Received: Fri 14, Nov 2025
Accepted: Tue 09, Dec 2025
Abstract
The supraorbital keyhole approach (SKA) is recognized as a minimally invasive procedure for managing anterior circulation aneurysms, particularly when performed by experienced neurosurgeons. This retrospective, non-randomized, cross-sectional study compared 30 cases of SKA in ruptured anterior circulation aneurysms with 32 cases managed using the conventional pterional approach (PtA). Both groups were comparable in preoperative characteristics. Intraoperatively, SKA demonstrated significant advantages over PtA in operative time (72.17 ± 38.48 vs. 108.12 ± 44.79 minutes) and blood loss (649.67 ± 1224.12 vs. 909.22 ± 890.33 mL). Postoperatively, SKA showed fewer complications and better six-month functional outcomes.
In conclusion, the minimally invasive SKA is a valuable operative technique for anterior and selected middle skull base lesions. When performed by an experienced surgeon, this approach offers improved safety, reduced morbidity, and superior cosmetic results.
Keywords
Supraorbital keyhole approach, minimal invasive surgery, intracranial aneurysm
1. Introduction
A ruptured intracranial aneurysm is a neurosurgical emergency. Delayed or inappropriate management can lead to serious sequelae, including increased intracranial pressure, focal neurological deficits, cerebral infarction, seizures, vasospasm, or death [1]. Currently, two primary treatment modalities are available: surgical transcranial clipping and endovascular coiling. The endovascular method is preferable for deep-seated, narrow-necked saccular aneurysms, patients with lower-grade subarachnoid hemorrhage (SAH), and those without significant intracranial pathology such as massive intracerebral hemorrhage (ICH) or cerebral infarction [1, 2]. Conversely, transcranial approaches allow prompt management of raised intracranial pressure and reduction of SAH clot burden, with higher rates of complete aneurysm obliteration, albeit with greater risk of brain manipulation [3, 4]. The International Subarachnoid Aneurysm Trial (ISAT) demonstrated that recurrence and rebleeding rates were higher following endovascular therapy than surgical clipping [5]. Moreover, transcranial surgery is available in most hospitals without the need for specialized interventional facilities.
Minimally invasive microsurgical (MIM) techniques for aneurysm clipping were first introduced by Yasargil [6] as alternatives to conventional craniotomy. These approaches aim to reduce brain manipulation, minimize surgical trauma, and facilitate faster recovery. This study compares the outcomes of minimally invasive SKA and conventional PtA for clipping anterior circulation aneurysms, detailing the operative techniques step-by-step.
2. Materials and Methods
This retrospective, non-randomized, cross-sectional study included all patients with ruptured anterior circulation aneurysms (involving the ICA, ACA, ACOM, and selected PCOM aneurysms) who underwent surgery between August 2019 and December 2024 at Mahasarakham Hospital, a general hospital in Thailand. After receiving counseling, patients and their families chose between the minimally invasive supraorbital keyhole approach (SKA) and the conventional pterional approach (PtA). Preoperative profiles were compared, including age, sex, aneurysm type and side, Fisher grade, Glasgow Coma Scale (GCS), and modified World Federation of Neurosurgical Societies (m-WFNS) score.
The m-WFNS grading system categorizes aneurysmal SAH prognosis as follows: Grade 1 = GCS 15, Grade 2 = GCS 14, Grade 3 = GCS 13, Grade 4 = GCS 7-12, and Grade 5 = GCS 3-6. Lower grades indicate better prognosis [7, 8]. Fisher grading classifies the amount of SAH on CT brain, with Grade 3 showing the highest incidence of symptomatic vasospasm [9]. Intraoperative parameters included operative time (OPT), blood loss (BL), and intraoperative complications. Postoperative outcomes included immediate complications, status at discharge (D/C), and length of hospital stay (LOS). Long-term outcomes were evaluated at six months using the modified Rankin Scale (mRS) and cosmetic satisfaction.
2.1. Surgical Techniques
2.1.1. Conventional Pterional Approach (PtA)
A frontotemporal skin incision was made, followed by reflection of the scalp flap and dissection of the temporalis muscle to expose the bone. A frontotemporal bone flap was created, and the dura was widely opened. The subfrontal corridor was used for microsurgical dissection, aneurysm clipping, and closure of all layers in the standard manner.
2.1.2. Minimally Invasive Supraorbital Keyhole Approach (SKA)
This technique emphasizes a compact, targeted exposure (Figure 1). Patient positioning varied with aneurysm type: 15-30° lateral rotation for ACA and ACOM aneurysms, and 30-45° for ICA and PCOM aneurysms. The head was elevated 30-45° above the heart with neck extension. A curvilinear skin incision was made along the outer half of the eyebrow, avoiding the supraorbital nerve (Figure 1a). The subgaleal layer was dissected inferiorly along the superior orbital rim and superiorly for approximately 5 cm. The scalp was tented with 1-0 silk sutures. The temporalis muscle was incised approximately 3 cm from muscle origin while preserving its origin for resuturing (Figure 1b).
