of air present inside brain ventricles and the VP shunt system, the software 3D Slicer 5.6.2 was used to segment the air bubbles. As shown in Figure 2, air appears hypodense on CT scans and can be easily delimited from surrounding structures. Regions with Hounsfield unit (HU) values below -1000 in the valve reservoir were considered to represent air and were segmented. All images were segmented by the same individual to minimize inter-user variability. The same measurement method was applied identically to both the case and control groups. The volume of air present in the ventricles and valve reservoir after initial shunt insertion (and after revision surgeries, if applicable) was determined. It was not possible to visualize the presence of air within the catheter components of the medical device, as these are radiopaque.
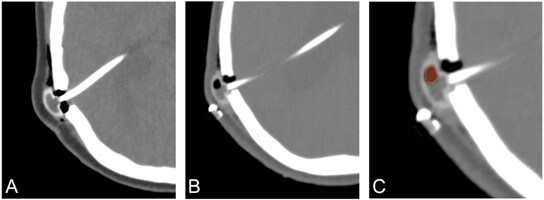
2.4. Statistical Methods
Statistical analyses were performed using IBM SPSS Statistics, version 29.0.2.0. All statistical tests conducted in this study were intended to compare patients who did not undergo revision surgery (controls) with those who underwent at least one revision surgery (cases). Each variable was first analyzed individually to assess its impact on the two groups. The Kaplan-Meier and log-rank statistical tests were used to explore the association between individual factors and the time to VP shunt failure. Survival time was the number of months without revision surgery following the installation procedure, and cases were censored at their last medical follow-up available. A significance threshold of p < .05 was used. These univariate analyses served to identify candidate variables for inclusion in the multivariate models. Subsequently, Binomial Logistic Regression analysis (multivariate) were performed to adjust for potential modifying or confounding effects among variables and to address the main research question regarding the impact of air on VP shunt failure. Missing data were handled by excluding patients with missing information for any relevant variable, except when the cause of failure was missing; in such cases, the patient was categorized as “Not specified in the files’’. No imputation techniques were used. Descriptive statistics are presented using the median (range) for continuous variables and frequency with valid percentage for categorical variables.
3. Results
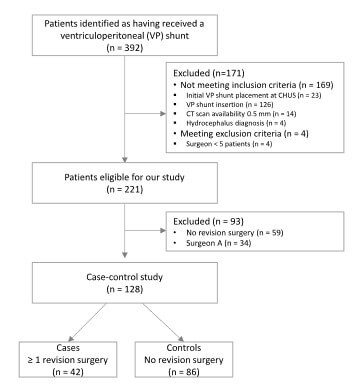
The number of patients at each stage of the study is shown in (Figure 3). Out of 392 patients initially screened, 128 were selected for the study (42 cases and 86 controls). Follow-up duration ranged from .1 to 156 months, with a mean of 41 months and a median of 24 months.
3.1. Descriptive Data
The characteristics of the patient population are summarized in (Table 1). The median age at the time of VP shunt insertion was 70 years (IQR: 15, range: 0-87). Cases (revision surgery) were statistically younger than controls (64.5 vs. 73 years, p = .003).
Regarding the type of hydrocephalus, the majority of patients had normal pressure hydrocephalus (50.0%). Controls (no revision surgery) also predominantly had normal pressure hydrocephalus (57.0%) while the majority of cases were communicating hydrocephalus (40.5%). This difference in hydrocephalus type was statistically significant (p = .018).
