Received: Thu 25, Sep 2025
Accepted: Thu 16, Oct 2025
Abstract
Objective: To evaluate the efficacy and safety of the "Catheter Shortening Method" for managing lumbar cistern drainage (LCD) catheter obstruction, compared to conventional treatment approaches.
Methods: A single-center retrospective case-control study was conducted. Patients requiring intervention for LCD catheter obstruction between January 2017 and December 2023 were included (47 patients, 64 obstruction events). Patients were allocated to two groups: Shortening Method Group (n=19): The catheter was incrementally shortened by approximately 1 cm segments from the distal end until cerebrospinal fluid (CSF) flow resumed, followed by connection to an extension catheter. Conventional Treatment Group (n=28): Patients underwent catheter replacement (34 instances) or conversion to external ventricular drainage (EVD) (4 instances). Primary outcomes included catheter patency restoration rate, re-obstruction rate, and frequency of invasive procedures.
Results: 1. Patency Restoration: The Shortening Method achieved 100% patency restoration in 19 patients (22 shortening procedures). 2. Re-obstruction Rate: The rate of secondary obstruction was significantly lower in the Shortening Method group than in the Conventional Treatment group (10.5% vs. 34.6%, p < 0.05). 3. Procedural Burden: The Conventional Treatment group required an average of 1.36 invasive procedures per patient, with 14.3% converted to EVD. The Shortening Method avoided additional invasive access procedures.
Conclusions: The Catheter Shortening Method is a simple, minimally invasive, and highly effective strategy for managing LCD catheter obstruction. It achieved a 100% success rate in restoring patency, significantly reduced the risk of re-obstruction and procedural burden compared to conventional re-catheterization or EVD conversion, and is particularly suitable for elderly, critically ill, or patients requiring prolonged drainage. The mechanism may relate to reducing catheter length (decreasing fluid resistance per Poiseuille's law) and mitigating issues related to protein deposition within the catheter lumen associated with temperature changes during CSF drainage.
Keywords
Lumbar cistern drainage, catheter obstruction, shortening method, cerebrospinal fluid (CSF) drainage, minimally invasive management
1. Introduction
Lumbar cistern drainage (LCD) entails the temporary placement of a fine catheter into the lumbar spinal canal connected to a closed system, facilitating cerebrospinal fluid (CSF) drainage. In 1963, Vourch first reported the continuous drainage of cerebrospinal fluid through a spinal canal catheter [1]. LCD is used to treat intracranial infection, vasospasm after hemorrhage, intractable intracranial hypertension following craniocerebral trauma, cerebrospinal fluid leakage, refractory intracranial hypertension in pediatric severe traumatic brain injury, and to prevent spinal cord ischemia in F/BEVAR [2-6]. Obstruction of the drainage system is a frequent mechanical complication. Common approaches to address obstruction include replacing the drainage catheter or initiating external ventricular drainage (EVD). However, these interventions not only increase treatment costs but also expose patients to additional discomfort and procedural risks. This study describes a validated simple and safe technique to manage LCD obstruction effectively.
2. Methods
2.1. Study Design and Reporting Guidelines
This retrospective case-control study was conducted in compliance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for cohort studies.
2.2. Ethical Considerations
The study protocol was approved by the Ethics Committee of Shandong Provincial Hospital, affiliated with Shandong First Medical University. The study adhered to the Declaration of Helsinki on medical research involving human participants. Written informed consent was obtained from all participants prior to the drainage procedure, in accordance with standard clinical protocols.
2.3. Study Population
We retrospectively reviewed data from consecutive patients who underwent continuous lumbar cistern drainage (LCD) in the Department of Neurosurgery at Shandong Provincial Hospital between January 2017 and December 2023. A total of 64 patients who required treatment for lumbar cistern drainage catheter obstruction were included in this analysis.