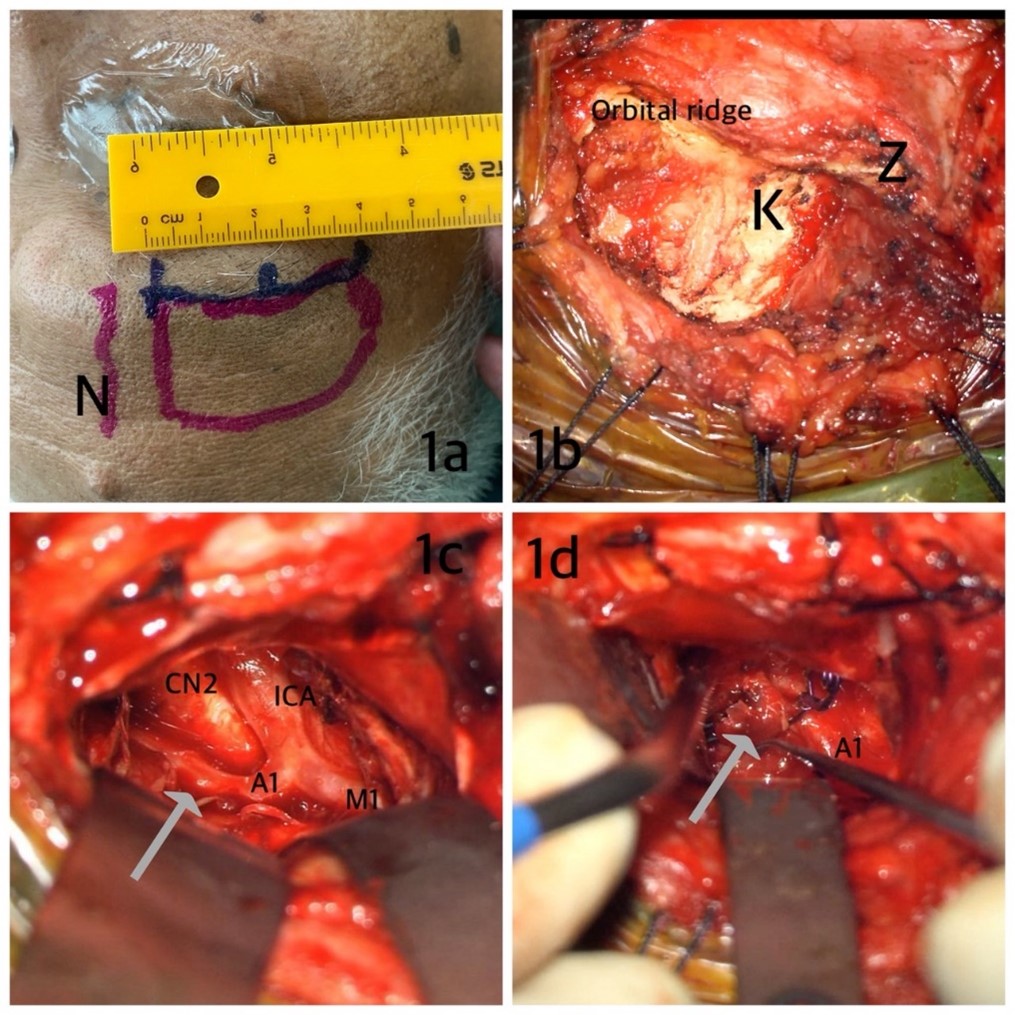
A burr hole was made at the sphenopterional (keyhole) point—an area providing simultaneous access to the anterior and middle cranial fossae [10]. A 5 cm bone flap was created along the superior lateral orbital ridge, and the sphenoid wing was flattened with a high-speed drill to widen the surgical corridor. The dura was opened in a U-shaped fashion based on the orbital ridge and tented with 3-0 silk sutures. Mannitol was administered pre-dural opening to facilitate brain relaxation. In cases of brain swelling, external CSF drainage (ventriculostomy or lumbar drainage) was employed. A subfrontal approach was performed under a microscope using self-retaining retractors (Figure 1c). Sharp arachnoid dissection along the sphenoid wing allowed identification of the optic nerve-ICA complex and CSF release, facilitating further relaxation. Two or three retractors provided adequate working space for bimanual microsurgery (Figures 1c & 1d).
The vascular structures—including both ICAs, A1 segments, ACOM, and ipsilateral PCOM—along with the optic apparatus and lamina terminalis, were identified. In thick SAH, opening the lamina terminalis aided brain relaxation. Aneurysm clipping was performed using the two-hand technique (Figure 1d). After clip application, the subarachnoid clot was irrigated with normal saline, and meticulous hemostasis was confirmed. The dura, bone flap, muscle, and scalp were closed in standard fashion. A subgaleal drain was generally unnecessary; gentle pressure dressing was sufficient.