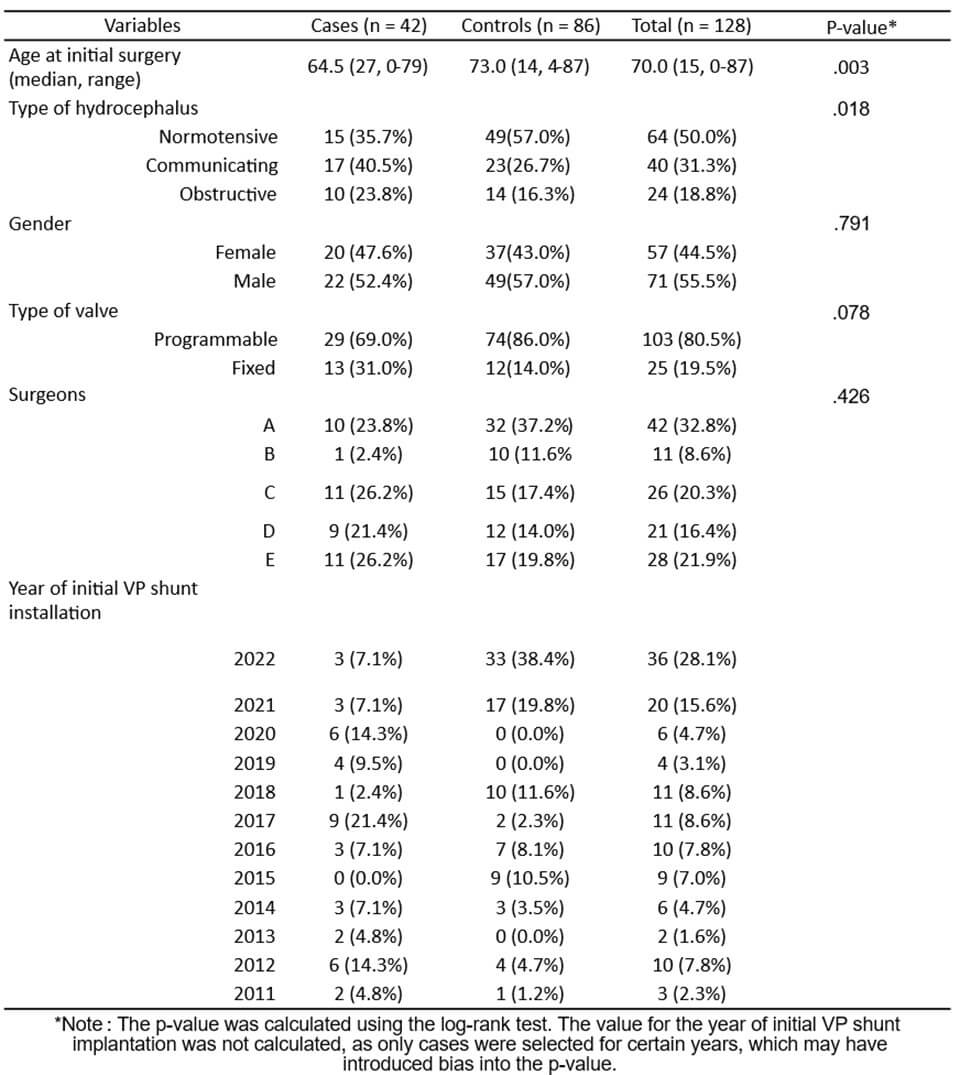
Most patients in this study were male (55.5%), and it was the same in both groups (52.4% male for cases and 57.0% for controls). There was therefore no statistical difference in gender between the groups (p = .791).
Overall, 80.5% of patients received a programmable valve. Both cases and controls predominantly received programmable valves, with 69.0% in the case group and 86.0% in the control group (p = .078).
Among surgeons, surgeon A performed the highest number of implantation surgeries, operating on 32.8% of patients. Surgeon E followed, accounting for 21.9% of shunt procedures. In the control group, a similar distribution was observed, with 37.2% of patients operated on by surgeon A and 19.8% by surgeon E. Among selected cases, surgeons C and E were the most represented, each performing surgeries on 26.2% of patients, followed by surgeon A with 23.8%.
Most patients included in this study underwent initial VP shunt installation in 2022 (28.1%) or 2021 (15.6%). The control group also predominantly consisted of patients from these two years. Selected cases predominantly included patients who had their initial shunt placement in 2017 (21.4%), 2020 (14.3%) and 2012 (14.3%).
The cause of the first revision surgery was identified from patients’ medical records (Table 2). The most common cause was VP shunt infection (35.7%). The second most frequent category was ‘’Not specified in the files’’, accounting for 33.3% of cases. Abdominal dysfunctions followed, representing 14.3% of revision causes. Thirteen out of the 14 patients with an unspecified dysfunction cause documented in the medical records also had air in the valve after revision surgery.