2.4. Inclusion and Exclusion Criteria
2.4.1. Inclusion Criteria
i) Patients who underwent LCD for the following indications:
ii) Central nervous system (CNS) infections requiring adjunctive drainage therapy.
iii) Subarachnoid hemorrhage (SAH) necessitating cerebrospinal fluid (CSF) diversion.
iv) Persistent post-operative subcutaneous fluid accumulation following craniotomy or spinal surgery.
v) Prevention of spinal cord ischemia after aortic surgery.
vi) Cerebrospinal fluid (CSF) leakage managed with therapeutic drainage.
2.4.2. Exclusion Criteria
i) Patients with unsuccessful catheter placement at insertion.
ii) Patients who expired from causes unrelated to drainage complications.
iii) Patients lost to follow-up.
iv) Inadvertent catheter dislodgement during the LCD procedure.
v) Patients managed with non-Medtronic® EDM™ systems.
2.5. Lumbar Cistern Drainage Equipment
The Medtronic® EDM™ Lumbar External Drainage and Monitoring System (Medtronic Inc.) used in this study consisted of the following components (Figure 1):
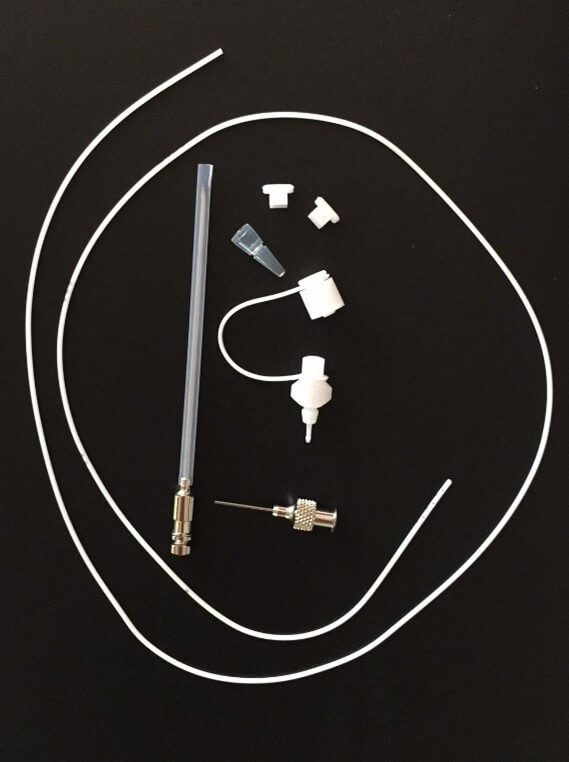
i) 14-gauge (F14) lumbar puncture needle (Tuohy needle).
ii) Lumbar puncture catheter with an outer diameter of 1.5 mm, inner diameter of 0.8 mm, and length of 80 cm.
2.6. Lumbar Cistern Catheterization Procedure
The catheterization procedure was performed as follows (Figure 2):
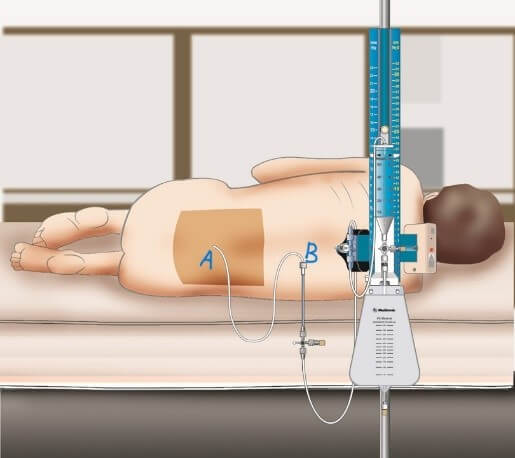
Patients were positioned laterally, and the skin was disinfected with antiseptic solution. Local infiltration anesthesia was administered using 2% lidocaine. A 14-gauge Tuohy needle was advanced into the L3-L4 or L4-L5 intervertebral space, and the subarachnoid space was confirmed by free-flowing cerebrospinal fluid (CSF). A multi-orifice catheter was threaded 15-20 cm caudally into the subarachnoid space. The catheter was sutured to the skin adjacent to the puncture site and secured with an integrated locking device and occlusive dressing. The catheter was connected via extension tubing with a three-way stopcock to a gravity-dependent collection system mounted on an infusion pole.