Postoperative period in ICU are closely follow up as the standard guideline [1] until discharge from the hospital. Rehabilitation program and community care are trained, follow up at least 6 months about functional status and cosmetic result.
3. Results
A total of 62 patients with ruptured anterior circulation aneurysms were admitted to Mahasarakham Hospital between August 2019 and January 2025. Of these, 32 patients underwent the conventional pterional approach (PtA), and 30 patients underwent the minimally invasive supraorbital keyhole approach (SKA). The majority of patients were female, with ages ranging from 23 to 79 years. Aneurysm locations included the internal carotid artery (ICA), anterior cerebral artery (ACA, A1 segment), anterior communicating artery (ACOM), and selected posterior communicating artery (PCOM) aneurysms. Most cases presented with Fisher grades 2 or 3. The most common presenting symptoms were sudden severe headache and altered consciousness. Preoperative severity, as measured by the modified World Federation of Neurosurgical Societies (m-WFNS) scale, was variable across both groups. Baseline characteristics between the SKA and PtA groups were not significantly different (Table 1).
Table. 1. Pre-operative profiles in patients with PtA
and SKA (n=62).
|
characteristics |
PtA (n=32) |
SKA (n=30) |
|
Age (years) |
56.84 (26-79) |
54.37 (23-77) |
|
Sex (no.) female |
18 (56.3%) |
18 (60.0%) |
|
male |
14 (43.8%) |
12 (40.0%) |
|
Fisher grade
(no.) 1 |
2 (6.3%) |
1 (3.3%) |
|
2 |
10 (31.3%) |
12 (40.0%) |
|
3 |
12 (37.5%) |
12 (40.0%) |
|
4 |
8 (25.0%) |
5 (16.7%) |
|
Location of
aneurysms (no.) ACA (A1, ACOM) ICA PCOM |
19 (59.4%) 11 (34.4%) 2 (6.3%) |
23 (76.7%) 6 (20.0%) 1 (3.3%) |
|
Side (no.) left right |
16 (50.0%) 16 (50.0%) |
7 (23.3%) 23 (76.7%) |
|
m-WFDS (no.) 1 2 3 4 5 |
6 (18.8%) 4 (12.5%) 8 (25.0%) 6 (18.8%) 8 (25.0%) |
5 (16.7%) 12 (40.0%) 5 (16.7%) 3 (10.0%) 5 (16.7%) |
Detailed operative findings are summarized in (Table 2). The mean operative time was significantly shorter for SKA than for PtA (72.17 ± 38.48 vs. 108.12 ± 44.79 minutes). Estimated blood loss was also lower in the SKA group (649.67 ± 1224.12 mL vs. 909.22 ± 890.33 mL). The main intraoperative complications were aneurysm rupture and marked brain swelling secondary to infarction in cases with severe vasospasm. In the SKA group, two cases experienced intraoperative rupture with controlled bleeding (1,000 mL and 2,700 mL). Excluding these two cases, the mean blood loss among the remaining 28 SKA cases was 241.92 mL.
Table. 2. Intra-operative and post-operative findings.
|
Variable |
PtA (n=32) |
SKA (n=30) |
|
|
Operation time
(minutes), mean±SD |
108.12±44.79 |
72.17±38.48 |
|
|
Blood loss (mL),
mean±SD |
909.22±890.33 |
649.67±1224.12 |
|
|
Intra-operative
complication, (no.) none rupture mark brain
swelling due to infartion |
20 (62.5%) 2 (6.3%) 10 (31.3%) |
26 (86.7%) 3 (10.0%) 1 (3.3%) |
|
|
Postoperative
complication, (no.) none seizure infarction other (HAP,
sepsis) |
11 (34.4%) 1 (3.1%) 10 (31.3%) 10 (31.3%) |
23 (76.6%) - 5 (16.7%) 2 (6.7) |
|
|
Result at d/c,
(no.) Dead Improve Bed ridden |
9 (28.1%) 22 (68.8%) 1 (3.1%) |
5 (16.7%) 25 (83.3%) - |
|
|
LOS (day), Min-max |
2-50 |
2-35 |
|
|
mRS at 6-month,
(no.) 0 1 2 3 4 5 6 |
2 (6.3%) 6 (18.8%) 7 (21.9%) 2 (6.3%) 3 (9.4%) 2 (6.3%) 10 (31.3%) |
5 (16.7%) 15 (50.0%) 2 (6.7%) 2 (6.7%) 1 (3.3%) - 5 (16.7%) |
|
Postoperative complications were slightly less frequent in the SKA group. The major postoperative events were infarction and hospital-acquired pneumonia (HAP). Vasospasm was observed in both groups and was associated with thick subarachnoid clots, delayed surgery, and intraoperative hypotension. Overall, most patients in both groups showed improvement at discharge. All fatalities were due to early cerebral infarction. At six-month follow-up, nearly all surviving patients had improved functional status, with some having no major neurological deficits.