The list of components of the shunt system replaced during revision surgery was also extracted from the medical records. In 40.5% of cases, the complete shunt system was replaced. The distal catheter alone was replaced in 26.2% of revision surgeries, and the valve alone in 19.0%. When combining categories, a total of 29 out of 42 patients underwent valve replacement, representing 69.1% of the revision surgeries.
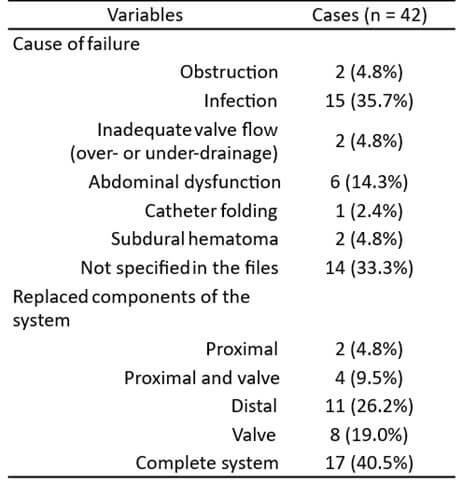
Age (log-rank p = .003) and type of hydrocephalus (log-rank p = .018) were statistically significantly associated with the risk of shunt revision, while the type of valve had no impact (log-rank p = .078) (Figure 4).
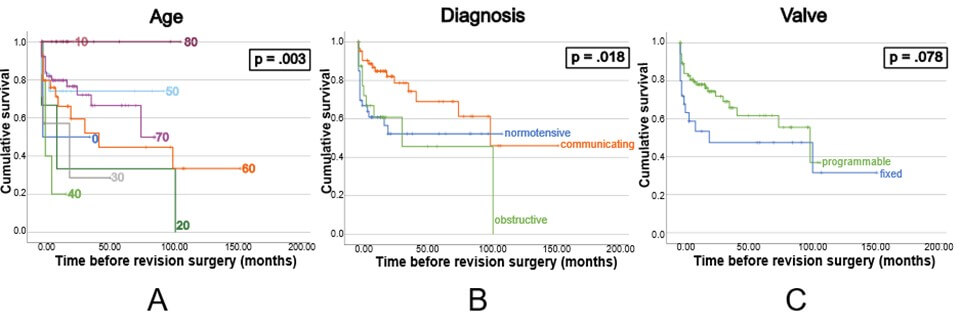
The effect of air in the VP shunt system was then analyzed. Air was present in 84% of investigated valves (93% in cases vs 80% in controls, chi-square p = .065). The volume of air (mL3) in the valve was similar between cases (median 27.51, range 0-243.93, IQR 47.53) and controls (median 23.92, range 0-215.04, IQR 59.51). Air was also present in the ventricles of 30.5% of patients after the installation surgery (28.6% in cases and 31.4% in controls). The volume of air in the ventricles was similar between cases (median 0, range 0-606.31, IQR 14.50) and controls (median 0, range 0-544.24, IQR 7.80). A Mann-Whitney U test for independent groups demonstrated that the occurrence of revision surgery is not linearly correlated with the amount of air present in the valve post-implantation (Supplementary Digital Content 2). Instead, an all-or-nothing effect was observed, as confirmed by a chi-square test examining the relationship between the presence of air in the valve (yes or no) and revision surgery, which yielded a p-value of .065. The same test was conducted for the presence of air in the ventricles, with a p-value of .744.
Actuarial analysis demonstrated that, after shunt implantation, the presence of air in the valve, but not in the ventricle, was associated with an increased chance of revision surgery (Figure 5).
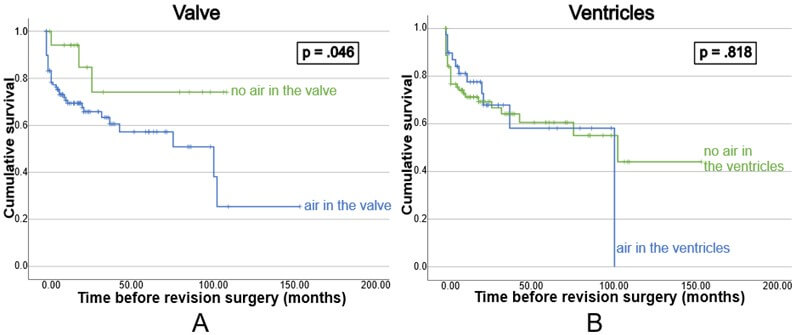
Binomial Logistic Regression analysis (multivariate) showed that patient age at the time of shunt insertion and the presence of air in the valve after surgery remained statistically significantly associated with revision surgery (Table 3). The type of hydrocephalus, however, is no longer statistically significant in the multivariate model (p = .887). Table 3 also indicates that when air is present in the valve postoperatively, there is 4.2 times higher likelihood (effect size) of revision surgery than if there is no air (p = .047; 95% CI = 1.022-17.295).