2.7. Management of the Lumbar Drainage System
The management of the lumbar drainage system was standardized as follows:
i) The drainage collection chamber was positioned vertically at the mid-axillary line, initially calibrated 10 cm above the external auditory meatus line.
ii) Drainage volume was recorded bi-hourly by trained nursing staff, who underwent standardized training for LCD system management.
iii) The drainage rate was maintained at 5-10 mL/hour (120-240 mL/day). If the output exceeded these parameters, adjustments to the chamber height (±5 cm) were made.
iv) Sterile dressings were changed every 5 days or sooner if compromised (e.g., saturation or detachment).
v) Patients were required to maintain strict bed rest in a supine position, except for essential procedures.
vi) Catheter clamping was permitted transiently when necessary.
2.8. Definition of Drainage Catheter Obstruction
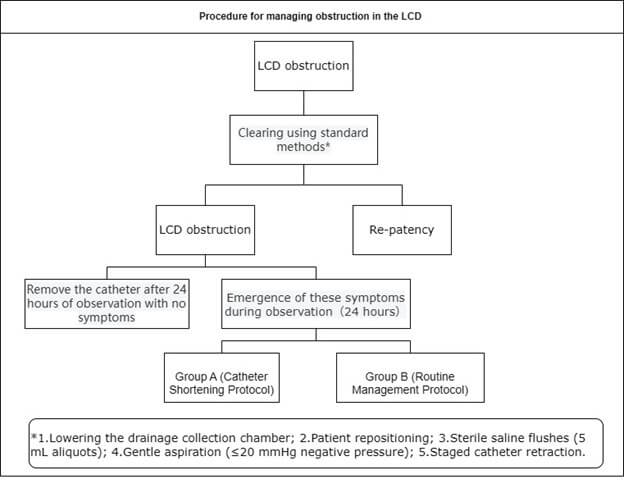
Catheter obstruction was defined as the absence of CSF outflow for ≥ 4 consecutive hours, despite following a standardized clearance protocol, which included:
i) Lowering the drainage collection chamber.
ii) Patient repositioning.
iii) Saline flushes.
iv) Gentle aspiration.
v) Staged catheter retraction.
If obstruction persisted despite these interventions, further management protocols were initiated.
2.9. Management of Catheter Obstruction
In the case of catheter obstruction, the catheter was retained in situ for a 24-hour observation period. Absence of new neurological symptoms (headache, nausea, altered consciousness) or systemic signs (fever >38°C) warranted catheter removal. Emergence of these symptoms during observation mandated escalation to therapeutic interventions, the following management strategies were employed:
• Group A (Catheter Shortening Protocol): The catheter was shortened distally (B-to-A) in 1 cm increments. This procedure continued until CSF flow was restored or the drainage rate reached ≥10 mL/hour. If CSF flow was restored, the catheter end was reconnected to the extension after disinfection.
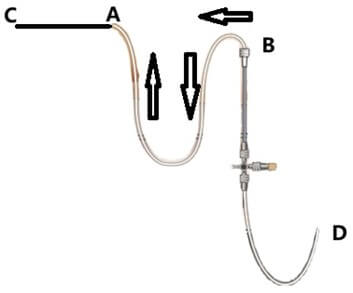
• Group B (Routine Management Protocol): If obstruction persisted, catheter replacement was performed at an adjacent spinal level. In cases where the CSF viscosity was high, external ventricular drainage (EVD) was considered.
2.10. Data Collection and Statistical Analysis
Patient data were collected retrospectively from medical records, including demographic information, clinical characteristics, and details of the drainage procedure. Descriptive statistics were used to summarize baseline characteristics. Continuous variables were expressed as mean ± standard deviation (SD), while categorical variables were presented as frequencies and percentages. Statistical analysis was performed using Microsoft Excel® (Version 2505; Microsoft Corp., Redmond, WA).