As summarized in Table 3, both surgical groups had comparable baseline characteristics, including age, sex, Fisher grade, m-WFNS score, and aneurysm type. Postoperative length of stay and discharge status were also not significantly different. However, the SKA technique demonstrated significant advantages in shorter operative time, reduced intraoperative blood loss, fewer postoperative complications, and improved six-month functional recovery (mRS) (p < 0.05).
Table. 3. Comparitive profiles of both operation.
|
Variable |
PtA (n=32) |
SKA (n=30) |
P value |
|
Ageb |
33.31 |
29.57 |
.414 |
|
Sexb |
30.94 |
32.10 |
.767 |
|
Diagnosis (type of
aneurysm) a |
2.41 |
2.17 |
.289 |
|
Fisher gradeb |
32.72 |
30.20 |
.560 |
|
m-WFDSb |
34.50 |
28.30 |
.167 |
|
Operation time
(minute) skin-to-skin a |
108.13 |
72.17 |
<.001* |
|
Blood loss(mL) b |
36.28 |
26.40 |
.031* |
|
LOS (day)a |
18.09 |
15.23 |
.329 |
|
Result at d/ca |
1.75 |
1.83 |
.465 |
|
mRS at 6-monthb |
37.48 |
25.12 |
.005* |
|
Postoperative
complicationb |
11.36 |
6.57 |
.018* |
|
|
|
|
|
a Independent-samples t-test ; b Mann-Whitney U
test
*p < 0.05 was considered statistically
significant.
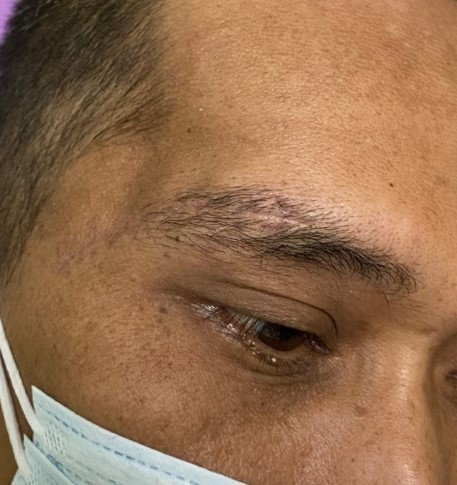
Long-term wound and cosmetic outcomes were satisfactory in all patients (Figure 2). At six months postoperatively, the surgical incision line was well-hidden along the eyebrow, with only minimal skin and scalp atrophy and no prominent aesthetic defects. Patients expressed high satisfaction with the small skull flap and short skin incision. None reported facial pain, masticatory discomfort, or numbness during facial movements.
4. Discussion
Ruptured intracranial aneurysm is a life-threatening neurosurgical emergency. Timely and appropriate management can restore patients to normal life, whereas delayed or inadequate treatment may result in disability or death [1, 11]. Computed tomography (CT) of the brain is the initial imaging modality for diagnosis. The Fisher scale [12] classifies aneurysmal subarachnoid hemorrhage (SAH) by clot thickness and predicts vasospasm risk. The grades are divided as grade 1 (not seen SAH or IVH), grade 2 (diffuse thin <1 mm SAH), grade 3 (localized clot or > 1 mm SAH thickness), and grade4 (diffuse SAH with IVH or ICH). The incidence of symptomatic vasospasm increases with higher Fisher grades—21%, 25%, 37%, and 31%, respectively [13]. Vasospasm is a major complication in aneurysmal SAH that cause arterial infarction especially unappropriated management. Clinically, the modified World Federation of Neurosurgical Societies (m-WFNS) scale grades patients based on the Glasgow Coma Scale (GCS); grades 3-5 (GCS ≤13) are associated with poorer outcomes [7, 8]. These grading systems are crucial for guiding treatment selection and counseling patients’ families regarding prognosis.
Surgical clipping remains a definitive treatment for aneurysmal rupture, effectively preventing rebleeding. This study compared the minimally invasive supraorbital keyhole approach (SKA) with the conventional pterional approach (PtA) for ruptured anterior circulation aneurysms. The SKA demonstrated clear advantages in shorter operative time, reduced blood loss, fewer postoperative complications, and superior six-month functional recovery (mRS). Findings from previous studies align with our results, showing that minimally invasive approaches, particularly the SKA, yield faster recovery, less tissue trauma, and better cosmetic outcomes than traditional craniotomy (Table 4).
Table. 4. Summary of related studies comparing minimally
invasive and conventional approaches.
|
Author |
Year |
N |
Outcome |
||
|
Shao et al.
[14] |
2021 |
235 |
Keyhole approach
provided faster recovery and favorable surgical outcomes (OPT 148 ± 47 min,
BL 201 ± 98 mL). Safe and effective for anterior circulation aneurysms. |
||
|
Park et al.