4. Discussion
The review of medical records for this research revealed that a substantial proportion of shunt malfunctions remain unexplained. Approximately one-third of revision surgeries were performed without a clearly identified cause (Table 2). Among factors that were analyzed, the presence of air within the valve was the most significant predictor of shunt dysfunction. We suspect that presence of air may lead to a phenomenon known as “air bubble train failure”. This type of malfunction may go undetected both radiologically and intraoperatively, as the catheters are opaque and radiopaque. In these cases, the valve may appear functional, depressible on examination, and CSF flow may still be observed at the distal end of the peritoneal catheter as the valve pressure intermittently overcomes the air obstruction. This mechanism could account for some of the otherwise unexplained shunt malfunctions.
Parameters influencing the probability of revision surgery were first evaluated individually. Advance age was associated with fewer revision surgeries, as reported in the literature [6]. Three age clusters emerged (20-40, 50-70, and 80 years), though the small sample of patients under 50 makes results from this subgroup less reliable. The type of hydrocephalus is also shown to influence the incidence and median time to shunt revision [3]. Patients with normal pressure hydrocephalus have fewer failures compared to communicating or obstructive hydrocephalus. Additionally, the valve type (programmable vs. fixed) was reported to impact VP shunt revision rates with programmable valves being associated with a reduced risk [6, 7]. In our cohort, a difference can be observed in the graph (Figure 4C); however, this difference did not reach statistical significance.
Kaplan-Meier survival curves (Figure 4) show that most dysfunctions occur in the first few months following shunt insertion. In our patient cohort, the median interval between initial VP shunt placement and the first revision surgery was 3 months, which is consistent with previously reports, where the median time to first shunt revision ranges from .5 to 9.31 months [3, 8]. This early failure pattern may be explained by improper valve function from the outset. While patients may initially experience symptom improvement due to CSF loss during surgery, the shunt may not have functioned correctly postoperatively, leading to early dysfunction. This may also explain the non-linear distribution for failures, which cluster shortly after implantation.
In the first few months of use, patient movement may facilitate the displacement of air from the valve into the catheters, increasing the risk of air obstruction in the small-diameter tubes. This explains why dysfunction may not occur immediately after the installation surgery.
Air bubble obstruction is an easily preventable cause of dysfunction. To mitigate the risk, we recommend carefully purging the valve during surgery. Specifically, we suggest aspirating any residual air bubbles in the valve reservoir with a 25-gauge or smaller needle before wound closure.
However, as previously mentioned, this type of dysfunction is difficult to detect radiologically when a patient presents clinical signs of shunt failure, as air within the catheters is typically not visible on CT imaging. Clinicians should consider this mechanism when a patient presents symptom recurrence or worsening within the first few months after shunt placement. In such cases, bedside purging of the system may be attempted by manually pumping the valve reservoir 10 times [5].
4.1. Limitations
This study has certain limitations worth mentioning. First, it is a 12-year retrospective study. The documentation of the causes and locations of dysfunction was not standardized. In addition, the study only included patients who had a shunt system implanted at the Centre Hospitalier Universitaire de Sherbrooke, in Quebec, Canada. This may introduce a selection bias, as surgical techniques can vary between surgeons and institutions.
The selective inclusion explained in the methods section may have introduced a selection bias, although this approach was necessary to achieve the required number of revision cases. Selecting patients over several years (from 2011 to 2022) allowed us to better capture the variability in surgical practices across different surgeons and over time.
Another limitation is that not all patients had long-term follow-up. However, since air bubble dysfunction, caused by air insertion during the initial surgery should typically manifests in the first few months, this limitation is likely to have less impact on our study.