3. Results
3.1. Demographic and Clinical Characteristics
Table 1 summarizes the demographic and clinical characteristics of the cohort. A total of 212 consecutive patients received lumbar cistern drainage (LCD) between January 2017 and December 2023. After applying exclusion criteria (unsuccessful placement: n=8; mortality unrelated to LCD: n=2; lost to follow-up: n=11; catheter dislodgement: n=10; non-Medtronic® systems: n=2), 179 patients comprised the analytical cohort. Catheter obstruction occurred in 64 patients (35.8%). 17 (9.5%) underwent uneventful catheter removal following 24-hour observation without neurological symptoms. The remaining 47 patients (26.3%) required intervention: 19 underwent Group A (Catheter Shortening Protocol) and 28 received Group B (Routine Management Protocol). Within this intervention cohort (n=47), 26 were male (55.3%) and 21 female (44.7%). Primary indications were: central nervous system (CNS) infections requiring adjunctive drainage (n=37, 78.7%), subarachnoid hemorrhage (SAH) necessitating diversion (n=2, 4.3%), post-operative subcutaneous fluid accumulation (n=5, 10.6%), prevention of spinal cord ischemia after aortic surgery (n=1, 2.1%), and cerebrospinal fluid (CSF) leakage (n=2, 4.3%).
Table. 1. Demographic and clinical characteristics.
|
Variable |
Shortening Catheter Management Protocol |
Routine Catheter Management Protocol |
|
|
No. of patients |
47 |
19 |
28 |
|
Age (years) |
44.89±16.64 |
47.43±16.67 |
|
|
Sex |
Male |
11(57.9) |
15(53.6) |
|
Female |
8(42.1) |
13(46.4) |
|
|
Reason for LD insertion |
CNS infections requiring adjunctive drainage therapy |
12(63.1) |
25(89.2) |
|
SAH necessitating cerebrospinal fluid diversion |
1(5.3) |
1(3.6) |
|
|
Persistent post-operative subcutaneous fluid accumulation
following craniotomy or spinal surgery |
4(21.0) |
1(3.6) |
|
|
Prevention of spinal cord ischemia after aortic
surgery |
1(5.3) |
0 |
|
|
CSF leakage managed with therapeutic drainage |
1(5.3) |
1(3.6) |
|
|
Primary disease |
Brain tumor |
11(57.8) |
19(67.8) |
|
Cerebrospinal fluid leak |
1(5.3) |
1(3.6) |
|
|
Aortic dissection |
1(5.3) |
0 |
|
|
Chiari malformation |
2(10.5) |
1(3.6) |
|
|
Cerebral hemorrhage |
3(15.8) |
7(25.0) |
|
|
Spinal canal tumor |
1(5.3) |
0 |
|
|
Lesion location |
Supratentorial |
10(52.6) |
20(71.4) |
|
Subtentorial |
7(36.8) |
8(28.6) |
|
|
Spinal canal |
1(5.3) |
0 |
|
|
Others |
1(5.3) |
0 |
|
3.2. Group A (Catheter Shortening Protocol) Outcomes
Group A (n=19) underwent 22 shortening procedures. Primary patency was achieved in 17 patients (89.5%) without recurrent obstruction. One patient (5.3%) required a second shortening procedure for recurrent obstruction, achieving successful recanalization. One patient (5.3%) with CSF rhinorrhea-associated meningitis required three shortening procedures; all resolved obstructions successfully, yielding a final catheter length of 12.3 cm. Complete recanalization was achieved in all 22 procedures (100%). The mean residual catheter length post-recanalization was 34.89 ± 9.05 cm (mean ± SD). Longitudinal sectioning of retrieved catheters revealed wall-adherent deposits in all cases.