[15] |
2009 |
108 |
Supraorbital
approach reduced intra- and postoperative complications, shortened operative
time, and improved cosmetic results (mean OPT 160 min). |
||
|
Genesan et al.
[16] |
2018 |
123 |
SKA patients had
better cosmetic outcomes and shorter operations (OPT 192 min, BL 433 mL). No
significant difference in major complications. |
||
|
Fonseca et al.
[17] |
2021 |
35 |
|
||
|
Cha et al.
[18] |
2012 |
137 |
Lateral
supraorbital (LSO) approach yielded shorter hospitalization and operative
time (mean 117.1 min). |
||
|
Noiphitak et
al. [19] |
2020 |
102 |
After propensity
score matching, minimally invasive craniotomy showed shorter operative time
(2.8 vs. 4.2 h), shorter hospital stay, and fewer hemorrhagic complications. |
||
|
Harris et al.
[20] |
2025 |
90 |
MIS showed shorter
operative time (310 vs. 383 min) and hospital stay; estimated blood loss was
not significantly different. |
||
|
Rocca et al.
[21] |
2018 |
50 |
LSO approach
reduced early complications, improved masticatory comfort and cosmetics; a
safe and effective alternative to PtA. LSO is safe and effective substitute
to standard PT craniotomy to treat unruptured ACOM and A1/A2 aneurysms. |
||
|
Lan et al.
[22] |
2006 |
105 |
Keyhole approaches
are effective and ideal in the hands of experienced microsurgeons. |
||
|
Mandel et al.
[23] |
2021 |
111 |
MIS superior to
PtA for small UIAs regarding cosmetic satisfaction and quality of life. |
||
|
Chalouhi et al.
[24] |
2013 |
87 |
SKA had shorter
operative time (205 vs. 256 min) with slightly higher procedural
complications; acceptable for small, simple aneurysms. |
||
|
Lindert et al.
[25] |
1998 |
139 |
SKA provided
equivalent exposure with fewer intraoperative ruptures and less
approach-related morbidity than PtA. |
With advances in microsurgical optics and instruments, neurosurgeons can now achieve a wide operative field through a small bony window [26]. This approach reduces brain parenchymal injury, minimizes exposure to the environment, and lowers postoperative complication rates [14, 27]. Regardless of the craniotomy size, both SKA and PtA provide access to the subfrontal region around the keyhole point. When adequate brain relaxation is achieved, the minimally invasive approach can provide the same exposure as a conventional craniotomy. Florez-Perdomo et al. [29] performed a systematic review and meta-analysis comparing SKA and PtA, concluding that the supraorbital approach reduces ischemic events and is a safe alternative to traditional methods. However, its use in ruptured aneurysms with cerebral edema or midline shift remains challenging. In experienced hands, SKA is a reliable and effective method for anterior circulation aneurysms and other skull base pathologies [14, 19].
The concept of “keyhole surgery” was first introduced by Wilson in 1971 [29]. Hernesniemi et al. [30], after more than 2,000 operations, described the lateral supraorbital approach as a simpler, faster, safer, and less invasive alternative to the classic PtA. For younger neurosurgeons, the main obstacles in adopting SKA are: limited confidence in managing intraoperative complications such as rupture or brain swelling, and concern about the cosmetic outcome.
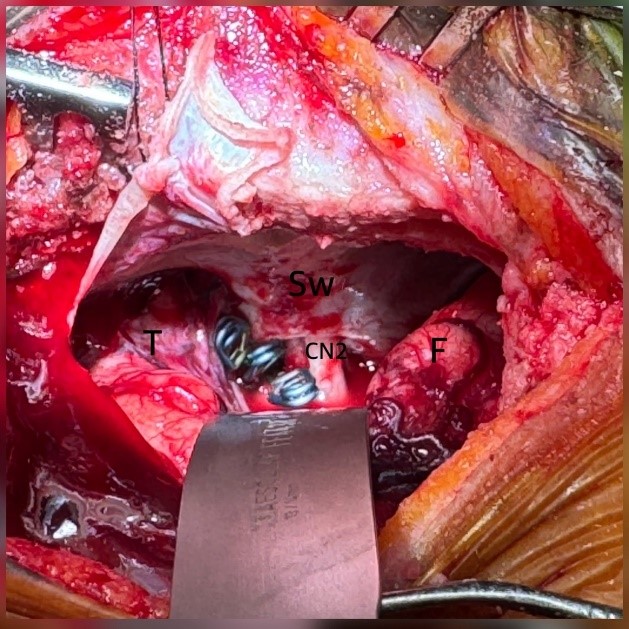
The key to a successful SKA lies in two main factors: adequate bone work and effective brain relaxation. Bone work: The craniotomy should extend inferiorly to the superior orbital ridge, medially to the lateral half of the superior orbital line, superiorly about 5 cm above the ridge, and laterally toward the anterior temporal fossa (keyhole point). The medial and superior edges should be beveled for smooth skull closure. Drilling and flattening the sphenoid wing is essential to widen the operative view across the anterior and middle cranial fossae (Figure 3). Brain relaxation: Techniques include external ventricular drainage (EVD), lumbar drainage, and osmotic therapy with mannitol or hypertonic saline are prefered. EVD is preferred when preoperative hydrocephalus is present [31, 32], while lumbar drainage is useful in mild hydrocephalus to reduce intracranial pressure [33, 34]. Lumbar drainage can reduce risk of delayed ischemic neurological deficit (DIND), both EVD and lumbar drainage are benefit for postoperative intracranial pressure monitor [35-38]. Osmotic agents further enhance relaxation when administered before dural opening [39-41]. Adequate arachnoid dissection is another critical step. The arachnoid membrane forms an envelope for vessels and bridging veins; careful sharp dissection with concurrent CSF release allows sufficient relaxation and safe exposure [14, 30]. The SKA provides a wide enough operative angle for bimanual microsurgical manipulation, enabling effective treatment of lesions in both the frontal and temporal fossae (Figure 3).