5. Conclusion
Air bubble in a VP shunt system is a possible cause of shunt failure and may account for up to 33% of currently unexplained dysfunctions. In our patient series, postoperative air in the valve increases the likelihood of revision surgery by 4.2 times. Age was also associated with revision surgery in our binomial logistic regression analysis. Neurosurgeons should consider this potential failure mechanism during both VP shunt implantation and when investigating postoperative dysfunction.
Conflicts of Interest
None.
Funding
This research was supported by NSERC Grant RGPIN-2019-06656; by the Bourse Technologies au service de la santé de la Fondation Huguette et Jean-Louis Fontaine.
Author Contributions
Anne-Sophie Pelletier : Study conception, data collection, data analysis and manuscript writing. Andréanne Hamel : Data collection. Denis Rancourt : Study conception and design, manuscript writing. Christian Iorio-Morin : Study conception and design, data analysis or interpretation, manuscript writing.
Registration of Research Studies
Given the retrospective nature of the study, the study was not registered. However, the study was approved and recorded with the Research Project Authorization Office of the CIUSSS de l’Estrie - CHUS and with the Research Ethics Board of the CIUSSS de l’Estrie - CHUS.
Consent
Given the retrospective nature of the study, the requirement for informed consent was not needed.
Ethical Approval
IRB#00003849. Authorization number: 55736.
Abbreviations
CSF: Cerebrospinal Fluid
CHUS: Centre Hospitalier Universitaire de Sherbrooke (Québec, Canada)
CT scan: Computer Tomography Scan
IQR: Interquartile Range
SE: Standard Error
VP shunt: Ventriculoperitoneal Shunt
REFERENCES
[1] Lauren N Schulz, Asma
Redwan, Sara Edwards, et al. “Hydrocephalus Pathophysiology and Epidemiology.” Neurosurg
Clin N Am, vol. 36, no. 2, pp. 113-126, 2025. View at: Publisher Site | PubMed
[2] Albert
M Isaacs, Runze Yang, Magalie Cadieux, et al. “Characteristics of shunt failure in
38,095 adult shunt insertion surgeries: a systematic review and meta-analysis.”
Neurosurg Focus, vol. 54, no. 4, pp. E2, 2023. View at: Publisher Site | PubMed
[3] G Kesava Reddy, Papireddy
Bollam, Runhua Shi, et al. “Management of Adult Hydrocephalus With
Ventriculoperitoneal Shunts: Long-term Single-Institution Experience.” Neurosurgery,
vol. 69, no. 4, pp. 774-781, 2011. View at: Publisher Site | PubMed
[4] Morgan
Broggi, Costanza M Zattra, Marco Schiariti, et al. “Diagnosis of Ventriculoperitoneal
Shunt Malfunction: A Practical Algorithm.” World Neurosurgery, vol. 137,
pp. e479-e486, 2020. View at: Publisher Site | PubMed
[5] Marouane Makhchoune,
Michel Triffaux, Triantafyllos Bouras, et al. “Obstruction of
ventriculoperitoneal shunt by air bubble: A case report.” Ann Med Surg
(Lond), vol. 81, pp. 104351, 2022. View at: Publisher Site | PubMed
[6] Geraint J Sunderland,
Elizabeth J Conroy, Alexandra Nelson, et al. “Factors affecting
ventriculoperitoneal shunt revision: a post hoc analysis of the British
Antibiotic and Silver Impregnated Catheter Shunt multicenter randomized
controlled trial.” J Neurosurg, vol. 138, no. 2, pp. 483-493, 2022. View
at: Publisher Site | PubMed
[7] Maria Mpakopoulou,
Alexandros G Brotis, Haralampos Gatos, et al. “Ten years of clinical experience
in the use of fixed-pressure versus programmable valves: a retrospective study
of 159 patients.” Acta Neurochir Suppl, vol. 113, pp. 25-28, 2012. View
at: Publisher
Site | PubMed
[8] G Kesava Reddy “Ventriculoperitoneal shunt surgery and the incidence of shunt revision in adult patients with hemorrhage-related hydrocephalus.” Clin Neurol Neurosurg, vol. 114, no. 9, pp. 1211-1216, 2012. View at: Publisher Site | PubMed