3.3. Group B (Routine Management Protocol) Outcomes
Group B (n=28) underwent 38 interventions (34 catheter replacements, 4 EVDs), representing a mean reintervention rate of 1.36 (38/28) procedures per patient. Obstruction recurrence distribution was: single episode (n=19, 67.9%), two episodes (n=6, 21.4%), and three episodes (n=3, 10.7%), totaling 40 obstruction events. Two patients (7.1%) had catheters retained without intervention after second obstructions; both were removed following asymptomatic 24-hour observation periods. Ultimately, 24 patients (85.7%) successfully completed treatment with LCD, while 4 (14.3%) required permanent conversion to EVD (Table 2).
Table. 2. Group B (Routine Management Protocol) outcomes.
|
Occlusion Episodes |
Cases |
Management Approach |
Cases |
|
once |
19(67.9) |
Catheter replacement |
17 |
|
EVD |
2 |
||
|
twice |
6(21.4) |
Catheter replacement twice |
2 |
|
First: Catheter replacement; Second: Observation followed by removal |
2 |
||
|
First: Catheter replacement; Second: EVD* |
2 |
||
|
three times |
3(10.7) |
Catheter replacement (three times) |
3 |
Group A demonstrated recurrent obstruction in 2/19 patients (10.5%). Among Group B patients requiring post-obstruction catheter replacement (n=28), secondary obstruction occurred in 9/28 cases (32.1%). Secondary obstruction rates differed significantly between Group A and Group B (10.5% vs 32.1%; χ²=3.95, p=0.047).
4. Discussion
Continuous lumbar cistern drainage (LCD) constitutes a frequently performed neurosurgical intervention. Its therapeutic mechanism relies on controlled cerebrospinal fluid (CSF) diversion. Catheter obstruction represents the predominant mechanical complication of LCD, with reported incidence rates exceeding 30% [7]. Obstruction necessitates immediate intervention, as therapeutic interruption can precipitate rapid clinical deterioration. Conventional management involves either catheter reinsertion or conversion to external ventricular drainage (EVD). Repeated insertion attempts elevate risks of dural trauma, adjacent tissue injury, procedural infections, and neurological sequelae, while prolonging intervention time and patient discomfort [8]. These factors collectively diminish patient satisfaction and potentially escalate clinician-patient discord.
In this retrospective controlled study, the catheter shortening technique (Group A) achieved 100% catheter recanalization while demonstrating non-invasive preservation of the original dural access. By contrast, the conventional management cohort (Group B) required a mean of 1.36 ± 0.64 additional invasive interventions per patient, with 85.7% undergoing repeated lumbar punctures for catheter replacement and 14.3% requiring conversion to external ventricular drainage (EVD)—a procedure carrying higher infection risks (5-20% incidence) 9and neurological complications [10]. Critically, the shortening method demonstrated a 69% relative reduction in secondary obstruction rates compared to catheter replacement (10.5% vs 32.1%; RR 0.31, 95% CI 0.08-0.83; p=0.047). These findings establish catheter shortening as an effective minimally invasive salvage technique for drainage catheter obstruction, offering statistically and clinically significant advantages over conventional approaches.
According to our research, the obstruction typically occurs in the middle section of the drainage catheter, and we conducted further analysis. A recent study determined that the protein concentration of cerebrospinal fluid in the lumbar cistern of adults was 52.2±18.4 mg/dL [11]. The protein concentration of cerebrospinal fluid in the lumbar cistern is elevated in almost all cases of bacterial meningitis and subarachnoid hemorrhage [12]. During the process of CSF drainage from the body to the external environment, the temperature of the CSF rapidly decreases from body temperature (around 37°C) to room temperature (around 20°C). As the temperature drops, proteins with increased concentrations in the CSF begin to precipitate. Meanwhile, the elevation in protein concentration leads to an increase in viscosity. According to Poiseuille's law [13] as Equation 1:
RT = 8Lμ/ρgr4 ---(1)
Fluid resistance is directly proportional to viscosity, thus the increase in viscosity results in an elevated resistance to flow, causing a decrease in the flow velocity of the CSF within the drainage tube. Consequently, the precipitated proteins are more likely to adhere to the inner walls of the drainage catheter, gradually leading to obstruction formation. When the drainage catheter is shortened and connected to an extension catheter with a larger diameter at the distal end, the resistance of the drainage system decreases, and the likelihood of re-blockage also reduces.