5. Conclusion
The minimally invasive supraorbital keyhole approach (SKA) is an excellent technique for managing anterior circulation aneurysms in the hands of experienced neurosurgeons. Compared to the conventional pterional approach, SKA offers significant advantages in operative time, blood loss, postoperative morbidity, and long-term functional recovery. Critical factors for success include meticulous bone exposure, sufficient sphenoid wing drilling, and appropriate management of brain swelling. The limitation of this study are single-center design, retrospective nature, surgeon experience variability. Beyond aneurysm surgery, the SKA can be effectively applied to other anterior and middle cranial fossa pathologies, such as tumors, third ventriculostomy via lamina terminalis fenestration, pituitary macroadenomas, and repair of cerebrospinal fluid leaks.
REFERENCES
[1] Brian L Hoh, Nerissa U Ko,
Sepideh Amin-Hanjani, et al. “2023 Guideline for the Management of Patients
With Aneurysmal Subarachnoid Hemorrhage: A Guideline From the American Heart
Association/American Stroke Association.” Stroke, vol. 54, no. 7, pp.
e314-e370, 2023. View at: Publisher
Site | PubMed
[2] Antti Lindgren, Mervyn DI
Vergouwen, Irene van der Schaaf, et al. “Endovascular coiling versus
neurosurgical clipping for people with aneurysmal subarachnoid haemorrhage.” Cochrane
Database Syst Rev, vol. 8, no. 8, pp. CD003085, 2018. View at: Publisher Site | PubMed
[3] L M Auer, M Mokry
“Disturbed cerebrospinal fluid circulation after subarachnoid hemorrhage and
acute aneurysm surgery.” Neurosurgery, vol. 26, no. 5, pp. 804-808,
1990. View at: Publisher
Site | PubMed
[4] Y Handa, B K Weir, M
Nosko, et al. “The effect of timing of clot removal on chronic vasospasm in a
primate model.” J Neurosurg, vol. 67, no. 4, pp. 558-564, 1987. View at:
Publisher Site | PubMed
[5] Andrew J Molyneux, Richard
S C Kerr, Jacqueline Birks, et al. “Risk of recurrent subarachnoid haemorrhage,
death, or dependence and standardised mortality ratios after clipping or
coiling of an intracranial aneurysm in the International Subarachnoid Aneurysm
Trial (ISAT): long-term follow-up.” Lancet Neurol, vol. 8, no. 5, pp.
427-433, 2009. View at: Publisher
Site | PubMed
[6] M G Yasargil, J L Fox “The
microsurgical approach to intracranial aneurysms.” Surg Neurol, vol. 3,
no. 1, pp. 7-14, 1975. View at: PubMed
[7] Hirotoshi Sano, Akira
Satoh, Yuichi Murayama, et al. “Modified World Federation of Neurosurgical
Societies subarachnoid hemorrhage grading system.” World Neurosurg, vol.
83, no. 5, pp. 801-807, 2015. View at: Publisher Site | PubMed
[8] “Report of World
Federation of Neurological Surgeons Committee on a Universal Subarachnoid
Hemorrhage Grading Scale.” J Neurosurg, vol. 68, no. 6, pp. 985-986,
1988. View at: Publisher
Site | PubMed
[9] Gaillard F. Radiopaedia
“Fisher scale of subarachnoid hemorrhage | Radiology Reference Article.
[10] Baltazar Leão Reis,
Roberto Leal Da Silveira, Sebastião Nataniel Silva Gusmão “Sphenopterional
Point: Strategic Point for Burr Role Placement in Frontotemporal Craniotomies.”