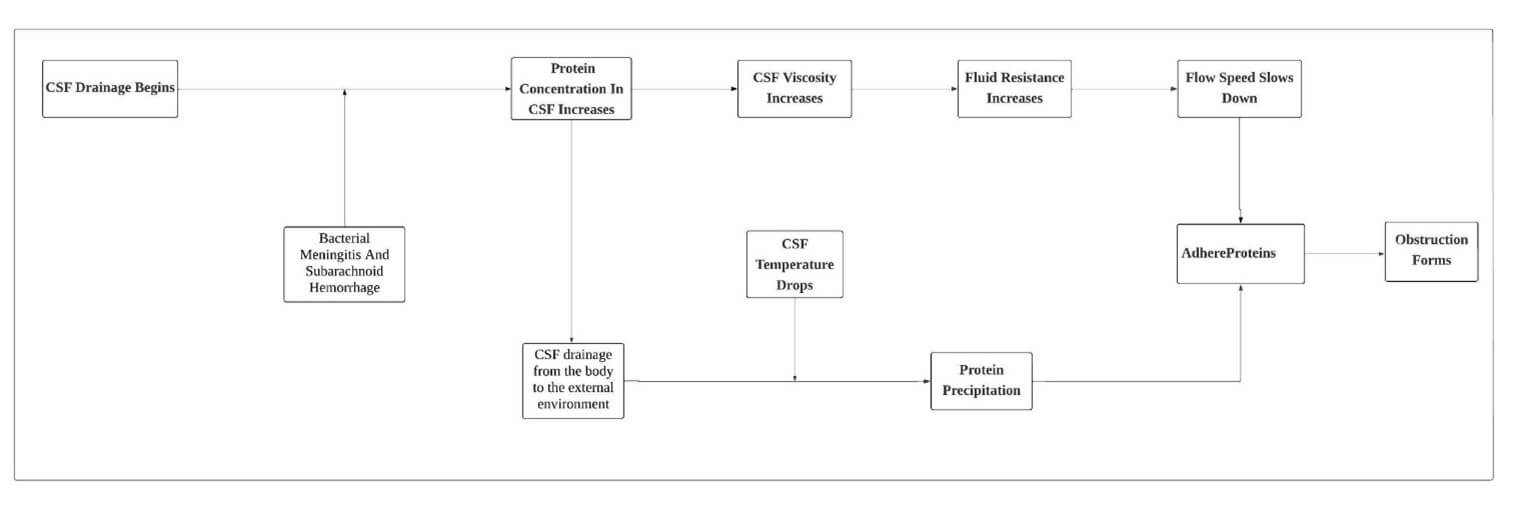
Catheter obstruction represents the predominant mechanical complication in lumbar drainage systems. Zheng M et al. reported a blockage rate of 13.7%, with extended drainage duration (≥7 days), low CSF output (<200 mL/day), CSF leakage, and abnormal CSF properties were predictors of lumbar cistern blockage [14]. Similarly, Pandit et al. reported obstruction in 21.3% of cases [7]. In cases of drainage catheter obstruction (slower drainage speed) or blockage, methods such as lowering the height of the titration catheter, adjusting the patient's position, repeatedly flushing with sterile saline, using negative pressure suction with a syringe, or removing part of the drainage catheter can be attempted. Some catheters may clear, but a significant proportion of the drainage catheters remain blocked. Based on our literature review, aside from replacing the drainage catheter and switching to external ventricular drainage, earlier authors appear not to have proposed better solutions for resolving the issue of catheter blockage. The shortening method we proposed is a new, simple, and effective approach.
Several limitations warrant consideration: 1. The single-center retrospective design introduces potential selection bias despite consecutive patient enrollment. 2. Modest cohort size (n=47 intervention cases) limits subgroup analyses such as indication-specific efficacy evaluation. 3. While we established 100% technical success for patency restoration, clinical outcomes (e.g., infection rates, neurological recovery, hospital stay duration) require prospective assessment. 4. Exclusive use of Medtronic® EDM™ catheters limits extrapolation to other systems with differing material properties (silicone vs. polyurethane) and internal diameters.
5. Conclusions
In summary, catheter obstruction constitutes a frequent mechanical complication during continuous lumbar cistern drainage (LCD). The routine treatment is to replace the drainage tube, and during the course of treatment, patients may experience repeated blockages, requiring multiple replacements, or even a shift to external ventricular drainage. Each procedural escalation imposes added resource utilization, patient discomfort, and complication risks. Our shortening technique offers particular advantages for vulnerable populations including elderly, critically ill, and extended-duration drainage candidates.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request. Data underlying the results of this study are not publicly available due to privacy and ethical restrictions. For further information, please contact the corresponding author at huawang@email.sdfmu.edu.cn.
Conflicts of Interest
None.
Funding
This research was supported entirely by institutional resources from Shandong Provincial Hospital Affiliated to Shandong First Medical University. No external funding or financial support was received for the study.
Acknowledgments
Patient Participation and Ethical Oversight
We extend our deepest gratitude to the 47 patients who participated in this study, particularly those who underwent interventions for lumbar cistern drainage (LCD) catheter obstruction. Their willingness to contribute to this research was essential for evaluating the efficacy and safety of the catheter shortening method. We also acknowledge the Ethics Committee of Shandong Provincial Hospital Affiliated to Shandong First Medical University for their rigorous review and approval of the study protocol (reference details available in the Methods section), ensuring full compliance with the Declaration of Helsinki. Patient informed consent procedures were meticulously managed by the hospital's clinical team, facilitating ethical data collection.
Clinical Team and Technical Support
Special thanks are due to the neurosurgical and nursing staff at Shandong Provincial Hospital for their expert execution of the standardized LCD management protocol. Their diligent bihourly monitoring of drainage volumes, sterile dressing changes, and precise catheter adjustments were critical to maintaining study integrity. We also recognize Medtronic Inc. for providing the EDM™ Lumbar External Drainage and Monitoring System (specifically, the F14 Tuohy needle and lumbar puncture catheter described in the Equipment section), which enabled the consistent application of the shortening technique. Technical assistance extended to the use of Microsoft Excel® (Version 2505; Microsoft Corp.) for dataset curation and preliminary statistical analysis, underscoring its role in handling retrospective patient data.
Methodological and Reporting Guidance
We appreciate the foundational guidance of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (von Elm et al., 2014), which informed the design and reporting of this single-center, retrospective case-control study. The clinical research unit at Shandong Provincial Hospital provided valuable insights during data interpretation, helping contextualize our findings on re-obstruction rates and procedural burden reduction.
Conflict of Interest Declaration
All authors declare no conflicts of interest related to this work. Medtronic Inc. had no involvement in the study design, data analysis, manuscript preparation, or decision to publish, and their equipment was used solely as part of routine clinical practice. Funding for this research was derived entirely from institutional resources, with no external financial support influencing the outcomes or conclusions.
Supplementary Materials
The supplementary materials for this study include additional data and figures that support the findings of the research. These materials are provided in the form of datasets and detailed images that further illustrate the results.
Figures 1-5: Comprehensive image analysis of catheter shortening procedures and associated changes in cerebrospinal fluid (CSF) flow.
Table 1: Detailed patient demographic information, including age, gender, and medical history of the 47 patients involved in the study.
Table 2: Raw data for the primary outcomes, including catheter patency restoration rates and re-obstruction events.
These supplementary materials are referenced at the appropriate points within the manuscript for further clarification and in-depth analysis.