World Neurosurg, vol. 105, pp. 399-405, 2017. View at: Publisher Site | PubMed
[11] Sebastian Sanchez, Jacob
M. Miller, Edgar A. Samaniego “Clinical Scales in Aneurysm Rupture Prediction.”
Stroke: Vascular and Interventional Neurology, vol. 4, no. 1, pp.
e000625, 2023. View at: Publisher
Site
[12] C M Fisher, J P Kistler, J
M Davis “Relation of cerebral vasospasm to subarachnoid hemorrhage visualized
by computerized tomographic scanning.” Neurosurgery, vol. 6, no. 1, pp.
1-9, 1980. View at: Publisher
Site | PubMed
[13] Jennifer A Frontera
“Decision Making in Neurocritical Care.” Thieme, pp 356, 2011.
[14] Dongqi Shao, Yu Li,
Zhixiang Sun, et al. “Keyhole Approach for Clipping Anterior Circulation
Aneurysms: Clinical Outcomes and Technical Note.” Front Surg, vol. 8,
pp. 783557, 20221. View at: Publisher
Site | PubMed
[15] Heung Sik Park, Sang Kyu
Park, Young Min Han “Microsurgical Experience with Supraorbital Keyhole
Operations on Anterior Circulation Aneurysms.” J Korean Neurosurg Soc,
vol. 46, no. 2, pp. 103-108, 2009. View at: Publisher Site | PubMed
[16] Pravinna Genesan, Mohammad
Saffari Mohammad Haspani, Saiful Razman Mohd Noor “A Comparative Study between
Supraorbital Keyhole and Pterional Approaches on Anterior Circulation
Aneurysms.” MJMS, vol. 25, no. 5, pp. 59-67, 2018. View at: Publisher Site | PubMed
[17] Ricardo Brandão Fonseca,
Alyne Oliveira Correia, Raysa Siqueira Vieira, et al. “Comparative study
between minimally invasive supraorbital craniotomy and pterional craniotomy for
treating anterior circulation cerebral aneurysms in a low-resource setting.” Sci
Rep, vol. 11, no. 1, pp. 5555, 2021. View at: Publisher Site | PubMed
[18] Ki-Chul Cha, Seung-Chyul
Hong, Jong-Soo Kim “Comparison between Lateral Supraorbital Approach and
Pterional Approach in the Surgical Treatment of Unruptured Intracranial
Aneurysms.” J Korean Neurosurg Soc, vol. 51, no. 6, pp. 334-337, 2012.
View at: Publisher
Site | PubMed
[19] Raywat Noiphithak, Vich
Yindeedej, Warot Ratanavinitkul, et al. “Comparison of Minimally Invasive and
Pterional Craniotomies for Ruptured Anterior Circulation Aneurysms: A
Propensity Score Matched Analysis.” World Neurosurg, vol. 138, pp.
e289-e298, 2020. View at: Publisher
Site | PubMed
[20] Peter Harris, Hayes H
Patrick, Keaton Piper, et al. “Comparison of patient-centric factors in
minimally invasive transcranial versus classic approaches: a matched cohort
study.” J Neurosurg, vol. 143, no. 4, pp. 1048-1058, 2025. View at: Publisher Site | PubMed
[21] Giuseppe La Rocca,
Giuseppe Maria Della Pepa, Carmelo Lucio Sturiale, et al. “Lateral Supraorbital
Versus Pterional Approach: Analysis of Surgical, Functional, and
Patient-Oriented Outcomes.” World Neurosurg, vol. 119, pp. e192-e199,
2018. View at: Publisher
Site | PubMed
[22] Qing Lan, Zhigang Gong,
Dezhi Kang, et al. “Microsurgical experience with keyhole operations on
intracranial aneurysms.” Surg Neurol, vol. 66, pp. S2-S9, 2006. View at:
Publisher Site | PubMed
[23] Mauricio Mandel, Rafael
Tutihashi, Yiping Li, et al. “MISIAN (Minimally Invasive Surgery for Treatment
of Unruptured Intracranial Aneurysms): A Prospective Randomized Single-Center
Clinical Trial With Long-Term Follow-Up Comparing Different Minimally Invasive
Surgery Techniques with Standard Open Surgery.” World Neurosurg, vol.
151, pp. e533-e544, 2021. View at: Publisher Site | PubMed
[24] Nohra Chalouhi, Pascal
Jabbour, Ibrahim Ibrahim, et al. “Surgical Treatment of Ruptured Anterior
Circulation Aneurysms: Comparison of Pterional and Supraorbital Keyhole
Approaches.” Neurosurgery, vol. 72, no. 3, pp. 437, 2013. View at: Publisher Site | PubMed
[25] E van Lindert, A
Perneczky, G Fries, et al. “The supraorbital keyhole approach to supratentorial
aneurysms: concept and technique.” Surg Neurol, vol. 49, no. 5, pp.
481-489, 1998. View at: Publisher
Site | PubMed
[26] Kentaro Mori “Keyhole
concept in cerebral aneurysm clipping and tumor removal by the supraciliary
lateral supraorbital approach.” Asian J Neurosurg, vol. 9, no. 1, pp.