REFERENCES
[1] G VOURC'H “Continuous
cerebrospinal fluid drainage by indwelling spinal catheter.” Br J Anaesth, vol. 35, pp. 118-120,
1963. View at: Publisher
Site | PubMed
[2] Stefan Wolf “Rationale for
lumbar drains in aneurysmal subarachnoid hemorrhage.” Curr Opin Crit Care, vol. 21, no. 2, pp. 120-126, 2015. View at: Publisher Site | PubMed
[3] Q-H Chen, D Lin, Q-G Yu,
et al. “Efficacy of lumbar cistern drainage combined with intrathecal
antibiotherapy for the treatment of ventriculo-subarachnoid infections
following surgery for hypertensive intracerebral hemorrhage.” Neurochirurgie, vol. 63, no. 1, pp.
13-16, 2017. View at: Publisher
Site | PubMed
[4] Nathan T Zwagerman, Eric W
Wang, Samuel S Shin, et al. “Does lumbar drainage reduce postoperative
cerebrospinal fluid leak after endoscopic endonasal skull base surgery? A
prospective, randomized controlled trial.” J
Neurosurg, vol. 131, no. 4, pp. 1172-1178, 2018. View at: Publisher Site | PubMed
[5] Lelio Guida, Alissa
Visentin, Sandro Benichi, et al. “External lumbar drainage for the management
of refractory intracranial hypertension in pediatric severe traumatic brain
injury: a retrospective single-center case series.” J Neurosurg Pediatr, vol. 34, no. 6, pp. 566-573, 2024. View at: Publisher Site | PubMed
[6] Jetan Badhiwala, Angela
Lumba-Brown, Gregory W J Hawryluk, et al. “External Lumbar Drainage following
Traumatic Intracranial Hypertension: A Systematic Review and Meta-Analysis.” Neurosurgery, vol. 89, no. 3, pp.
395-405, 2021. View at: Publisher
Site | PubMed
[7] Anand S Pandit, Joanna
Palasz, Parashkev Nachev, et al. “Mechanical Complications of External
Ventricular and Lumbar Drains.” World
Neurosurg, vol. 166, pp. e140-e154, 2022. View at: Publisher Site | PubMed
[8] Saqib Kamran Bakhshi,
Nawal Suhail, Rida Mitha, et al. “Lumbar Drain for Temporary Cerebrospinal
Fluid Diversion: Factors Related to the Risks of Complications at a University
Hospital.” World Neurosurg, vol. 143,
pp. e193-e198, 2020. View at: Publisher Site | PubMed
[9] H Humphreys, P J Jenks
“Surveillance and management of ventriculitis following neurosurgery.” J Hosp Infect, vol. 89, no. 4, pp.
281-286, 2015. View at: Publisher
Site | PubMed
[10] Ayoub Dakson, Michelle
Kameda-Smith, Michael D Staudt, et al. “A nationwide prospective multicenter
study of external ventricular drainage: accuracy, safety, and related
complications.” J Neurosurg, vol.
137, no. 1, pp. 249-257, 2021. View at: Publisher Site | PubMed
[11] Kalli J Fautsch, Darci R
Block, Jonathan Graff-Radford, et al. “Population-Based Evaluation of Total
Protein in Cerebrospinal Fluid.” Mayo
Clin Proc, vol. 98, no. 2, pp. 239-251, 2023. View at: Publisher Site | PubMed
[12] Brian Shahan, Edwin Y
Choi, Gilberto Nieves “Cerebrospinal Fluid Analysis.” Am Fam Physician, vol. 103, no. 7, pp. 422-428, 2021. View at: PubMed
[13] H H.
Richard Winn “YOUMANS & WINN
NEUROLOGICAL SURGERY.” EIGHTH EDITION ed. Elsevier; 2023.
[14] Min Zheng, Qilong Tian, Xuejiao Wang, et al. “Analysis of risk factors and treatment strategies for lumbar cistern blockage after craniocerebral surgery.” Front Neurosci, vol. 17, pp. 1124395, 2023. View at: Publisher Site | PubMed