14-20, 2014. View at: Publisher
Site | PubMed
[27] Mohamed Elnokaly, Gordon
Mao 1, Khaled A Aziz “Transpalpebral Approach “Eyelid Incision” for Surgical
Treatment of Intracerebral Aneurysms: Lessons Learned During a 10-Year
Experience.” Oper Neurosurg, vol. 18, no. 3, pp. 309-315, 2020. View at:
Publisher Site | PubMed
[28] William A Florez-Perdomo,
Cesar E Zabala-Otero, Harrison R Herrea, et al. “Supraorbital vs pterional
keyhole for anterior circulation aneurysms: A systematic review and
meta-analysis.” World Neurosurg X, vol. 19, pp. 100177, 2023. View at: Publisher Site | PubMed
[29] D H Wilson “Limited
exposure in cerebral surgery. Technical note.” J Neurosurg, vol. 34, no.
1, pp. 102-106, 1971. View at: Publisher Site | PubMed
[30] J Hernesniemi, K Ishii, M
Niemelä, et al. “Lateral supraorbital approach as an alternative to the
classical pterional approach.” Acta Neurochir Suppl, vol. 94, pp. 17-21,
2005. View at: Publisher
Site | PubMed
[31] Giacomo Pavesi, Davide
Nasi, Elisa Moriconi, et al. “Management and safety of intraoperative
ventriculostomy during early surgery for ruptured intracranial aneurysms.” Acta
Neurochir (Wien), vol. 164, no. 11, pp. 2909-2916, 2022. View at: Publisher Site | PubMed
[32] Jae Hoon Kim, Hee In Kang
“Intraoperative Ventriculostomy Using K Point in Surgical Management of
Aneurysmal Subarachnoid Hemorrhage.” World Neurosurg, vol. 122, pp.
e248-e252, 2019. View at: Publisher
Site | PubMed
[33] Sunil Munakomi; Joe M. Das
“Brain Herniation.” In: StatPearls [Internet]. Treasure Island (FL): StatPearls
Publishing; 2025. View at: PubMed
[34] Toshitsugu Terakado,
Yoshiro Ito, Koji Hirata, et al. “Efficacy and Safety of Lumbar Drainage before
Endovascular Treatment for Ruptured Intracranial Aneurysms.” J Neuroendovasc
Ther, vol. 18, no. 2, pp. 29-36, 2024. View at: Publisher Site | PubMed
[35] E S Connolly Jr, A A
Kader, V I Frazzini, et al. “The safety of intraoperative lumbar subarachnoid
drainage for acutely ruptured intracranial aneurysm: Technical note.” Surg
Neurol, vol. 48, no. 4, pp. 338-344, 1997. View at: Publisher Site | PubMed
[36] Stefan Wolf, Dorothee
Mielke, Christoph Barner, et al. “Effectiveness of Lumbar Cerebrospinal Fluid
Drain Among Patients With Aneurysmal Subarachnoid Hemorrhage: A Randomized
Clinical Trial.” JAMA Neurol, vol. 80, no. 8, pp. 833-842, 2023 View at:
Publisher Site | PubMed
[37] Cheol Young Lee, Kyoung
Min Jang, Seong Hyun Wui, et al. “The Benefits and Feasibility of External
Lumbar Cerebrospinal Fluid Drainage for Cerebral Vasospasm in Patients with
Aneurysmal Subarachnoid Hemorrhage: Meta-Analysis and Trial Sequential Analysis.”
World Neurosurg, vol. 167, pp. e549-e560, 2022. View at: Publisher Site | PubMed
[38] Paul Klimo Jr, John R W
Kestle, Joel D MacDonald, et al. “Marked reduction of cerebral vasospasm with
lumbar drainage of cerebrospinal fluid after subarachnoid hemorrhage.” J
Neurosurg, vol. 100, no. 2, pp. 215-224, 2004. View at: Publisher Site | PubMed
[39] Yuming Peng, Xiaoyuan Liu,
Aidong Wang, et al. “The effect of mannitol on intraoperative brain relaxation
in patients undergoing supratentorial tumor surgery: study protocol for a
randomized controlled trial.” Trials, vol. 15, pp. 165, 2014. View at: Publisher Site | PubMed
[40] Hee-Pyoung Park “Erratum.
A prospective randomized trial of the optimal dose of mannitol for
intraoperative brain relaxation in patients undergoing craniotomy for
supratentorial brain tumor resection.” J Neurosurg, vol. 129, no. 2, pp.
551-552, 2018. View at: Publisher
Site | PubMed
[41] Hyungseok Seo, Eugene Kim, Haesun Jung, et al. “A prospective randomized trial of the optimal dose of mannitol for intraoperative brain relaxation in patients undergoing craniotomy for supratentorial brain tumor resection.” J Neurosurg, vol. 126, no. 6, pp. 1839-1846, 2017. View at: Publisher Site | PubMed
